5841:
5498:, referred to by many since as the "father of the chemical bond". Pauling also considered the nature of the interatomic forces in metals, and concluded that about half of the five d-orbitals in the transition metals are involved in bonding, with the remaining nonbonding d-orbitals being responsible for the magnetic properties. Pauling was therefore able to correlate the number of d-orbitals in bond formation with the bond length, as well as with many of the physical properties of the substance. He subsequently introduced the metallic orbital, an extra orbital necessary to permit uninhibited resonance of valence bonds among various electronic structures.
5821:
179:) along the cell edges, measured from a reference point. It is thus only necessary to report the coordinates of a smallest asymmetric subset of particles, called the crystallographic asymmetric unit. The asymmetric unit may be chosen so that it occupies the smallest physical space, which means that not all particles need to be physically located inside the boundaries given by the lattice parameters. All other particles of the unit cell are generated by the symmetry operations that characterize the symmetry of the unit cell. The collection of symmetry operations of the unit cell is expressed formally as the
5251:
7622:
2052:
214:
202:
5873:
2243:
2233:
2223:
5696:} units linked together by shared vertices in different arrangements. In different minerals the tetrahedra show different degrees of networking and polymerization. For example, they occur singly, joined in pairs, in larger finite clusters including rings, in chains, double chains, sheets, and three-dimensional frameworks. The minerals are classified into groups based on these structures. In each of the 7 thermodynamically stable crystalline forms or polymorphs of crystalline quartz, only 2 out of 4 of each the edges of the {SiO
5505:, the factors that determine the choice of one from among alternative crystal structures of a metal or intermetallic compound revolve around the energy of resonance of bonds among interatomic positions. It is clear that some modes of resonance would make larger contributions (be more mechanically stable than others), and that in particular a simple ratio of number of bonds to number of positions would be exceptional. The resulting principle is that a special stability is associated with the simplest ratios or "bond numbers":
2279:
2185:
5567:
group number. The operation of directional forces were emphasized in one article on the relation between bond hybrids and the metallic structures. The resulting correlation between electronic and crystalline structures is summarized by a single parameter, the weight of the d-electrons per hybridized metallic orbital. The "d-weight" calculates out to 0.5, 0.7 and 0.9 for the fcc, hcp and bcc structures respectively. The relationship between d-electrons and crystal structure thus becomes apparent.
1607:
5888:
2335:
5903:
5801:, in that, in the absence of an electric field during production, the ferroelectric crystal does not exhibit a polarization. Upon the application of an electric field of sufficient magnitude, the crystal becomes permanently polarized. This polarization can be reversed by a sufficiently large counter-charge, in the same way that a ferromagnet can be reversed. However, although they are called ferroelectrics, the effect is due to the crystal structure (not the presence of a ferrous metal).
2387:
2377:
452:
190:
31:
5861:
293:, or some multiple thereof. That is, the Miller indices are proportional to the inverses of the intercepts of the plane with the unit cell (in the basis of the lattice vectors). If one or more of the indices is zero, it means that the planes do not intersect that axis (i.e., the intercept is "at infinity"). A plane containing a coordinate axis is translated so that it no longer contains that axis before its Miller indices are determined. The Miller indices for a plane are
5358:
5302:
5214:
230:
8015:
5915:
8199:
8157:
2213:
2308:
2267:
2175:
2047:{\displaystyle {\frac {1}{d^{2}}}={\frac {{\frac {h^{2}}{a^{2}}}\sin ^{2}\alpha +{\frac {k^{2}}{b^{2}}}\sin ^{2}\beta +{\frac {\ell ^{2}}{c^{2}}}\sin ^{2}\gamma +{\frac {2k\ell }{bc}}(\cos \beta \cos \gamma -\cos \alpha )+{\frac {2h\ell }{ac}}(\cos \gamma \cos \alpha -\cos \beta )+{\frac {2hk}{ab}}(\cos \alpha \cos \beta -\cos \gamma )}{1-\cos ^{2}\alpha -\cos ^{2}\beta -\cos ^{2}\gamma +2\cos \alpha \cos \beta \cos \gamma }}}
5580:
4896:
8152:
8132:
2145:
8027:
6892:
8142:
8122:
8112:
5931:
2365:
8219:
8209:
8189:
8168:
1596:
1275:
2074:
in three directions, but some have other symmetry elements as well. For example, rotating the crystal 180° about a certain axis may result in an atomic configuration that is identical to the original configuration; the crystal has twofold rotational symmetry about this axis. In addition to rotational
5482:
The difficulty of predicting stable crystal structures based on the knowledge of only the chemical composition has long been a stumbling block on the way to fully computational materials design. Now, with more powerful algorithms and high-performance computing, structures of medium complexity can be
5570:
In crystal structure predictions/simulations, the periodicity is usually applied, since the system is imagined as being unlimited in all directions. Starting from a triclinic structure with no further symmetry property assumed, the system may be driven to show some additional symmetry properties by
5455:
Grain boundaries are in general only a few nanometers wide. In common materials, crystallites are large enough that grain boundaries account for a small fraction of the material. However, very small grain sizes are achievable. In nanocrystalline solids, grain boundaries become a significant volume
5566:
analyzed the trends in melting points, compressibilities and bond lengths as a function of group number in the periodic table in order to establish a system of valencies of the transition elements in the metallic state. This treatment thus emphasized the increasing bond strength as a function of
5428:
cut into two parts, one of which is rotated, we see that there are five variables required to define a grain boundary. The first two numbers come from the unit vector that specifies a rotation axis. The third number designates the angle of rotation of the grain. The final two numbers specify the
4966:
One important characteristic of a crystalline structure is its atomic packing factor (APF). This is calculated by assuming that all the atoms are identical spheres, with a radius large enough that each sphere abuts on the next. The atomic packing factor is the proportion of space filled by these
2482:
A crystal system is a set of point groups in which the point groups themselves and their corresponding space groups are assigned to a lattice system. Of the 32 point groups that exist in three dimensions, most are assigned to only one lattice system, in which case the crystal system and lattice
5416:
Grain boundaries are interfaces where crystals of different orientations meet. A grain boundary is a single-phase interface, with crystals on each side of the boundary being identical except in orientation. The term "crystallite boundary" is sometimes, though rarely, used. Grain boundary areas
2075:
symmetry, a crystal may have symmetry in the form of mirror planes, and also the so-called compound symmetries, which are a combination of translation and rotation or mirror symmetries. A full classification of a crystal is achieved when all inherent symmetries of the crystal are identified.
5777:
polarization when an electric field is applied, but a substance that has such a natural charge separation even in the absence of a field is called a polar material. Whether or not a material is polar is determined solely by its crystal structure. Only 10 of the 32 point groups are
2453:, describe the geometric arrangement of the lattice points, and therefore the translational symmetry of the crystal. The three dimensions of space afford 14 distinct Bravais lattices describing the translational symmetry. All crystalline materials recognized today, not including
4947:
The unit cell of a ccp arrangement of atoms is the face-centered cubic (fcc) unit cell. This is not immediately obvious as the closely packed layers are parallel to the {111} planes of the fcc unit cell. There are four different orientations of the close-packed layers.
2464:, positioned around each and every lattice point. This group of atoms therefore repeats indefinitely in three dimensions according to the arrangement of one of the Bravais lattices. The characteristic rotation and mirror symmetries of the unit cell is described by its
5731:
respectively. Two more allotropes, γ and σ, exist at temperatures above 161 °C and pressures above several GPa. White tin is metallic, and is the stable crystalline form at or above room temperature. Below 13.2 °C, tin exists in the gray form, which has a
1029:
4907:
atomic planes in three dimensions. For example, if plane A lies beneath plane B, there are two possible ways of placing an additional atom on top of layer B. If an additional layer were placed directly over plane A, this would give rise to the following series:
2483:
system both have the same name. However, five point groups are assigned to two lattice systems, rhombohedral and hexagonal, because both lattice systems exhibit threefold rotational symmetry. These point groups are assigned to the trigonal crystal system.
137:
Crystal structure is described in terms of the geometry of arrangement of particles in the unit cells. The unit cell is defined as the smallest repeating unit having the full symmetry of the crystal structure. The geometry of the unit cell is defined as a
1407:
5081:
1044:
5337:
impurities in certain materials. Research on magnetic impurities demonstrates that substantial alteration of certain properties such as specific heat may be affected by small concentrations of an impurity, as for example impurities in semiconducting
5269:
Interstitial sites refer to the empty spaces in between the atoms in the crystal lattice. These spaces can be filled by oppositely charged ions to form multi-element structures. They can also be filled by impurity atoms or self-interstitials to form
1396:
5752:
applications. Although the α–β transformation temperature of tin is nominally 13.2 °C, impurities (e.g. Al, Zn, etc.) lower the transition temperature well below 0 °C, and upon addition of Sb or Bi the transformation may not occur at all.
6032:– (for elements that are solid at standard temperature and pressure) gives the crystalline structure of the most thermodynamically stable form(s) in those conditions. In all other cases the structure given is for the element at its melting point.
4880:
By considering the arrangement of atoms relative to each other, their coordination numbers, interatomic distances, types of bonding, etc., it is possible to form a general view of the structures and alternative ways of visualizing them.
893:
447:: The condensation of a material means that the atoms, ions or molecules are more stable if they are surrounded by other similar species. The surface tension of an interface thus varies according to the density on the surface.
5571:
applying Newton's Second Law on particles in the unit cell and a recently developed dynamical equation for the system period vectors (lattice parameters including angles), even if the system is subject to external stress.
4418:
4336:
4254:
3938:
3856:
3774:
3478:
3391:
3309:
784:
4108:
4026:
3692:
621:
2986:
2908:
2827:
5332:
When one atom substitutes for one of the principal atomic components within the crystal structure, alteration in the electrical and thermal properties of the material may ensue. Impurities may also manifest as
5288:
Real crystals feature defects or irregularities in the ideal arrangements described above and it is these defects that critically determine many of the electrical and mechanical properties of real materials.
4927:
If, however, all three planes are staggered relative to each other and it is not until the fourth layer is positioned directly over plane A that the sequence is repeated, then the following sequence arises:
2083:
Lattice systems are a grouping of crystal structures according to the point groups of their lattice. All crystals fall into one of seven lattice systems. They are related to, but not the same as the seven
386:
4788:
is the mathematical group comprising the symmetry operations that leave at least one point unmoved and that leave the appearance of the crystal structure unchanged. These symmetry operations include
4482:
4765:
4600:
4172:
3542:
3227:
508:
is the plane perpendicular to the principal axis in these crystal systems. For triclinic, orthorhombic, and cubic crystal systems the axis designation is arbitrary and there is no principal axis.
3051:
904:
7631:
6970:
1591:{\displaystyle {\frac {1}{d^{2}}}=\left({\frac {h^{2}}{a^{2}}}+{\frac {k^{2}\sin ^{2}\beta }{b^{2}}}+{\frac {\ell ^{2}}{c^{2}}}-{\frac {2h\ell \cos \beta }{ac}}\right)\csc ^{2}\beta }
5962:
5641:. According to Gibbs' rules of phase equilibria, these unique crystalline phases are dependent on intensive variables such as pressure and temperature. Polymorphism is related to
1270:{\displaystyle {\frac {1}{d^{2}}}={\frac {(h^{2}+k^{2}+\ell ^{2})\sin ^{2}\alpha +2(hk+k\ell +h\ell )(\cos ^{2}\alpha -\cos \alpha )}{a^{2}(1-3\cos ^{2}\alpha +2\cos ^{3}\alpha )}}}
665:(bcc) lattices, the primitive lattice vectors are not orthogonal. However, in these cases the Miller indices are conventionally defined relative to the lattice vectors of the cubic
496:
Some directions and planes are defined by symmetry of the crystal system. In monoclinic, trigonal, tetragonal, and hexagonal systems there is one unique axis (sometimes called the
5398:
at lower stress than that needed for a perfect crystal structure. The local stress fields result in interactions between the dislocations which then result in strain hardening or
4973:
7892:
8058:
5559:(which determines the relative bond lengths) are thus a result of the effort of an atom to use its valency in the formation of stable bonds with simple fractional bond numbers.
1286:
7887:
4701:
4649:
4536:
3605:
3163:
3111:
2750:
2693:
2633:
2576:
5464:. In the limit of small crystallites, as the volume fraction of grain boundaries approaches 100%, the material ceases to have any crystalline character, and thus becomes an
5444:
of the material. The high interfacial energy and relatively weak bonding in most grain boundaries often makes them preferred sites for the onset of corrosion and for the
5086:
Another important characteristic of a crystalline structure is its coordination number (CN). This is the number of nearest neighbours of a central atom in the structure.
7427:
7943:
2070:
The defining property of a crystal is its inherent symmetry. Performing certain symmetry operations on the crystal lattice leaves it unchanged. All crystals have
420:
linking nodes. Some directions and planes have a higher density of nodes. These high density planes have an influence on the behavior of the crystal as follows:
795:
6963:
5432:
Grain boundaries disrupt the motion of dislocations through a material, so reducing crystallite size is a common way to improve strength, as described by the
5661:. Polymorphs have different stabilities and may spontaneously and irreversibly transform from a metastable form (or thermodynamically unstable form) to the
7171:
653:
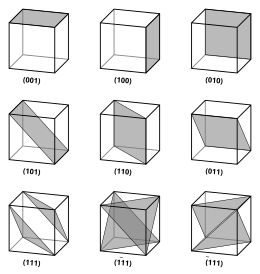
such as {100} denote a family of plane normals that are equivalent due to symmetry operations, much the way angle brackets denote a family of directions.
6956:
441:: Physical adsorption and chemical reactions occur at or near surface atoms or molecules. These phenomena are thus sensitive to the density of nodes.
301:
3). In an orthogonal coordinate system for a cubic cell, the Miller indices of a plane are the
Cartesian components of a vector normal to the plane.
66:. Ordered structures occur from intrinsic nature of constituent particles to form symmetric patterns that repeat along the principal directions of
626:
Because of the symmetry of cubic crystals, it is possible to change the place and sign of the integers and have equivalent directions and planes:
4364:
4282:
4200:
3884:
3802:
3720:
3424:
3337:
3255:
700:
4054:
3972:
3638:
545:
7761:
6913:
2939:
2861:
2780:
6796:
Molodets, A. M.; Nabatov, S. S. (2000). "Thermodynamic
Potentials, Diagram of State, and Phase Transitions of Tin on Shock Compression".
7948:
7673:
8065:
7839:
7131:
5201:
The 74% packing efficiency of the FCC and HCP is the maximum density possible in unit cells constructed of spheres of only one size.
4903:
The principles involved can be understood by considering the most efficient way of packing together equal-sized spheres and stacking
6928:
4967:
spheres which can be worked out by calculating the total volume of the spheres and dividing by the volume of the cell as follows:
492:) is along a dense direction. The shift of one node in a more dense direction requires a lesser distortion of the crystal lattice.
77:
of the structure. The unit cell completely reflects symmetry and structure of the entire crystal, which is built up by repetitive
326:
6923:
2410:) with respect to each other. These threefold axes lie along the body diagonals of the cube. The other six lattice systems, are
7938:
7930:
6611:
Hume-rothery, W.; Irving, H. M.; Williams, R. J. P. (1951). "The
Valencies of the Transition Elements in the Metallic State".
5748:. Gray tin has no metallic properties at all, is a dull gray powdery material, and has few uses, other than a few specialized
4443:
7991:
7969:
6873:
6848:
6656:
Altmann, S. L.; Coulson, C. A.; Hume-Rothery, W. (1957). "On the
Relation between Bond Hybrids and the Metallic Structures".
6459:
6197:
6111:
4726:
4561:
4133:
3503:
3188:
67:
2457:, fit in one of these arrangements. The fourteen three-dimensional lattices, classified by lattice system, are shown above.
7984:
7834:
7500:
7365:
7214:
7067:
5820:
4772:
In total there are seven crystal systems: triclinic, monoclinic, orthorhombic, tetragonal, trigonal, hexagonal, and cubic.
7974:
7872:
7568:
6896:
3019:
6918:
5692:, the Si atom shows tetrahedral coordination by 4 oxygens. All but one of the crystalline forms involve tetrahedral {SiO
2477:
1024:{\displaystyle {\frac {1}{d^{2}}}={\frac {4}{3}}\left({\frac {h^{2}+hk+k^{2}}{a^{2}}}\right)+{\frac {\ell ^{2}}{c^{2}}}}
7221:
6658:
6613:
6574:
6356:
6297:
6261:
6229:
6029:
516:
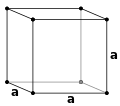
For the special case of simple cubic crystals, the lattice vectors are orthogonal and of equal length (usually denoted
642:
of directions that are equivalent due to symmetry operations, such as , , or the negative of any of those directions.
7996:
7854:
7824:
7753:
6086:
6701:
Liu, Gang (2015). "Dynamical equations for the period vectors in a periodic system under constant external stress".
5394:
Dislocations in a crystal lattice are line defects that are associated with local stress fields. Dislocations allow
108:. All possible symmetric arrangements of particles in three-dimensional space may be described by 230 space groups.
8031:
7706:
5629:
is the occurrence of multiple crystalline forms of a material. It is found in many crystalline materials including
5626:
5840:
5562:
After postulating a direct correlation between electron concentration and crystal structure in beta-phase alloys,
7979:
7902:
7776:
7375:
5490:
The crystal structures of simple ionic solids (e.g., NaCl or table salt) have long been rationalized in terms of
2242:
2232:
2222:
17:
7814:
7736:
6329:
5502:
4890:
213:
201:
154:) and the angles between them (α, β, γ). The positions of particles inside the unit cell are described by the
7829:
7819:
7124:
6943:
5076:{\displaystyle \mathrm {APF} ={\frac {N_{\mathrm {particle} }V_{\mathrm {particle} }}{V_{\text{unit cell}}}}}
2278:
488:
glide occurs preferentially parallel to higher density planes. The perturbation carried by the dislocation (
8240:
8090:
7953:
7601:
7226:
7204:
6144:, G.L. Trigg, VHC publishers, 1991, ISBN (Verlagsgesellschaft) 3-527-26954-1, ISBN (VHC Inc.) 0-89573-752-3
6008:
6002:
5477:
4836:. There are 32 possible crystal classes. Each one can be classified into one of the seven crystal systems.
4781:
2465:
1391:{\displaystyle {\frac {1}{d^{2}}}={\frac {h^{2}}{a^{2}}}+{\frac {k^{2}}{b^{2}}}+{\frac {\ell ^{2}}{c^{2}}}}
8127:
7505:
7259:
7154:
2833:
2419:
111:
The crystal structure and symmetry play a critical role in determining many physical properties, such as
5369:
5313:
5225:
4832:
Rotation axes (proper and improper), reflection planes, and centers of symmetry are collectively called
2184:
8250:
7862:
7159:
6407:
Zhang, X. Y.; Suhl, H (1985). "Spin-wave-related period doublings and chaos under transverse pumping".
4677:
4625:
4512:
3581:
3139:
3087:
2726:
2669:
2609:
2552:
8137:
8117:
7877:
7806:
7264:
7254:
5987:
5723:
that are encountered at normal pressure and temperature, α-tin and β-tin, are more commonly known as
5445:
3057:
2639:
116:
5436:
relationship. Since grain boundaries are defects in the crystal structure they tend to decrease the
8245:
8147:
8106:
8051:
8019:
7743:
7639:
7512:
7475:
7390:
7269:
7249:
7117:
5192:
5182:
3548:
2524:
2411:
2386:
2376:
2291:
7867:
7711:
7656:
7405:
7370:
5997:
5658:
5649:. The complete morphology of a material is described by polymorphism and other variables such as
5484:
5437:
5283:
2423:
461:
7621:
7563:
7380:
6154:
5992:
5790:
5250:
2399:
2071:
438:
189:
155:
78:
6908:
2504:
7920:
7716:
7678:
7485:
7437:
6249:
6047:
6024:
5104:
4957:
4612:
4499:
4187:
3956:
3707:
3565:
3242:
3071:
2848:
2653:
2538:
670:
525:
6903:
2212:
8162:
7644:
7517:
7353:
7244:
7093:
6948:
6805:
6768:
6721:
6667:
6622:
6583:
6515:
6416:
6381:
5461:
5441:
4488:
2517:
2351:
2334:
2106:
888:{\displaystyle {\frac {1}{d^{2}}}={\frac {h^{2}+k^{2}}{a^{2}}}+{\frac {\ell ^{2}}{c^{2}}}}
8:
8194:
7661:
7649:
7524:
7490:
7470:
5976:
5954:
5887:
5786:, so the 10 polar crystal classes are sometimes referred to as the pyroelectric classes.
5449:
5271:
5262:
5164:
5146:
4961:
4664:
4351:
4041:
3411:
3126:
666:
662:
658:
501:
481:
413:
6809:
6772:
6725:
6671:
6626:
6587:
6519:
6420:
6385:
5914:
5902:
2266:
2174:
8260:
7910:
7721:
7666:
7209:
7034:
6821:
6737:
6711:
6683:
6638:
6286:
6075:
6020:
5872:
5770:
5662:
5491:
2915:
475:
397:
317:
112:
73:
The smallest group of particles in material that constitutes this repeating pattern is
6938:
5700:} tetrahedra are shared with others, yielding the net chemical formula for silica: SiO
520:); similarly for the reciprocal lattice. So, in this common case, the Miller indices (
8255:
8214:
7844:
7683:
7611:
7591:
7311:
6980:
6869:
6844:
6825:
6480:
6455:
6432:
6352:
6325:
6293:
6257:
6225:
6193:
6107:
6082:
5950:
5779:
5670:
5646:
5245:
4824:
4507:
4269:
3959:
3789:
3568:
3324:
3074:
2926:
2713:
2656:
2541:
2407:
297:
with no common factors. Negative indices are indicated with horizontal bars, as in (1
6741:
6687:
6642:
5715:
when cooled. This change in mechanical properties due to existence of its two major
4848:
of the crystal structure contains translational symmetry operations. These include:
2307:
7882:
7688:
7606:
7596:
7395:
7328:
7299:
7292:
6813:
6776:
6729:
6675:
6630:
6591:
6550:
6523:
6488:
6424:
6389:
6185:
5766:
5448:
of new phases from the solid. They are also important to many of the mechanisms of
4868:, which reflect a point through a plane while translating it parallel to the plane.
2934:
2856:
2775:
2756:
529:
428:
89:
81:
of unit cell along its principal axes. The translation vectors define the nodes of
8184:
8085:
7771:
7766:
7731:
7551:
7450:
7385:
7348:
7343:
7194:
7140:
6933:
6346:
6256:(Reprint of WA Benjamin corrected 1975 ed.). Courier-Dover. pp. 70–71.
6005:– a notation to represent symmetry in point groups, plane groups and space groups
5681:
5654:
5465:
4716:
4672:
4620:
4551:
4433:
4123:
3871:
3625:
3493:
3178:
3009:
2767:
2596:
2512:
2446:
2403:
444:
82:
43:
35:
6126:
International Tables for
Crystallography (2006). Volume A, Space-group symmetry.
5417:
contain those atoms that have been perturbed from their original lattice sites,
8204:
8074:
7581:
7546:
7534:
7529:
7495:
7465:
7455:
7414:
7358:
7282:
7236:
7098:
6372:
Hogan, C. M. (1969). "Density of States of an
Insulating Ferromagnetic Alloy".
6141:
6044:– a small piece of a single crystal used to initiate growth of a larger crystal
6035:
6014:
5981:
5970:
5798:
5666:
5433:
5425:
5411:
4359:
4277:
4195:
3879:
3797:
3715:
3419:
3332:
3250:
2085:
2065:
489:
139:
120:
6781:
6756:
6478:(1929). "The principles determining the structure of complex ionic crystals".
6189:
5860:
2406:, that is, it exhibits four threefold rotational axes oriented at 109.5° (the
2364:
2144:
88:
The lengths of principal axes/edges, of unit cell and angles between them are
8234:
7726:
7539:
7338:
7072:
7027:
6703:
6475:
6428:
6213:
6017:– a state of matter with properties of both conventional liquids and crystals
5794:
5762:
5749:
5733:
5650:
5495:
5429:
plane of the grain boundary (or a unit vector that is normal to this plane).
5339:
5334:
5174:
5112:
4862:, which rotate a point around an axis while translating parallel to the axis.
6393:
7432:
7422:
7316:
7199:
6679:
6634:
6596:
6569:
6541:
Pauling, Linus (1947). "Atomic Radii and
Interatomic Distances in Metals".
6527:
6217:
6041:
5783:
5611:
5563:
5418:
5399:
5395:
5129:
4904:
4813:, which changes the sign of the coordinate of each point with respect to a
4049:
3967:
3633:
3576:
3134:
3082:
2721:
2664:
2604:
2454:
2427:
2199:
238:
142:, providing six lattice parameters taken as the lengths of the cell edges (
30:
6733:
6436:
6050:– a primitive cell of a crystal lattice with Voronoi decomposition applied
4413:{\displaystyle \mathbb {D} _{12}=\mathbb {Z} _{6}\rtimes \mathbb {Z} _{2}}
4331:{\displaystyle \mathbb {D} _{12}=\mathbb {Z} _{6}\rtimes \mathbb {Z} _{2}}
4249:{\displaystyle \mathbb {D} _{12}=\mathbb {Z} _{6}\rtimes \mathbb {Z} _{2}}
3933:{\displaystyle \mathbb {D} _{12}=\mathbb {Z} _{6}\rtimes \mathbb {Z} _{2}}
451:
27:
Ordered arrangement of atoms, ions, or molecules in a crystalline material
7915:
7586:
7460:
7287:
6944:
Crystallography Open
Database (with more than 140,000 crystal structures)
6716:
6506:
Pauling, Linus (1938). "The Nature of the
Interatomic Forces in Metals".
6038:– a repeating unit formed by the vectors spanning the points of a lattice
5587:
5556:
5389:
5121:
4845:
3851:{\displaystyle \mathbb {D} _{6}=\mathbb {Z} _{3}\rtimes \mathbb {Z} _{2}}
3769:{\displaystyle \mathbb {D} _{6}=\mathbb {Z} _{3}\rtimes \mathbb {Z} _{2}}
3473:{\displaystyle \mathbb {D} _{8}=\mathbb {Z} _{4}\rtimes \mathbb {Z} _{2}}
3386:{\displaystyle \mathbb {D} _{8}=\mathbb {Z} _{4}\rtimes \mathbb {Z} _{2}}
3304:{\displaystyle \mathbb {D} _{8}=\mathbb {Z} _{4}\rtimes \mathbb {Z} _{2}}
2498:
779:{\displaystyle {\frac {1}{d^{2}}}={\frac {h^{2}+k^{2}+\ell ^{2}}{a^{2}}}}
485:
478:: This typically occurs preferentially parallel to higher density planes.
469:
237:
Vectors and planes in a crystal lattice are described by the three-value
180:
105:
6570:"A Resonating-Valence-Bond Theory of Metals and Intermetallic Compounds"
6554:
6492:
5930:
5765:, and crystals belonging to one of these classes (point groups) display
5421:, and impurities that have migrated to the lower energy grain boundary.
5357:
5301:
5213:
5089:
The APFs and CNs of the most common crystal structures are shown below:
4103:{\displaystyle \mathbb {Z} _{6}=\mathbb {Z} _{3}\times \mathbb {Z} _{2}}
4021:{\displaystyle \mathbb {Z} _{6}=\mathbb {Z} _{3}\times \mathbb {Z} _{2}}
3687:{\displaystyle \mathbb {Z} _{6}=\mathbb {Z} _{3}\times \mathbb {Z} _{2}}
7480:
7166:
6817:
5958:
5949:
For more detailed information in specific technology applications, see
5774:
5619:
5345:
may lead to different properties as first predicted in the late 1960s.
5138:
2431:
2415:
2253:
2161:
472:
tend to have straight grain boundaries following higher density planes.
434:
229:
5456:
fraction of the material, with profound effects on such properties as
4828:, which consists of a rotation about an axis followed by an inversion.
4804:, which rotates the structure a specified portion of a circle about a
616:{\displaystyle d_{\ell mn}={\frac {a}{\sqrt {\ell ^{2}+m^{2}+n^{2}}}}}
7189:
5745:
5720:
5716:
5689:
5642:
5607:
5457:
2993:
2435:
2131:
1036:
465:
132:
74:
8198:
8156:
6843:(in German) (91–100 ed.). Walter de Gruyter. pp. 793–800.
2981:{\displaystyle \mathbb {V} =\mathbb {Z} _{2}\times \mathbb {Z} _{2}}
2903:{\displaystyle \mathbb {V} =\mathbb {Z} _{2}\times \mathbb {Z} _{2}}
2822:{\displaystyle \mathbb {V} =\mathbb {Z} _{2}\times \mathbb {Z} _{2}}
7786:
7556:
7304:
7051:
6904:
The internal structure of crystals... Crystallography for beginners
6081:. Manchester Physics Series (2nd ed.). John Wiley & Sons.
409:
101:
59:
8043:
7109:
431:
is directly related to density (or periodic density fluctuations).
7796:
7017:
5963:
List of biophysically important macromolecular crystal structures
5741:
5737:
5712:
5634:
5630:
5615:
294:
63:
6838:
5579:
4895:
8151:
8131:
6891:
6839:
Holleman, Arnold F.; Wiberg, Egon; Wiberg, Nils (1985). "Tin".
5973:– a primitive cell in the reciprocal space lattice of a crystal
5677:
5603:
5599:
5591:
5583:
2460:
The crystal structure consists of the same group of atoms, the
424:
5665:
form at a particular temperature. They also exhibit different
8141:
8121:
7791:
7044:
7039:
7022:
5638:
5424:
Treating a grain boundary geometrically as an interface of a
5342:
4920:
This arrangement of atoms in a crystal structure is known as
316:
between adjacent lattice planes is related to the (shortest)
8111:
6978:
6924:
6324:(3rd ed.). Washington, DC: American Chemical Society.
5156:
401:
381:{\displaystyle d={\frac {2\pi }{|\mathbf {g} _{hk\ell }|}}}
51:
6655:
6610:
6068:
6066:
6064:
5711:(Sn), which is malleable near ambient temperatures but is
7893:
Zeitschrift für Kristallographie – New Crystal Structures
6322:
Teaching General Chemistry: A Materials Science Companion
5708:
405:
55:
8218:
8208:
8188:
8167:
7888:
Zeitschrift für Kristallographie – Crystalline Materials
4477:{\displaystyle \mathbb {D} _{12}\times \mathbb {Z} _{2}}
6914:
Appendix A from the manual for Atoms, software for XAFS
6061:
4760:{\displaystyle \mathbb {S} _{4}\times \mathbb {Z} _{2}}
4595:{\displaystyle \mathbb {A} _{4}\times \mathbb {Z} _{2}}
4167:{\displaystyle \mathbb {Z} _{6}\times \mathbb {Z} _{2}}
3537:{\displaystyle \mathbb {D} _{8}\times \mathbb {Z} _{2}}
3222:{\displaystyle \mathbb {Z} _{4}\times \mathbb {Z} _{2}}
539:
between adjacent (ℓmn) lattice planes is (from above):
7781:
6795:
6757:"Review of the anatase to rutile phase transformation"
5804:
4844:
In addition to the operations of the point group, the
308:) planes intersecting one or more lattice points (the
233:
Planes with different Miller indices in cubic crystals
4976:
4729:
4680:
4628:
4564:
4515:
4446:
4367:
4285:
4203:
4136:
4057:
3975:
3887:
3805:
3723:
3641:
3584:
3506:
3427:
3340:
3258:
3191:
3142:
3090:
3022:
2942:
2864:
2783:
2729:
2672:
2612:
2555:
1610:
1410:
1289:
1047:
907:
798:
703:
548:
329:
6864:
Schwartz, Mel (2002). "Tin and Alloys, Properties".
5555:, etc. The choice of structure and the value of the
6120:
3046:{\displaystyle \mathbb {V} \times \mathbb {Z} _{2}}
260:) denotes a plane that intercepts the three points
6755:Hanaor, Dorian A. H.; Sorrell, Charles C. (2011).
6285:
6159:CSIC Dept de Cristalografia y Biologia Estructural
6074:
5075:
4899:The hpc lattice (left) and the ccf lattice (right)
4759:
4695:
4643:
4594:
4530:
4476:
4412:
4330:
4248:
4166:
4102:
4020:
3932:
3850:
3768:
3686:
3599:
3536:
3472:
3385:
3303:
3221:
3157:
3105:
3045:
2980:
2902:
2821:
2744:
2687:
2627:
2570:
2046:
1590:
1390:
1269:
1023:
887:
778:
615:
380:
4487:
3056:
2832:
2638:
2523:
104:properties of crystal are described byconcept of
8232:
6406:
6023:– a function used to solve the phase problem in
5789:There are a few crystal structures, notably the
4940:This type of structural arrangement is known as
2478:Crystallographic point group § Isomorphisms
524:) and both simply denote normals/directions in
6247:
6182:Electron Diffraction in the Electron Microscope
412:) of a crystal. Likewise, the crystallographic
320:vector orthogonal to the planes by the formula
6454:. Long Grove, IL: Waveland Press. p. 85.
6279:
6277:
6275:
6273:
6212:
6136:
6134:
6132:
5598:. The most important forms of silica include:
396:The crystallographic directions are geometric
8059:
7125:
6964:
6866:Encyclopedia of Materials, Parts and Finishes
6754:
6468:
2059:
6371:
5984:– a major stage of a crystallization process
7962:
6919:Intro to Minerals: Crystal Class and System
6270:
6129:
2402:or isometric system, has the symmetry of a
50:is a description of ordered arrangement of
8066:
8052:
7132:
7118:
6971:
6957:
6474:
5471:
6780:
6715:
6595:
5277:
4747:
4732:
4683:
4631:
4582:
4567:
4518:
4464:
4449:
4400:
4385:
4370:
4318:
4303:
4288:
4236:
4221:
4206:
4154:
4139:
4090:
4075:
4060:
4008:
3993:
3978:
3920:
3905:
3890:
3838:
3823:
3808:
3756:
3741:
3726:
3674:
3659:
3644:
3587:
3524:
3509:
3460:
3445:
3430:
3373:
3358:
3343:
3291:
3276:
3261:
3209:
3194:
3145:
3093:
3033:
3024:
2968:
2953:
2944:
2890:
2875:
2866:
2809:
2794:
2785:
2732:
2675:
2615:
2558:
6863:
6543:Journal of the American Chemical Society
6449:
6315:
6313:
6311:
6309:
6224:. Brooks/Cole (Thomson Learning, Inc.).
6179:
6072:
5578:
5249:
4894:
4795:, which reflects the structure across a
450:
391:
228:
29:
6567:
6540:
6505:
6250:"§4-2 Screw axes and §4-3 Glide planes"
6243:
6241:
6140:Encyclopedia of Physics (2nd Edition),
241:notation. This syntax uses the indices
14:
8233:
7235:
6344:
6320:Ellis, Arthur B.; et al. (1995).
6283:
5756:
4875:
3547:
676:
8047:
7113:
6952:
6319:
6306:
5761:Twenty of the 32 crystal classes are
5204:
4872:There are 230 distinct space groups.
3943:
2350:
2290:
2252:
2198:
2160:
2130:
2111:
2102:
2099:
2096:
38:(sodium in purple, chlorine in green)
8026:
7366:Phase transformation crystallography
6909:Different types of crystal structure
6288:McGraw Hill Encyclopaedia of Physics
6238:
6101:
5487:, random sampling, or metadynamics.
5352:
5296:
5208:
8073:
7873:Journal of Chemical Crystallography
7139:
6700:
6155:"4. Direct and reciprocal lattices"
5811:Some examples of crystal structures
5805:Some examples of crystal structures
5483:predicted using such approaches as
5405:
4856:, which move a point along a vector
2441:
2092:Overview of common lattice systems
511:
24:
6659:Proceedings of the Royal Society A
6614:Proceedings of the Royal Society A
6575:Proceedings of the Royal Society A
6106:(2nd ed.). Wiley. p. 1.
6030:Periodic table (crystal structure)
5054:
5051:
5048:
5045:
5042:
5039:
5036:
5033:
5021:
5018:
5015:
5012:
5009:
5006:
5003:
5000:
4984:
4981:
4978:
2471:
2078:
25:
8272:
6929:Crystal planes and Miller indices
6885:
6841:Lehrbuch der Anorganischen Chemie
5769:. All piezoelectric classes lack
224:
8217:
8207:
8197:
8187:
8166:
8155:
8150:
8140:
8130:
8120:
8110:
8025:
8014:
8013:
7620:
6890:
6452:Mechanical Behavior of Materials
5929:
5913:
5901:
5886:
5871:
5859:
5839:
5819:
5676:One good example of this is the
5356:
5300:
5212:
4884:
4696:{\displaystyle \mathbb {S} _{4}}
4644:{\displaystyle \mathbb {S} _{4}}
4531:{\displaystyle \mathbb {A} _{4}}
3600:{\displaystyle \mathbb {Z} _{3}}
3158:{\displaystyle \mathbb {Z} _{4}}
3106:{\displaystyle \mathbb {Z} _{4}}
2745:{\displaystyle \mathbb {Z} _{2}}
2688:{\displaystyle \mathbb {Z} _{2}}
2628:{\displaystyle \mathbb {Z} _{2}}
2571:{\displaystyle \mathbb {Z} _{1}}
2385:
2375:
2363:
2333:
2306:
2277:
2265:
2241:
2231:
2221:
2211:
2183:
2173:
2143:
354:
212:
200:
188:
6857:
6832:
6789:
6748:
6694:
6649:
6604:
6561:
6534:
6499:
6443:
6400:
6365:
6338:
6254:Introduction to Crystallography
6073:Hook, J.R.; Hall, H.E. (2010).
5797:behavior. This is analogous to
5574:
5348:
5261:(blue) interstitial sites in a
4839:
4775:
669:and hence are again simply the
7815:Bilbao Crystallographic Server
6206:
6173:
6147:
6095:
5736:crystal structure, similar to
5503:resonating valence bond theory
4891:Close-packing of equal spheres
1943:
1910:
1881:
1848:
1819:
1786:
1261:
1211:
1196:
1165:
1162:
1135:
1110:
1071:
691:) lattice planes is given by:
371:
348:
13:
1:
6934:Interactive 3D Crystal models
6292:(2nd ed.). McGraw-Hill.
6055:
5707:Another example is elemental
5292:
4951:
4922:hexagonal close packing (hcp)
504:than the other two axes. The
455:Dense crystallographic planes
8091:Crystallographic point group
6761:Journal of Materials Science
6011:– a crystal growth technique
6009:Laser-heated pedestal growth
5478:Crystal structure prediction
4782:crystallographic point group
2487:Overview of crystal systems
2466:crystallographic point group
2349:
2322:
2289:
2251:
2197:
2159:
2129:
126:
7:
7863:Crystal Growth & Design
7155:Timeline of crystallography
6868:(2nd ed.). CRC Press.
6104:Basic Solid State Chemistry
5944:
5494:, first set out in 1929 by
256:By definition, the syntax (
253:as directional parameters.
10:
8277:
7674:Nuclear magnetic resonance
6939:Specific Crystal 3D models
6284:Parker, C.B., ed. (1994).
5948:
5773:. Any material develops a
5688:. In the vast majority of
5475:
5409:
5387:
5281:
5243:
4955:
4888:
2475:
2063:
2060:Classification by symmetry
528:. For cubic crystals with
183:of the crystal structure.
130:
8177:
8099:
8081:
8009:
7929:
7901:
7878:Journal of Crystal Growth
7853:
7805:
7752:
7699:
7630:
7618:
7413:
7404:
7327:
7180:
7147:
7081:
7068:Characterization analysis
7060:
7010:
6987:
6782:10.1007/s10853-010-5113-0
6450:Courtney, Thomas (2000).
6190:10.1007/978-1-349-02595-4
6102:West, Anthony R. (1999).
5988:Crystallographic database
5782:. All polar crystals are
4942:cubic close packing (ccp)
3862:ditrigonal-scalenohedral
3552:
3484:ditetragonal-dipyramidal
3397:tetragonal-scalenohedral
3233:tetragonal-trapezohedral
2516:
2511:
2508:
2503:
2497:
2494:
2491:
2422:(often confused with the
2323:
2125:
2122:
2119:
2116:
117:electronic band structure
8148:trigonal & hexagonal
7744:Single particle analysis
7602:Hermann–Mauguin notation
7082:Material characteristics
6429:10.1103/PhysRevA.32.2530
6248:Donald E. Sands (1994).
6180:Edington, J. W. (1975).
6003:Hermann–Mauguin notation
5719:, α- and β-tin. The two
5659:crystallographic defects
5193:Triangular orthobicupola
4424:dihexagonal-dipyramidal
4178:hexagonal-trapezohedral
3117:tetragonal-disphenoidal
2398:The most symmetric, the
7868:Crystallography Reviews
7712:Isomorphous replacement
7506:Lomer–Cottrell junction
6979:Fundamental aspects of
6394:10.1103/PhysRev.188.870
6345:Kallay, Nikola (2000).
5485:evolutionary algorithms
5472:Prediction of structure
5284:Crystallographic defect
4342:ditrigonal-dipyramidal
3698:trigonal-trapezohedral
3315:ditetragonal-pyramidal
3169:tetragonal-dipyramidal
2424:trigonal crystal system
219:Face-centered cubic (F)
207:Body-centered cubic (I)
68:three-dimensional space
7381:Spinodal decomposition
6680:10.1098/rspa.1957.0073
6635:10.1098/rspa.1951.0172
6597:10.1098/rspa.1949.0032
6528:10.1103/PhysRev.54.899
5993:Fractional coordinates
5623:
5586:is one of the several
5278:Defects and impurities
5266:
5183:Hexagonal close-packed
5098:Atomic packing factor
5077:
4900:
4761:
4697:
4645:
4596:
4532:
4478:
4414:
4332:
4260:dihexagonal-pyramidal
4250:
4168:
4114:hexagonal-dipyramidal
4104:
4022:
3934:
3852:
3770:
3688:
3601:
3538:
3474:
3387:
3305:
3223:
3159:
3107:
3047:
2982:
2904:
2823:
2746:
2689:
2629:
2572:
2449:, also referred to as
2072:translational symmetry
2048:
1592:
1392:
1271:
1025:
889:
780:
617:
456:
382:
234:
156:fractional coordinates
39:
7921:Gregori Aminoff Prize
7717:Molecular replacement
6899:at Wikimedia Commons
6734:10.1139/cjp-2014-0518
6220:(1976). "Chapter 7".
6025:X-ray crystallography
5582:
5253:
5078:
4958:Atomic packing factor
4898:
4762:
4698:
4646:
4597:
4533:
4479:
4415:
4333:
4251:
4169:
4105:
4032:trigonal-dipyramidal
4023:
3935:
3853:
3780:ditrigonal-pyramidal
3771:
3689:
3602:
3539:
3475:
3388:
3306:
3224:
3160:
3108:
3062:tetragonal-pyramidal
3048:
2983:
2905:
2838:rhombic-disphenoidal
2824:
2747:
2690:
2630:
2573:
2049:
1593:
1393:
1272:
1026:
890:
781:
618:
526:Cartesian coordinates
454:
392:Planes and directions
383:
232:
34:Crystal structure of
33:
7227:Structure prediction
7094:Electronic structure
7011:Classes of materials
6568:Pauling, L. (1949).
6348:Interfacial Dynamics
5791:perovskite structure
5669:, solubilities, and
5442:thermal conductivity
5272:interstitial defects
4974:
4727:
4678:
4626:
4562:
4513:
4444:
4365:
4283:
4201:
4134:
4055:
3973:
3947:hexagonal-pyramidal
3885:
3803:
3721:
3639:
3582:
3504:
3425:
3338:
3256:
3189:
3140:
3088:
3020:
2940:
2862:
2781:
2727:
2670:
2610:
2553:
2112:14 Bravais lattices
1608:
1408:
1287:
1045:
905:
796:
701:
671:Cartesian directions
546:
327:
121:optical transparency
64:crystalline material
8241:Chemical properties
7491:Cottrell atmosphere
7471:Partial dislocation
7215:Restriction theorem
6810:2000HTemp..38..715M
6773:2011JMatS..46..855H
6726:2015CaJPh..93..974L
6672:1957RSPSA.240..145A
6627:1951RSPSA.208..431H
6588:1949RSPSA.196..343P
6555:10.1021/ja01195a024
6520:1938PhRv...54..899P
6493:10.1021/ja01379a006
6421:1985PhRvA..32.2530Z
6386:1969PhRv..188..870H
6222:Solid State Physics
6077:Solid State Physics
5998:Frank–Kasper phases
5977:Crystal engineering
5955:Ceramic engineering
5757:Physical properties
5263:face-centered cubic
5165:Face-centered cubic
5147:Body-centered cubic
5101:Coordination number
4962:Coordination number
4876:Atomic coordination
4665:non-centrosymmetric
4352:non-centrosymmetric
4042:non-centrosymmetric
3556:trigonal-pyramidal
3412:non-centrosymmetric
3127:non-centrosymmetric
2488:
2107:Schönflies notation
2093:
677:Interplanar spacing
663:body-centered cubic
659:face-centered cubic
636:⟨100⟩
502:rotational symmetry
500:) which has higher
482:Plastic deformation
7911:Carl Hermann Medal
7722:Molecular dynamics
7569:Defects in diamond
7564:Stone–Wales defect
7210:Reciprocal lattice
7172:Biocrystallography
6897:Crystal structures
6818:10.1007/BF02755923
6021:Patterson function
5771:inversion symmetry
5655:amorphous fraction
5645:, which refers to
5624:
5368:. You can help by
5312:. You can help by
5267:
5224:. You can help by
5205:Interstitial sites
5095:Crystal structure
5073:
4901:
4815:center of symmetry
4757:
4693:
4641:
4592:
4528:
4474:
4410:
4328:
4246:
4164:
4100:
4018:
3930:
3848:
3766:
3684:
3597:
3534:
3470:
3383:
3301:
3219:
3155:
3103:
3043:
2978:
2900:
2819:
2742:
2685:
2625:
2568:
2486:
2126:Face-centered (F)
2123:Body-centered (I)
2120:Base-centered (S)
2091:
2044:
1588:
1388:
1267:
1021:
885:
776:
687:between adjacent (
613:
457:
425:Optical properties
378:
318:reciprocal lattice
304:Considering only (
235:
94:lattice parameters
40:
8251:Materials science
8228:
8227:
8041:
8040:
8005:
8004:
7612:Thermal ellipsoid
7577:
7576:
7486:Frank–Read source
7446:
7445:
7312:Aperiodic crystal
7278:
7277:
7160:Crystallographers
7107:
7106:
7089:Crystal structure
6988:Materials science
6981:materials science
6895:Media related to
6875:978-1-56676-661-6
6850:978-3-11-007511-3
6582:(1046): 343–362.
6481:J. Am. Chem. Soc.
6461:978-1-57766-425-3
6409:Physical Review A
6199:978-0-333-18292-5
6113:978-0-471-98756-7
6048:Wigner–Seitz cell
5951:Materials science
5671:X-ray diffraction
5386:
5385:
5330:
5329:
5246:Interstitial site
5242:
5241:
5199:
5198:
5071:
5068:
4834:symmetry elements
4825:Improper rotation
4770:
4769:
2408:tetrahedral angle
2396:
2395:
2042:
1908:
1846:
1784:
1742:
1699:
1656:
1626:
1565:
1530:
1503:
1458:
1426:
1386:
1359:
1332:
1305:
1265:
1063:
1037:primitive setting
1019:
988:
936:
923:
883:
856:
814:
774:
719:
611:
610:
376:
90:lattice constants
48:crystal structure
16:(Redirected from
8268:
8221:
8211:
8201:
8191:
8170:
8159:
8154:
8144:
8134:
8124:
8114:
8100:Seven 3D systems
8068:
8061:
8054:
8045:
8044:
8029:
8028:
8017:
8016:
7960:
7959:
7883:Kristallografija
7737:Gerchberg–Saxton
7632:Characterisation
7624:
7607:Structure factor
7411:
7410:
7396:Ostwald ripening
7233:
7232:
7178:
7177:
7134:
7127:
7120:
7111:
7110:
7061:Analysis methods
6973:
6966:
6959:
6950:
6949:
6894:
6880:
6879:
6861:
6855:
6854:
6836:
6830:
6829:
6798:High Temperature
6793:
6787:
6786:
6784:
6752:
6746:
6745:
6719:
6717:cond-mat/0209372
6698:
6692:
6691:
6653:
6647:
6646:
6608:
6602:
6601:
6599:
6565:
6559:
6558:
6538:
6532:
6531:
6503:
6497:
6496:
6487:(4): 1010–1026.
6472:
6466:
6465:
6447:
6441:
6440:
6415:(4): 2530–2533.
6404:
6398:
6397:
6369:
6363:
6362:
6342:
6336:
6335:
6317:
6304:
6303:
6291:
6281:
6268:
6267:
6245:
6236:
6235:
6210:
6204:
6203:
6177:
6171:
6170:
6168:
6166:
6151:
6145:
6138:
6127:
6124:
6118:
6117:
6099:
6093:
6092:
6080:
6070:
5933:
5917:
5905:
5890:
5875:
5863:
5843:
5823:
5793:, which exhibit
5767:piezoelectricity
5647:elemental solids
5554:
5553:
5549:
5544:
5543:
5539:
5534:
5533:
5529:
5524:
5523:
5519:
5514:
5513:
5509:
5406:Grain boundaries
5381:
5378:
5360:
5353:
5325:
5322:
5304:
5297:
5237:
5234:
5216:
5209:
5092:
5091:
5082:
5080:
5079:
5074:
5072:
5070:
5069:
5066:
5060:
5059:
5058:
5057:
5026:
5025:
5024:
4992:
4987:
4797:reflection plane
4766:
4764:
4763:
4758:
4756:
4755:
4750:
4741:
4740:
4735:
4702:
4700:
4699:
4694:
4692:
4691:
4686:
4650:
4648:
4647:
4642:
4640:
4639:
4634:
4601:
4599:
4598:
4593:
4591:
4590:
4585:
4576:
4575:
4570:
4537:
4535:
4534:
4529:
4527:
4526:
4521:
4483:
4481:
4480:
4475:
4473:
4472:
4467:
4458:
4457:
4452:
4419:
4417:
4416:
4411:
4409:
4408:
4403:
4394:
4393:
4388:
4379:
4378:
4373:
4337:
4335:
4334:
4329:
4327:
4326:
4321:
4312:
4311:
4306:
4297:
4296:
4291:
4255:
4253:
4252:
4247:
4245:
4244:
4239:
4230:
4229:
4224:
4215:
4214:
4209:
4173:
4171:
4170:
4165:
4163:
4162:
4157:
4148:
4147:
4142:
4109:
4107:
4106:
4101:
4099:
4098:
4093:
4084:
4083:
4078:
4069:
4068:
4063:
4027:
4025:
4024:
4019:
4017:
4016:
4011:
4002:
4001:
3996:
3987:
3986:
3981:
3939:
3937:
3936:
3931:
3929:
3928:
3923:
3914:
3913:
3908:
3899:
3898:
3893:
3857:
3855:
3854:
3849:
3847:
3846:
3841:
3832:
3831:
3826:
3817:
3816:
3811:
3775:
3773:
3772:
3767:
3765:
3764:
3759:
3750:
3749:
3744:
3735:
3734:
3729:
3693:
3691:
3690:
3685:
3683:
3682:
3677:
3668:
3667:
3662:
3653:
3652:
3647:
3606:
3604:
3603:
3598:
3596:
3595:
3590:
3543:
3541:
3540:
3535:
3533:
3532:
3527:
3518:
3517:
3512:
3479:
3477:
3476:
3471:
3469:
3468:
3463:
3454:
3453:
3448:
3439:
3438:
3433:
3392:
3390:
3389:
3384:
3382:
3381:
3376:
3367:
3366:
3361:
3352:
3351:
3346:
3310:
3308:
3307:
3302:
3300:
3299:
3294:
3285:
3284:
3279:
3270:
3269:
3264:
3228:
3226:
3225:
3220:
3218:
3217:
3212:
3203:
3202:
3197:
3164:
3162:
3161:
3156:
3154:
3153:
3148:
3112:
3110:
3109:
3104:
3102:
3101:
3096:
3052:
3050:
3049:
3044:
3042:
3041:
3036:
3027:
2987:
2985:
2984:
2979:
2977:
2976:
2971:
2962:
2961:
2956:
2947:
2909:
2907:
2906:
2901:
2899:
2898:
2893:
2884:
2883:
2878:
2869:
2828:
2826:
2825:
2820:
2818:
2817:
2812:
2803:
2802:
2797:
2788:
2751:
2749:
2748:
2743:
2741:
2740:
2735:
2694:
2692:
2691:
2686:
2684:
2683:
2678:
2634:
2632:
2631:
2626:
2624:
2623:
2618:
2577:
2575:
2574:
2569:
2567:
2566:
2561:
2501:/ Crystal class
2489:
2485:
2447:Bravais lattices
2442:Bravais lattices
2389:
2379:
2367:
2337:
2310:
2281:
2269:
2245:
2235:
2225:
2215:
2187:
2177:
2147:
2094:
2090:
2053:
2051:
2050:
2045:
2043:
2041:
2001:
2000:
1982:
1981:
1963:
1962:
1946:
1909:
1907:
1899:
1888:
1847:
1845:
1837:
1826:
1785:
1783:
1775:
1764:
1753:
1752:
1743:
1741:
1740:
1731:
1730:
1721:
1710:
1709:
1700:
1698:
1697:
1688:
1687:
1678:
1667:
1666:
1657:
1655:
1654:
1645:
1644:
1635:
1632:
1627:
1625:
1624:
1612:
1597:
1595:
1594:
1589:
1581:
1580:
1571:
1567:
1566:
1564:
1556:
1536:
1531:
1529:
1528:
1519:
1518:
1509:
1504:
1502:
1501:
1492:
1485:
1484:
1475:
1474:
1464:
1459:
1457:
1456:
1447:
1446:
1437:
1427:
1425:
1424:
1412:
1397:
1395:
1394:
1389:
1387:
1385:
1384:
1375:
1374:
1365:
1360:
1358:
1357:
1348:
1347:
1338:
1333:
1331:
1330:
1321:
1320:
1311:
1306:
1304:
1303:
1291:
1276:
1274:
1273:
1268:
1266:
1264:
1254:
1253:
1232:
1231:
1210:
1209:
1199:
1177:
1176:
1122:
1121:
1109:
1108:
1096:
1095:
1083:
1082:
1069:
1064:
1062:
1061:
1049:
1030:
1028:
1027:
1022:
1020:
1018:
1017:
1008:
1007:
998:
993:
989:
987:
986:
977:
976:
975:
954:
953:
943:
937:
929:
924:
922:
921:
909:
894:
892:
891:
886:
884:
882:
881:
872:
871:
862:
857:
855:
854:
845:
844:
843:
831:
830:
820:
815:
813:
812:
800:
785:
783:
782:
777:
775:
773:
772:
763:
762:
761:
749:
748:
736:
735:
725:
720:
718:
717:
705:
637:
622:
620:
619:
614:
612:
609:
608:
596:
595:
583:
582:
573:
569:
564:
563:
530:lattice constant
512:Cubic structures
460:Microstructural
429:Refractive index
387:
385:
384:
379:
377:
375:
374:
369:
368:
357:
351:
345:
337:
312:), the distance
300:
216:
204:
195:Simple cubic (P)
192:
21:
8276:
8275:
8271:
8270:
8269:
8267:
8266:
8265:
8246:Crystallography
8231:
8230:
8229:
8224:
8178:Four 2D systems
8173:
8095:
8086:Bravais lattice
8077:
8075:Crystal systems
8072:
8042:
8037:
8001:
7958:
7925:
7897:
7849:
7801:
7772:CrystalExplorer
7748:
7732:Phase retrieval
7695:
7626:
7625:
7616:
7573:
7552:Schottky defect
7451:Perfect crystal
7442:
7438:Abnormal growth
7400:
7386:Supersaturation
7349:Miscibility gap
7330:
7323:
7274:
7231:
7195:Bravais lattice
7176:
7143:
7141:Crystallography
7138:
7108:
7103:
7077:
7056:
7006:
6983:
6977:
6888:
6883:
6876:
6862:
6858:
6851:
6837:
6833:
6794:
6790:
6753:
6749:
6699:
6695:
6654:
6650:
6609:
6605:
6566:
6562:
6539:
6535:
6514:(11): 899–904.
6508:Physical Review
6504:
6500:
6473:
6469:
6462:
6448:
6444:
6405:
6401:
6374:Physical Review
6370:
6366:
6359:
6343:
6339:
6332:
6318:
6307:
6300:
6282:
6271:
6264:
6246:
6239:
6232:
6211:
6207:
6200:
6178:
6174:
6164:
6162:
6153:
6152:
6148:
6139:
6130:
6125:
6121:
6114:
6100:
6096:
6089:
6071:
6062:
6058:
6053:
5966:
5947:
5942:
5941:
5940:
5937:
5934:
5925:
5923:
5918:
5909:
5906:
5897:
5896:
5891:
5882:
5881:
5876:
5867:
5864:
5855:
5853:
5849:
5844:
5835:
5833:
5829:
5824:
5813:
5812:
5807:
5759:
5703:
5699:
5695:
5687:
5682:silicon dioxide
5597:
5577:
5551:
5547:
5546:
5541:
5537:
5536:
5531:
5527:
5526:
5521:
5517:
5516:
5511:
5507:
5506:
5492:Pauling's rules
5480:
5474:
5466:amorphous solid
5414:
5408:
5392:
5382:
5376:
5373:
5366:needs expansion
5351:
5326:
5320:
5317:
5310:needs expansion
5295:
5286:
5280:
5248:
5238:
5232:
5229:
5222:needs expansion
5207:
5102:
5065:
5061:
5032:
5031:
5027:
4999:
4998:
4994:
4993:
4991:
4977:
4975:
4972:
4971:
4964:
4956:Main articles:
4954:
4893:
4887:
4878:
4842:
4819:inversion point
4778:
4751:
4746:
4745:
4736:
4731:
4730:
4728:
4725:
4724:
4717:centrosymmetric
4713:
4687:
4682:
4681:
4679:
4676:
4675:
4661:
4655:hextetrahedral
4635:
4630:
4629:
4627:
4624:
4623:
4586:
4581:
4580:
4571:
4566:
4565:
4563:
4560:
4559:
4552:centrosymmetric
4548:
4522:
4517:
4516:
4514:
4511:
4510:
4468:
4463:
4462:
4453:
4448:
4447:
4445:
4442:
4441:
4434:centrosymmetric
4430:
4404:
4399:
4398:
4389:
4384:
4383:
4374:
4369:
4368:
4366:
4363:
4362:
4348:
4322:
4317:
4316:
4307:
4302:
4301:
4292:
4287:
4286:
4284:
4281:
4280:
4266:
4240:
4235:
4234:
4225:
4220:
4219:
4210:
4205:
4204:
4202:
4199:
4198:
4184:
4158:
4153:
4152:
4143:
4138:
4137:
4135:
4132:
4131:
4124:centrosymmetric
4120:
4094:
4089:
4088:
4079:
4074:
4073:
4064:
4059:
4058:
4056:
4053:
4052:
4038:
4012:
4007:
4006:
3997:
3992:
3991:
3982:
3977:
3976:
3974:
3971:
3970:
3953:
3924:
3919:
3918:
3909:
3904:
3903:
3894:
3889:
3888:
3886:
3883:
3882:
3872:centrosymmetric
3868:
3842:
3837:
3836:
3827:
3822:
3821:
3812:
3807:
3806:
3804:
3801:
3800:
3786:
3760:
3755:
3754:
3745:
3740:
3739:
3730:
3725:
3724:
3722:
3719:
3718:
3704:
3678:
3673:
3672:
3663:
3658:
3657:
3648:
3643:
3642:
3640:
3637:
3636:
3626:centrosymmetric
3621:
3617:
3591:
3586:
3585:
3583:
3580:
3579:
3562:
3528:
3523:
3522:
3513:
3508:
3507:
3505:
3502:
3501:
3494:centrosymmetric
3490:
3464:
3459:
3458:
3449:
3444:
3443:
3434:
3429:
3428:
3426:
3423:
3422:
3407:
3403:
3377:
3372:
3371:
3362:
3357:
3356:
3347:
3342:
3341:
3339:
3336:
3335:
3321:
3295:
3290:
3289:
3280:
3275:
3274:
3265:
3260:
3259:
3257:
3254:
3253:
3239:
3213:
3208:
3207:
3198:
3193:
3192:
3190:
3187:
3186:
3179:centrosymmetric
3175:
3149:
3144:
3143:
3141:
3138:
3137:
3123:
3097:
3092:
3091:
3089:
3086:
3085:
3068:
3037:
3032:
3031:
3023:
3021:
3018:
3017:
3010:centrosymmetric
3005:
3001:
2972:
2967:
2966:
2957:
2952:
2951:
2943:
2941:
2938:
2937:
2923:
2894:
2889:
2888:
2879:
2874:
2873:
2865:
2863:
2860:
2859:
2844:
2813:
2808:
2807:
2798:
2793:
2792:
2784:
2782:
2779:
2778:
2768:centrosymmetric
2764:
2736:
2731:
2730:
2728:
2725:
2724:
2709:
2705:
2679:
2674:
2673:
2671:
2668:
2667:
2650:
2619:
2614:
2613:
2611:
2608:
2607:
2597:centrosymmetric
2592:
2588:
2562:
2557:
2556:
2554:
2551:
2550:
2535:
2509:Point symmetry
2495:Crystal system
2492:Crystal family
2480:
2474:
2472:Crystal systems
2444:
2360:
2330:
2303:
2262:
2208:
2170:
2140:
2104:
2100:Lattice system
2097:Crystal family
2086:crystal systems
2081:
2079:Lattice systems
2068:
2062:
1996:
1992:
1977:
1973:
1958:
1954:
1947:
1900:
1889:
1887:
1838:
1827:
1825:
1776:
1765:
1763:
1748:
1744:
1736:
1732:
1726:
1722:
1720:
1705:
1701:
1693:
1689:
1683:
1679:
1677:
1662:
1658:
1650:
1646:
1640:
1636:
1634:
1633:
1631:
1620:
1616:
1611:
1609:
1606:
1605:
1576:
1572:
1557:
1537:
1535:
1524:
1520:
1514:
1510:
1508:
1497:
1493:
1480:
1476:
1470:
1466:
1465:
1463:
1452:
1448:
1442:
1438:
1436:
1435:
1431:
1420:
1416:
1411:
1409:
1406:
1405:
1380:
1376:
1370:
1366:
1364:
1353:
1349:
1343:
1339:
1337:
1326:
1322:
1316:
1312:
1310:
1299:
1295:
1290:
1288:
1285:
1284:
1249:
1245:
1227:
1223:
1205:
1201:
1200:
1172:
1168:
1117:
1113:
1104:
1100:
1091:
1087:
1078:
1074:
1070:
1068:
1057:
1053:
1048:
1046:
1043:
1042:
1013:
1009:
1003:
999:
997:
982:
978:
971:
967:
949:
945:
944:
942:
938:
928:
917:
913:
908:
906:
903:
902:
877:
873:
867:
863:
861:
850:
846:
839:
835:
826:
822:
821:
819:
808:
804:
799:
797:
794:
793:
768:
764:
757:
753:
744:
740:
731:
727:
726:
724:
713:
709:
704:
702:
699:
698:
679:
645:Coordinates in
635:
630:Coordinates in
604:
600:
591:
587:
578:
574:
568:
553:
549:
547:
544:
543:
514:
445:Surface tension
400:linking nodes (
394:
370:
358:
353:
352:
347:
346:
338:
336:
328:
325:
324:
298:
288:
277:
266:
227:
220:
217:
208:
205:
196:
193:
177:
170:
163:
135:
129:
98:cell parameters
83:Bravais lattice
44:crystallography
28:
23:
22:
18:Crystal lattice
15:
12:
11:
5:
8274:
8264:
8263:
8258:
8253:
8248:
8243:
8226:
8225:
8223:
8222:
8212:
8202:
8192:
8181:
8179:
8175:
8174:
8172:
8171:
8160:
8145:
8135:
8125:
8115:
8103:
8101:
8097:
8096:
8094:
8093:
8088:
8082:
8079:
8078:
8071:
8070:
8063:
8056:
8048:
8039:
8038:
8036:
8035:
8023:
8010:
8007:
8006:
8003:
8002:
8000:
7999:
7994:
7989:
7988:
7987:
7982:
7977:
7966:
7964:
7957:
7956:
7951:
7946:
7941:
7935:
7933:
7927:
7926:
7924:
7923:
7918:
7913:
7907:
7905:
7899:
7898:
7896:
7895:
7890:
7885:
7880:
7875:
7870:
7865:
7859:
7857:
7851:
7850:
7848:
7847:
7842:
7837:
7832:
7827:
7822:
7817:
7811:
7809:
7803:
7802:
7800:
7799:
7794:
7789:
7784:
7779:
7774:
7769:
7764:
7758:
7756:
7750:
7749:
7747:
7746:
7741:
7740:
7739:
7729:
7724:
7719:
7714:
7709:
7707:Direct methods
7703:
7701:
7697:
7696:
7694:
7693:
7692:
7691:
7686:
7676:
7671:
7670:
7669:
7664:
7654:
7653:
7652:
7647:
7636:
7634:
7628:
7627:
7619:
7617:
7615:
7614:
7609:
7604:
7599:
7594:
7592:Ewald's sphere
7589:
7584:
7578:
7575:
7574:
7572:
7571:
7566:
7561:
7560:
7559:
7554:
7544:
7543:
7542:
7537:
7535:Frenkel defect
7532:
7530:Bjerrum defect
7522:
7521:
7520:
7510:
7509:
7508:
7503:
7498:
7496:Peierls stress
7493:
7488:
7483:
7478:
7473:
7468:
7466:Burgers vector
7458:
7456:Stacking fault
7453:
7447:
7444:
7443:
7441:
7440:
7435:
7430:
7425:
7419:
7417:
7415:Grain boundary
7408:
7402:
7401:
7399:
7398:
7393:
7388:
7383:
7378:
7373:
7368:
7363:
7362:
7361:
7359:Liquid crystal
7356:
7351:
7346:
7335:
7333:
7325:
7324:
7322:
7321:
7320:
7319:
7309:
7308:
7307:
7297:
7296:
7295:
7290:
7279:
7276:
7275:
7273:
7272:
7267:
7262:
7257:
7252:
7247:
7241:
7239:
7230:
7229:
7224:
7222:Periodic table
7219:
7218:
7217:
7212:
7207:
7202:
7197:
7186:
7184:
7175:
7174:
7169:
7164:
7163:
7162:
7151:
7149:
7145:
7144:
7137:
7136:
7129:
7122:
7114:
7105:
7104:
7102:
7101:
7099:Microstructure
7096:
7091:
7085:
7083:
7079:
7078:
7076:
7075:
7070:
7064:
7062:
7058:
7057:
7055:
7054:
7049:
7048:
7047:
7037:
7032:
7031:
7030:
7028:Semiconductors
7025:
7014:
7012:
7008:
7007:
7005:
7004:
7001:
6998:
6995:
6991:
6989:
6985:
6984:
6976:
6975:
6968:
6961:
6953:
6947:
6946:
6941:
6936:
6931:
6926:
6921:
6916:
6911:
6906:
6887:
6886:External links
6884:
6882:
6881:
6874:
6856:
6849:
6831:
6804:(5): 715–721.
6788:
6767:(4): 855–874.
6747:
6710:(9): 974–978.
6693:
6648:
6603:
6560:
6549:(3): 542–553.
6533:
6498:
6467:
6460:
6442:
6399:
6380:(2): 870–874.
6364:
6358:978-0824700065
6357:
6337:
6330:
6305:
6299:978-0070514003
6298:
6269:
6263:978-0486678399
6262:
6237:
6231:978-0030493461
6230:
6205:
6198:
6172:
6146:
6128:
6119:
6112:
6094:
6087:
6059:
6057:
6054:
6052:
6051:
6045:
6039:
6036:Primitive cell
6033:
6027:
6018:
6015:Liquid crystal
6012:
6006:
6000:
5995:
5990:
5985:
5982:Crystal growth
5979:
5974:
5971:Brillouin zone
5967:
5946:
5943:
5939:
5938:
5935:
5928:
5926:
5921:
5919:
5912:
5910:
5907:
5900:
5898:
5894:
5892:
5885:
5883:
5879:
5877:
5870:
5868:
5865:
5858:
5856:
5851:
5847:
5845:
5838:
5836:
5831:
5827:
5825:
5818:
5815:
5814:
5810:
5809:
5808:
5806:
5803:
5799:ferromagnetism
5758:
5755:
5701:
5697:
5693:
5685:
5667:melting points
5595:
5576:
5573:
5476:Main article:
5473:
5470:
5426:single crystal
5412:Grain boundary
5410:Main article:
5407:
5404:
5388:Main article:
5384:
5383:
5363:
5361:
5350:
5347:
5328:
5327:
5307:
5305:
5294:
5291:
5282:Main article:
5279:
5276:
5244:Main article:
5240:
5239:
5219:
5217:
5206:
5203:
5197:
5196:
5189:
5186:
5179:
5178:
5171:
5168:
5161:
5160:
5153:
5150:
5143:
5142:
5135:
5132:
5126:
5125:
5118:
5115:
5109:
5108:
5099:
5096:
5084:
5083:
5064:
5056:
5053:
5050:
5047:
5044:
5041:
5038:
5035:
5030:
5023:
5020:
5017:
5014:
5011:
5008:
5005:
5002:
4997:
4990:
4986:
4983:
4980:
4953:
4950:
4938:
4937:
4918:
4917:
4889:Main article:
4886:
4883:
4877:
4874:
4870:
4869:
4863:
4857:
4841:
4838:
4830:
4829:
4821:
4808:
4799:
4777:
4774:
4768:
4767:
4754:
4749:
4744:
4739:
4734:
4722:
4719:
4714:
4711:
4708:
4707:hexoctahedral
4704:
4703:
4690:
4685:
4670:
4667:
4662:
4659:
4656:
4652:
4651:
4638:
4633:
4618:
4615:
4613:enantiomorphic
4610:
4607:
4603:
4602:
4589:
4584:
4579:
4574:
4569:
4557:
4554:
4549:
4546:
4543:
4539:
4538:
4525:
4520:
4505:
4502:
4500:enantiomorphic
4497:
4494:
4491:
4485:
4484:
4471:
4466:
4461:
4456:
4451:
4439:
4436:
4431:
4428:
4425:
4421:
4420:
4407:
4402:
4397:
4392:
4387:
4382:
4377:
4372:
4357:
4354:
4349:
4346:
4343:
4339:
4338:
4325:
4320:
4315:
4310:
4305:
4300:
4295:
4290:
4275:
4272:
4267:
4264:
4261:
4257:
4256:
4243:
4238:
4233:
4228:
4223:
4218:
4213:
4208:
4193:
4190:
4188:enantiomorphic
4185:
4182:
4179:
4175:
4174:
4161:
4156:
4151:
4146:
4141:
4129:
4126:
4121:
4118:
4115:
4111:
4110:
4097:
4092:
4087:
4082:
4077:
4072:
4067:
4062:
4047:
4044:
4039:
4036:
4033:
4029:
4028:
4015:
4010:
4005:
4000:
3995:
3990:
3985:
3980:
3965:
3962:
3957:enantiomorphic
3954:
3951:
3948:
3945:
3941:
3940:
3927:
3922:
3917:
3912:
3907:
3902:
3897:
3892:
3877:
3874:
3869:
3866:
3863:
3859:
3858:
3845:
3840:
3835:
3830:
3825:
3820:
3815:
3810:
3795:
3792:
3787:
3784:
3781:
3777:
3776:
3763:
3758:
3753:
3748:
3743:
3738:
3733:
3728:
3713:
3710:
3708:enantiomorphic
3705:
3702:
3699:
3695:
3694:
3681:
3676:
3671:
3666:
3661:
3656:
3651:
3646:
3631:
3628:
3623:
3619:
3615:
3612:
3608:
3607:
3594:
3589:
3574:
3571:
3566:enantiomorphic
3563:
3560:
3557:
3554:
3551:
3545:
3544:
3531:
3526:
3521:
3516:
3511:
3499:
3496:
3491:
3488:
3485:
3481:
3480:
3467:
3462:
3457:
3452:
3447:
3442:
3437:
3432:
3417:
3414:
3409:
3405:
3401:
3398:
3394:
3393:
3380:
3375:
3370:
3365:
3360:
3355:
3350:
3345:
3330:
3327:
3322:
3319:
3316:
3312:
3311:
3298:
3293:
3288:
3283:
3278:
3273:
3268:
3263:
3248:
3245:
3243:enantiomorphic
3240:
3237:
3234:
3230:
3229:
3216:
3211:
3206:
3201:
3196:
3184:
3181:
3176:
3173:
3170:
3166:
3165:
3152:
3147:
3132:
3129:
3124:
3121:
3118:
3114:
3113:
3100:
3095:
3080:
3077:
3072:enantiomorphic
3069:
3066:
3063:
3060:
3054:
3053:
3040:
3035:
3030:
3026:
3015:
3012:
3007:
3003:
2999:
2996:
2989:
2988:
2975:
2970:
2965:
2960:
2955:
2950:
2946:
2932:
2929:
2924:
2921:
2918:
2911:
2910:
2897:
2892:
2887:
2882:
2877:
2872:
2868:
2854:
2851:
2849:enantiomorphic
2846:
2842:
2839:
2836:
2830:
2829:
2816:
2811:
2806:
2801:
2796:
2791:
2787:
2773:
2770:
2765:
2762:
2759:
2753:
2752:
2739:
2734:
2719:
2716:
2711:
2707:
2703:
2700:
2696:
2695:
2682:
2677:
2662:
2659:
2654:enantiomorphic
2651:
2648:
2645:
2642:
2636:
2635:
2622:
2617:
2602:
2599:
2594:
2590:
2586:
2583:
2579:
2578:
2565:
2560:
2547:
2544:
2539:enantiomorphic
2536:
2533:
2530:
2527:
2521:
2520:
2518:Abstract group
2515:
2510:
2507:
2502:
2496:
2493:
2473:
2470:
2451:space lattices
2443:
2440:
2394:
2393:
2383:
2373:
2371:
2361:
2358:
2355:
2348:
2347:
2345:
2343:
2341:
2331:
2328:
2325:
2321:
2320:
2318:
2316:
2314:
2304:
2301:
2298:
2295:
2288:
2287:
2285:
2275:
2273:
2263:
2260:
2257:
2250:
2249:
2239:
2229:
2219:
2209:
2206:
2203:
2196:
2195:
2193:
2191:
2181:
2171:
2168:
2165:
2158:
2157:
2155:
2153:
2151:
2141:
2138:
2135:
2128:
2127:
2124:
2121:
2118:
2117:Primitive (P)
2114:
2113:
2110:
2101:
2098:
2080:
2077:
2066:Crystal system
2064:Main article:
2061:
2058:
2057:
2056:
2055:
2054:
2040:
2037:
2034:
2031:
2028:
2025:
2022:
2019:
2016:
2013:
2010:
2007:
2004:
1999:
1995:
1991:
1988:
1985:
1980:
1976:
1972:
1969:
1966:
1961:
1957:
1953:
1950:
1945:
1942:
1939:
1936:
1933:
1930:
1927:
1924:
1921:
1918:
1915:
1912:
1906:
1903:
1898:
1895:
1892:
1886:
1883:
1880:
1877:
1874:
1871:
1868:
1865:
1862:
1859:
1856:
1853:
1850:
1844:
1841:
1836:
1833:
1830:
1824:
1821:
1818:
1815:
1812:
1809:
1806:
1803:
1800:
1797:
1794:
1791:
1788:
1782:
1779:
1774:
1771:
1768:
1762:
1759:
1756:
1751:
1747:
1739:
1735:
1729:
1725:
1719:
1716:
1713:
1708:
1704:
1696:
1692:
1686:
1682:
1676:
1673:
1670:
1665:
1661:
1653:
1649:
1643:
1639:
1630:
1623:
1619:
1615:
1600:
1599:
1598:
1587:
1584:
1579:
1575:
1570:
1563:
1560:
1555:
1552:
1549:
1546:
1543:
1540:
1534:
1527:
1523:
1517:
1513:
1507:
1500:
1496:
1491:
1488:
1483:
1479:
1473:
1469:
1462:
1455:
1451:
1445:
1441:
1434:
1430:
1423:
1419:
1415:
1400:
1399:
1398:
1383:
1379:
1373:
1369:
1363:
1356:
1352:
1346:
1342:
1336:
1329:
1325:
1319:
1315:
1309:
1302:
1298:
1294:
1281:Orthorhombic:
1279:
1278:
1277:
1263:
1260:
1257:
1252:
1248:
1244:
1241:
1238:
1235:
1230:
1226:
1222:
1219:
1216:
1213:
1208:
1204:
1198:
1195:
1192:
1189:
1186:
1183:
1180:
1175:
1171:
1167:
1164:
1161:
1158:
1155:
1152:
1149:
1146:
1143:
1140:
1137:
1134:
1131:
1128:
1125:
1120:
1116:
1112:
1107:
1103:
1099:
1094:
1090:
1086:
1081:
1077:
1073:
1067:
1060:
1056:
1052:
1035:Rhombohedral (
1033:
1032:
1031:
1016:
1012:
1006:
1002:
996:
992:
985:
981:
974:
970:
966:
963:
960:
957:
952:
948:
941:
935:
932:
927:
920:
916:
912:
897:
896:
895:
880:
876:
870:
866:
860:
853:
849:
842:
838:
834:
829:
825:
818:
811:
807:
803:
788:
787:
786:
771:
767:
760:
756:
752:
747:
743:
739:
734:
730:
723:
716:
712:
708:
678:
675:
655:
654:
647:curly brackets
643:
632:angle brackets
624:
623:
607:
603:
599:
594:
590:
586:
581:
577:
572:
567:
562:
559:
556:
552:
535:, the spacing
513:
510:
498:principal axis
494:
493:
490:Burgers vector
479:
473:
449:
448:
442:
432:
416:are geometric
393:
390:
389:
388:
373:
367:
364:
361:
356:
350:
344:
341:
335:
332:
310:lattice planes
286:
275:
264:
226:
225:Miller indices
223:
222:
221:
218:
211:
209:
206:
199:
197:
194:
187:
175:
168:
161:
140:parallelepiped
131:Main article:
128:
125:
92:, also called
26:
9:
6:
4:
3:
2:
8273:
8262:
8259:
8257:
8254:
8252:
8249:
8247:
8244:
8242:
8239:
8238:
8236:
8220:
8216:
8213:
8210:
8206:
8203:
8200:
8196:
8193:
8190:
8186:
8183:
8182:
8180:
8176:
8169:
8164:
8161:
8158:
8153:
8149:
8146:
8143:
8139:
8136:
8133:
8129:
8126:
8123:
8119:
8116:
8113:
8108:
8105:
8104:
8102:
8098:
8092:
8089:
8087:
8084:
8083:
8080:
8076:
8069:
8064:
8062:
8057:
8055:
8050:
8049:
8046:
8034:
8033:
8024:
8022:
8021:
8012:
8011:
8008:
7998:
7995:
7993:
7990:
7986:
7983:
7981:
7978:
7976:
7973:
7972:
7971:
7968:
7967:
7965:
7961:
7955:
7952:
7950:
7947:
7945:
7942:
7940:
7937:
7936:
7934:
7932:
7928:
7922:
7919:
7917:
7914:
7912:
7909:
7908:
7906:
7904:
7900:
7894:
7891:
7889:
7886:
7884:
7881:
7879:
7876:
7874:
7871:
7869:
7866:
7864:
7861:
7860:
7858:
7856:
7852:
7846:
7843:
7841:
7838:
7836:
7833:
7831:
7828:
7826:
7823:
7821:
7818:
7816:
7813:
7812:
7810:
7808:
7804:
7798:
7795:
7793:
7790:
7788:
7785:
7783:
7780:
7778:
7775:
7773:
7770:
7768:
7765:
7763:
7760:
7759:
7757:
7755:
7751:
7745:
7742:
7738:
7735:
7734:
7733:
7730:
7728:
7727:Patterson map
7725:
7723:
7720:
7718:
7715:
7713:
7710:
7708:
7705:
7704:
7702:
7698:
7690:
7687:
7685:
7682:
7681:
7680:
7677:
7675:
7672:
7668:
7665:
7663:
7660:
7659:
7658:
7655:
7651:
7648:
7646:
7643:
7642:
7641:
7638:
7637:
7635:
7633:
7629:
7623:
7613:
7610:
7608:
7605:
7603:
7600:
7598:
7597:Friedel's law
7595:
7593:
7590:
7588:
7585:
7583:
7580:
7579:
7570:
7567:
7565:
7562:
7558:
7555:
7553:
7550:
7549:
7548:
7545:
7541:
7540:Wigner effect
7538:
7536:
7533:
7531:
7528:
7527:
7526:
7525:Interstitials
7523:
7519:
7516:
7515:
7514:
7511:
7507:
7504:
7502:
7499:
7497:
7494:
7492:
7489:
7487:
7484:
7482:
7479:
7477:
7474:
7472:
7469:
7467:
7464:
7463:
7462:
7459:
7457:
7454:
7452:
7449:
7448:
7439:
7436:
7434:
7431:
7429:
7426:
7424:
7421:
7420:
7418:
7416:
7412:
7409:
7407:
7403:
7397:
7394:
7392:
7389:
7387:
7384:
7382:
7379:
7377:
7374:
7372:
7371:Precipitation
7369:
7367:
7364:
7360:
7357:
7355:
7352:
7350:
7347:
7345:
7342:
7341:
7340:
7339:Phase diagram
7337:
7336:
7334:
7332:
7326:
7318:
7315:
7314:
7313:
7310:
7306:
7303:
7302:
7301:
7298:
7294:
7291:
7289:
7286:
7285:
7284:
7281:
7280:
7271:
7268:
7266:
7263:
7261:
7258:
7256:
7253:
7251:
7248:
7246:
7243:
7242:
7240:
7238:
7234:
7228:
7225:
7223:
7220:
7216:
7213:
7211:
7208:
7206:
7203:
7201:
7198:
7196:
7193:
7192:
7191:
7188:
7187:
7185:
7183:
7179:
7173:
7170:
7168:
7165:
7161:
7158:
7157:
7156:
7153:
7152:
7150:
7146:
7142:
7135:
7130:
7128:
7123:
7121:
7116:
7115:
7112:
7100:
7097:
7095:
7092:
7090:
7087:
7086:
7084:
7080:
7074:
7073:Phase diagram
7071:
7069:
7066:
7065:
7063:
7059:
7053:
7050:
7046:
7043:
7042:
7041:
7038:
7036:
7033:
7029:
7026:
7024:
7021:
7020:
7019:
7016:
7015:
7013:
7009:
7002:
6999:
6996:
6993:
6992:
6990:
6986:
6982:
6974:
6969:
6967:
6962:
6960:
6955:
6954:
6951:
6945:
6942:
6940:
6937:
6935:
6932:
6930:
6927:
6925:
6922:
6920:
6917:
6915:
6912:
6910:
6907:
6905:
6902:
6901:
6900:
6898:
6893:
6877:
6871:
6867:
6860:
6852:
6846:
6842:
6835:
6827:
6823:
6819:
6815:
6811:
6807:
6803:
6799:
6792:
6783:
6778:
6774:
6770:
6766:
6762:
6758:
6751:
6743:
6739:
6735:
6731:
6727:
6723:
6718:
6713:
6709:
6706:
6705:
6704:Can. J. Phys.
6697:
6689:
6685:
6681:
6677:
6673:
6669:
6666:(1221): 145.
6665:
6661:
6660:
6652:
6644:
6640:
6636:
6632:
6628:
6624:
6621:(1095): 431.
6620:
6616:
6615:
6607:
6598:
6593:
6589:
6585:
6581:
6577:
6576:
6571:
6564:
6556:
6552:
6548:
6544:
6537:
6529:
6525:
6521:
6517:
6513:
6509:
6502:
6494:
6490:
6486:
6483:
6482:
6477:
6471:
6463:
6457:
6453:
6446:
6438:
6434:
6430:
6426:
6422:
6418:
6414:
6410:
6403:
6395:
6391:
6387:
6383:
6379:
6375:
6368:
6360:
6354:
6351:. CRC Press.
6350:
6349:
6341:
6333:
6327:
6323:
6316:
6314:
6312:
6310:
6301:
6295:
6290:
6289:
6280:
6278:
6276:
6274:
6265:
6259:
6255:
6251:
6244:
6242:
6233:
6227:
6223:
6219:
6215:
6209:
6201:
6195:
6191:
6187:
6183:
6176:
6160:
6156:
6150:
6143:
6137:
6135:
6133:
6123:
6115:
6109:
6105:
6098:
6090:
6088:9780471928041
6084:
6079:
6078:
6069:
6067:
6065:
6060:
6049:
6046:
6043:
6040:
6037:
6034:
6031:
6028:
6026:
6022:
6019:
6016:
6013:
6010:
6007:
6004:
6001:
5999:
5996:
5994:
5991:
5989:
5986:
5983:
5980:
5978:
5975:
5972:
5969:
5968:
5964:
5960:
5956:
5952:
5932:
5927:
5920:β-quartz (SiO
5916:
5911:
5904:
5899:
5889:
5884:
5874:
5869:
5862:
5857:
5842:
5837:
5822:
5817:
5816:
5802:
5800:
5796:
5795:ferroelectric
5792:
5787:
5785:
5781:
5776:
5772:
5768:
5764:
5763:piezoelectric
5754:
5751:
5750:semiconductor
5747:
5743:
5739:
5735:
5734:diamond cubic
5730:
5726:
5722:
5718:
5714:
5710:
5705:
5691:
5683:
5679:
5674:
5672:
5668:
5664:
5660:
5656:
5652:
5651:crystal habit
5648:
5644:
5640:
5636:
5632:
5628:
5621:
5617:
5613:
5609:
5605:
5601:
5593:
5589:
5585:
5581:
5572:
5568:
5565:
5560:
5558:
5504:
5499:
5497:
5496:Linus Pauling
5493:
5488:
5486:
5479:
5469:
5467:
5463:
5459:
5453:
5451:
5447:
5446:precipitation
5443:
5439:
5435:
5430:
5427:
5422:
5420:
5413:
5403:
5401:
5397:
5391:
5380:
5371:
5367:
5364:This section
5362:
5359:
5355:
5354:
5346:
5344:
5341:
5340:ferromagnetic
5336:
5335:electron spin
5324:
5315:
5311:
5308:This section
5306:
5303:
5299:
5298:
5290:
5285:
5275:
5273:
5264:
5260:
5256:
5252:
5247:
5236:
5227:
5223:
5220:This section
5218:
5215:
5211:
5210:
5202:
5194:
5190:
5187:
5184:
5181:
5180:
5176:
5175:Cuboctahedron
5172:
5169:
5166:
5163:
5162:
5158:
5154:
5151:
5148:
5145:
5144:
5140:
5136:
5133:
5131:
5128:
5127:
5123:
5119:
5116:
5114:
5113:Diamond cubic
5111:
5110:
5106:
5100:
5097:
5094:
5093:
5090:
5087:
5062:
5028:
4995:
4988:
4970:
4969:
4968:
4963:
4959:
4949:
4945:
4943:
4935:
4931:
4930:
4929:
4925:
4923:
4915:
4911:
4910:
4909:
4906:
4897:
4892:
4885:Close packing
4882:
4873:
4867:
4864:
4861:
4858:
4855:
4851:
4850:
4849:
4847:
4837:
4835:
4827:
4826:
4822:
4820:
4816:
4812:
4809:
4807:
4806:rotation axis
4803:
4800:
4798:
4794:
4791:
4790:
4789:
4787:
4786:crystal class
4783:
4773:
4752:
4742:
4737:
4723:
4720:
4718:
4715:
4709:
4706:
4705:
4688:
4674:
4671:
4668:
4666:
4663:
4657:
4654:
4653:
4636:
4622:
4619:
4616:
4614:
4611:
4608:
4605:
4604:
4587:
4577:
4572:
4558:
4555:
4553:
4550:
4544:
4541:
4540:
4523:
4509:
4506:
4503:
4501:
4498:
4495:
4492:
4490:
4486:
4469:
4459:
4454:
4440:
4437:
4435:
4432:
4426:
4423:
4422:
4405:
4395:
4390:
4380:
4375:
4361:
4358:
4355:
4353:
4350:
4344:
4341:
4340:
4323:
4313:
4308:
4298:
4293:
4279:
4276:
4273:
4271:
4268:
4262:
4259:
4258:
4241:
4231:
4226:
4216:
4211:
4197:
4194:
4191:
4189:
4186:
4180:
4177:
4176:
4159:
4149:
4144:
4130:
4127:
4125:
4122:
4116:
4113:
4112:
4095:
4085:
4080:
4070:
4065:
4051:
4048:
4045:
4043:
4040:
4034:
4031:
4030:
4013:
4003:
3998:
3988:
3983:
3969:
3966:
3963:
3961:
3958:
3955:
3949:
3946:
3942:
3925:
3915:
3910:
3900:
3895:
3881:
3878:
3875:
3873:
3870:
3864:
3861:
3860:
3843:
3833:
3828:
3818:
3813:
3799:
3796:
3793:
3791:
3788:
3782:
3779:
3778:
3761:
3751:
3746:
3736:
3731:
3717:
3714:
3711:
3709:
3706:
3700:
3697:
3696:
3679:
3669:
3664:
3654:
3649:
3635:
3632:
3629:
3627:
3624:
3613:
3611:rhombohedral
3610:
3609:
3592:
3578:
3575:
3572:
3570:
3567:
3564:
3558:
3555:
3550:
3546:
3529:
3519:
3514:
3500:
3497:
3495:
3492:
3486:
3483:
3482:
3465:
3455:
3450:
3440:
3435:
3421:
3418:
3415:
3413:
3410:
3399:
3396:
3395:
3378:
3368:
3363:
3353:
3348:
3334:
3331:
3328:
3326:
3323:
3317:
3314:
3313:
3296:
3286:
3281:
3271:
3266:
3252:
3249:
3246:
3244:
3241:
3235:
3232:
3231:
3214:
3204:
3199:
3185:
3182:
3180:
3177:
3171:
3168:
3167:
3150:
3136:
3133:
3130:
3128:
3125:
3119:
3116:
3115:
3098:
3084:
3081:
3078:
3076:
3073:
3070:
3064:
3061:
3059:
3055:
3038:
3028:
3016:
3013:
3011:
3008:
2997:
2995:
2991:
2990:
2973:
2963:
2958:
2948:
2936:
2933:
2930:
2928:
2925:
2919:
2917:
2913:
2912:
2895:
2885:
2880:
2870:
2858:
2855:
2852:
2850:
2847:
2840:
2837:
2835:
2831:
2814:
2804:
2799:
2789:
2777:
2774:
2771:
2769:
2766:
2760:
2758:
2755:
2754:
2737:
2723:
2720:
2717:
2715:
2712:
2701:
2698:
2697:
2680:
2666:
2663:
2660:
2658:
2655:
2652:
2646:
2643:
2641:
2637:
2620:
2606:
2603:
2600:
2598:
2595:
2584:
2581:
2580:
2563:
2548:
2545:
2543:
2540:
2537:
2531:
2528:
2526:
2522:
2519:
2514:
2506:
2500:
2490:
2484:
2479:
2469:
2467:
2463:
2458:
2456:
2455:quasicrystals
2452:
2448:
2439:
2437:
2433:
2429:
2425:
2421:
2417:
2413:
2409:
2405:
2401:
2392:
2388:
2384:
2382:
2378:
2374:
2372:
2370:
2366:
2362:
2356:
2353:
2346:
2344:
2342:
2340:
2336:
2332:
2326:
2319:
2317:
2315:
2313:
2309:
2305:
2299:
2297:Rhombohedral
2296:
2293:
2286:
2284:
2280:
2276:
2274:
2272:
2268:
2264:
2258:
2255:
2248:
2244:
2240:
2238:
2234:
2230:
2228:
2224:
2220:
2218:
2214:
2210:
2204:
2201:
2194:
2192:
2190:
2186:
2182:
2180:
2176:
2172:
2166:
2163:
2156:
2154:
2152:
2150:
2146:
2142:
2136:
2133:
2115:
2108:
2095:
2089:
2087:
2076:
2073:
2067:
2038:
2035:
2032:
2029:
2026:
2023:
2020:
2017:
2014:
2011:
2008:
2005:
2002:
1997:
1993:
1989:
1986:
1983:
1978:
1974:
1970:
1967:
1964:
1959:
1955:
1951:
1948:
1940:
1937:
1934:
1931:
1928:
1925:
1922:
1919:
1916:
1913:
1904:
1901:
1896:
1893:
1890:
1884:
1878:
1875:
1872:
1869:
1866:
1863:
1860:
1857:
1854:
1851:
1842:
1839:
1834:
1831:
1828:
1822:
1816:
1813:
1810:
1807:
1804:
1801:
1798:
1795:
1792:
1789:
1780:
1777:
1772:
1769:
1766:
1760:
1757:
1754:
1749:
1745:
1737:
1733:
1727:
1723:
1717:
1714:
1711:
1706:
1702:
1694:
1690:
1684:
1680:
1674:
1671:
1668:
1663:
1659:
1651:
1647:
1641:
1637:
1628:
1621:
1617:
1613:
1604:
1603:
1601:
1585:
1582:
1577:
1573:
1568:
1561:
1558:
1553:
1550:
1547:
1544:
1541:
1538:
1532:
1525:
1521:
1515:
1511:
1505:
1498:
1494:
1489:
1486:
1481:
1477:
1471:
1467:
1460:
1453:
1449:
1443:
1439:
1432:
1428:
1421:
1417:
1413:
1404:
1403:
1401:
1381:
1377:
1371:
1367:
1361:
1354:
1350:
1344:
1340:
1334:
1327:
1323:
1317:
1313:
1307:
1300:
1296:
1292:
1283:
1282:
1280:
1258:
1255:
1250:
1246:
1242:
1239:
1236:
1233:
1228:
1224:
1220:
1217:
1214:
1206:
1202:
1193:
1190:
1187:
1184:
1181:
1178:
1173:
1169:
1159:
1156:
1153:
1150:
1147:
1144:
1141:
1138:
1132:
1129:
1126:
1123:
1118:
1114:
1105:
1101:
1097:
1092:
1088:
1084:
1079:
1075:
1065:
1058:
1054:
1050:
1041:
1040:
1038:
1034:
1014:
1010:
1004:
1000:
994:
990:
983:
979:
972:
968:
964:
961:
958:
955:
950:
946:
939:
933:
930:
925:
918:
914:
910:
901:
900:
898:
878:
874:
868:
864:
858:
851:
847:
840:
836:
832:
827:
823:
816:
809:
805:
801:
792:
791:
789:
769:
765:
758:
754:
750:
745:
741:
737:
732:
728:
721:
714:
710:
706:
697:
696:
694:
693:
692:
690:
686:
685:
674:
672:
668:
664:
660:
652:
648:
644:
641:
633:
629:
628:
627:
605:
601:
597:
592:
588:
584:
579:
575:
570:
565:
560:
557:
554:
550:
542:
541:
540:
538:
534:
531:
527:
523:
519:
509:
507:
503:
499:
491:
487:
483:
480:
477:
474:
471:
467:
463:
459:
458:
453:
446:
443:
440:
436:
433:
430:
426:
423:
422:
421:
419:
415:
411:
407:
403:
399:
365:
362:
359:
342:
339:
333:
330:
323:
322:
321:
319:
315:
311:
307:
302:
296:
292:
285:
281:
274:
270:
263:
259:
254:
252:
248:
244:
240:
231:
215:
210:
203:
198:
191:
186:
185:
184:
182:
178:
171:
164:
157:
153:
149:
145:
141:
134:
124:
122:
118:
114:
109:
107:
103:
99:
95:
91:
86:
84:
80:
76:
71:
69:
65:
61:
57:
53:
49:
45:
37:
32:
19:
8165:(isometric)
8128:orthorhombic
8030:
8018:
7963:Associations
7931:Organisation
7423:Disclination
7354:Polymorphism
7317:Quasicrystal
7260:Orthorhombic
7200:Miller index
7181:
7148:Key concepts
7088:
6889:
6865:
6859:
6840:
6834:
6801:
6797:
6791:
6764:
6760:
6750:
6707:
6702:
6696:
6663:
6657:
6651:
6618:
6612:
6606:
6579:
6573:
6563:
6546:
6542:
6536:
6511:
6507:
6501:
6484:
6479:
6470:
6451:
6445:
6412:
6408:
6402:
6377:
6373:
6367:
6347:
6340:
6321:
6287:
6253:
6221:
6214:Ashcroft, N.
6208:
6181:
6175:
6163:. Retrieved
6161:. 6 Apr 2017
6158:
6149:
6122:
6103:
6097:
6076:
6042:Seed crystal
5788:
5784:pyroelectric
5760:
5728:
5724:
5706:
5675:
5627:Polymorphism
5625:
5612:cristobalite
5575:Polymorphism
5569:
5564:Hume-Rothery
5561:
5500:
5489:
5481:
5454:
5431:
5423:
5419:dislocations
5415:
5400:cold working
5393:
5374:
5370:adding to it
5365:
5349:Dislocations
5331:
5318:
5314:adding to it
5309:
5287:
5268:
5258:
5254:
5230:
5226:adding to it
5221:
5200:
5130:Simple cubic
5088:
5085:
4965:
4946:
4941:
4939:
4933:
4926:
4921:
4919:
4913:
4905:close-packed
4902:
4879:
4871:
4866:Glide planes
4865:
4859:
4854:translations
4853:
4843:
4840:Space groups
4833:
4831:
4823:
4818:
4814:
4810:
4805:
4801:
4796:
4792:
4785:
4779:
4776:Point groups
4771:
4493:tetartoidal
2834:orthorhombic
2481:
2461:
2459:
2450:
2445:
2428:orthorhombic
2420:rhombohedral
2397:
2390:
2380:
2368:
2338:
2311:
2282:
2270:
2246:
2236:
2226:
2216:
2200:Orthorhombic
2188:
2178:
2148:
2103:Point group
2082:
2069:
1402:Monoclinic:
790:Tetragonal:
688:
683:
682:
681:The spacing
680:
656:
650:
646:
639:
631:
625:
536:
532:
521:
517:
515:
505:
497:
495:
470:crystallites
417:
395:
313:
309:
305:
303:
290:
283:
279:
272:
268:
261:
257:
255:
250:
246:
242:
239:Miller index
236:
173:
166:
159:
151:
147:
143:
136:
110:
106:space groups
97:
93:
87:
72:
47:
41:
8195:rectangular
8109:(anorthic)
7916:Ewald Prize
7684:Diffraction
7662:Diffraction
7645:Diffraction
7587:Bragg plane
7582:Bragg's law
7461:Dislocation
7376:Segregation
7288:Crystallite
7205:Point group
7003:Performance
6142:R.G. Lerner
5588:crystalline
5557:axial ratio
5390:Dislocation
5377:August 2022
5321:August 2022
5259:tetrahedral
5233:August 2022
5122:Tetrahedron
4846:space group
4508:alternating
2994:dipyramidal
2644:sphenoidal
2582:pinacoidal
2499:Point group
1602:Triclinic:
899:Hexagonal:
506:basal plane
486:Dislocation
181:space group
79:translation
70:in matter.
8235:Categories
8138:tetragonal
8118:monoclinic
7700:Algorithms
7689:Scattering
7667:Scattering
7650:Scattering
7518:Slip bands
7481:Cross slip
7331:transition
7265:Tetragonal
7255:Monoclinic
7167:Metallurgy
7035:Composites
7000:Processing
6997:Properties
6476:L. Pauling
6331:084122725X
6218:Mermin, D.
6056:References
5959:Metallurgy
5775:dielectric
5721:allotropes
5717:allotropes
5673:patterns.
5620:stishovite
5462:plasticity
5438:electrical
5434:Hall–Petch
5293:Impurities
5257:(red) and
5255:Octahedral
5139:Octahedron
4952:APF and CN
4860:Screw axes
4793:Reflection
4542:diploidal
3944:hexagonal
3058:tetragonal
2935:Klein four
2857:Klein four
2776:Klein four
2640:monoclinic
2505:Schönflies
2476:See also:
2432:monoclinic
2416:tetragonal
2324:Hexagonal
2254:Tetragonal
2162:Monoclinic
661:(fcc) and
439:reactivity
435:Adsorption
36:table salt
8261:Structure
8215:hexagonal
8107:triclinic
7807:Databases
7270:Triclinic
7250:Hexagonal
7190:Unit cell
7182:Structure
6994:Structure
6826:120417927
5746:germanium
5729:white tin
5690:silicates
5643:allotropy
5608:tridymite
5590:forms of
5458:diffusion
5067:unit cell
4934:ABCABCABC
4811:Inversion
4743:×
4673:symmetric
4621:symmetric
4606:gyroidal
4578:×
4460:×
4396:⋊
4314:⋊
4232:⋊
4150:×
4086:×
4004:×
3916:⋊
3834:⋊
3752:⋊
3670:×
3553:trigonal
3549:hexagonal
3520:×
3456:⋊
3369:⋊
3287:⋊
3205:×
3029:×
2964:×
2916:pyramidal
2886:×
2805:×
2757:prismatic
2525:triclinic
2436:triclinic
2412:hexagonal
2292:Hexagonal
2132:Triclinic
2039:γ
2036:
2030:β
2027:
2021:α
2018:
2006:γ
2003:
1990:−
1987:β
1984:
1971:−
1968:α
1965:
1952:−
1941:γ
1938:
1932:−
1929:β
1926:
1920:α
1917:
1879:β
1876:
1870:−
1867:α
1864:
1858:γ
1855:
1835:ℓ
1817:α
1814:
1808:−
1805:γ
1802:
1796:β
1793:
1773:ℓ
1758:γ
1755:
1724:ℓ
1715:β
1712:
1672:α
1669:
1586:β
1583:
1554:β
1551:
1545:ℓ
1533:−
1512:ℓ
1490:β
1487:
1368:ℓ
1259:α
1256:
1237:α
1234:
1218:−
1194:α
1191:
1185:−
1182:α
1179:
1160:ℓ
1151:ℓ
1127:α
1124:
1102:ℓ
1001:ℓ
865:ℓ
755:ℓ
667:supercell
638:denote a
576:ℓ
555:ℓ
410:molecules
366:ℓ
343:π
133:Unit cell
127:Unit cell
75:unit cell
60:molecules
8256:Crystals
8020:Category
7855:Journals
7787:OctaDist
7782:JANA2020
7754:Software
7640:Electron
7557:F-center
7344:Eutectic
7305:Fiveling
7300:Twinning
7293:Equiaxed
7052:Polymers
7018:Ceramics
6742:54966950
6688:94113118
6643:95981632
5945:See also
5830:COONa.3H
5725:gray tin
5684:, or SiO
5680:form of
5635:minerals
5631:polymers
5604:β-quartz
5600:α-quartz
5265:lattice.
5105:Geometry
4914:ABABABAB
4802:Rotation
4360:dihedral
4278:dihedral
4196:dihedral
3880:dihedral
3798:dihedral
3716:dihedral
3420:dihedral
3333:dihedral
3251:dihedral
2992:rhombic-
2914:rhombic-
2699:domatic
2549:trivial
634:such as
476:Cleavage
295:integers
113:cleavage
102:symmetry
8185:oblique
8032:Commons
7980:Germany
7657:Neutron
7547:Vacancy
7406:Defects
7391:GP-zone
7237:Systems
6806:Bibcode
6769:Bibcode
6722:Bibcode
6668:Bibcode
6623:Bibcode
6584:Bibcode
6516:Bibcode
6437:9896377
6417:Bibcode
6382:Bibcode
5742:silicon
5738:diamond
5713:brittle
5616:coesite
5550:⁄
5540:⁄
5530:⁄
5520:⁄
5510:⁄
5501:In the
2529:pedial
695:Cubic:
462:defects
8205:square
7975:France
7970:Europe
7903:Awards
7433:Growth
7283:Growth
7045:Alloys
6872:
6847:
6824:
6740:
6686:
6641:
6458:
6435:
6355:
6328:
6296:
6260:
6228:
6196:
6165:18 May
6110:
6085:
5961:, and
5678:quartz
5663:stable
5639:metals
5637:, and
5618:, and
5592:silica
5584:Quartz
5343:alloys
5185:(HCP)
5167:(FCC)
5149:(BCC)
4050:cyclic
3968:cyclic
3634:cyclic
3577:cyclic
3135:cyclic
3083:cyclic
2722:cyclic
2665:cyclic
2605:cyclic
651:braces
640:family
418:planes
414:planes
282:, and
249:, and
119:, and
100:. The
8163:cubic
7997:Japan
7944:IOBCr
7797:SHELX
7792:Olex2
7679:X-ray
7329:Phase
7245:Cubic
7040:Metal
7023:Glass
6822:S2CID
6738:S2CID
6712:arXiv
6684:S2CID
6639:S2CID
5780:polar
5594:, SiO
5450:creep
5396:shear
5188:0.74
5170:0.74
5152:0.68
5134:0.52
5117:0.34
4852:Pure
4489:cubic
4270:polar
3960:polar
3790:polar
3569:polar
3325:polar
3075:polar
2927:polar
2714:polar
2657:polar
2542:polar
2513:Order
2462:basis
2400:cubic
2352:Cubic
466:Pores
402:atoms
398:lines
62:in a
58:, or
52:atoms
7939:IUCr
7840:ICDD
7835:ICSD
7820:CCDC
7767:Coot
7762:CCP4
7513:Slip
7476:Kink
6870:ISBN
6845:ISBN
6456:ISBN
6433:PMID
6353:ISBN
6326:ISBN
6294:ISBN
6258:ISBN
6226:ISBN
6194:ISBN
6167:2017
6108:ISBN
6083:ISBN
5936:Ruby
5908:NaCl
5866:CsCl
5727:and
5460:and
5440:and
5191:12 (
5173:12 (
5157:Cube
4960:and
4780:The
2845:(V)
2434:and
2404:cube
2354:(c)
2294:(h)
2256:(t)
2202:(o)
2164:(m)
2134:(a)
657:For
468:and
437:and
406:ions
56:ions
7954:DMG
7949:RAS
7845:PDB
7830:COD
7825:CIF
7777:DSR
7501:GND
7428:CSL
6814:doi
6777:doi
6730:doi
6676:doi
6664:240
6631:doi
6619:208
6592:doi
6580:196
6551:doi
6524:doi
6489:doi
6425:doi
6390:doi
6378:188
6186:doi
5893:TiO
5878:CaF
5854:ONa
5744:or
5709:tin
5657:or
5372:.
5316:.
5228:.
5155:8 (
5137:6 (
5120:4 (
4936:...
4932:...
4916:...
4912:...
4817:or
4784:or
4721:48
4669:24
4617:24
4556:24
4504:12
4438:24
4356:12
4274:12
4192:12
4128:12
3876:12
3498:16
2426:),
2391:cF
2381:cI
2369:cP
2339:hP
2312:hR
2283:tI
2271:tP
2247:oF
2237:oI
2227:oS
2217:oP
2189:mS
2179:mP
2149:aP
2088:.
2033:cos
2024:cos
2015:cos
1994:cos
1975:cos
1956:cos
1935:cos
1923:cos
1914:cos
1873:cos
1861:cos
1852:cos
1811:cos
1799:cos
1790:cos
1746:sin
1703:sin
1660:sin
1574:csc
1548:cos
1478:sin
1247:cos
1225:cos
1188:cos
1170:cos
1115:sin
1039:):
689:hkℓ
649:or
522:ℓmn
408:or
306:hkℓ
258:hkℓ
96:or
42:In
8237::
7992:US
7985:UK
6820:.
6812:.
6802:38
6800:.
6775:.
6765:46
6763:.
6759:.
6736:.
6728:.
6720:.
6708:93
6682:.
6674:.
6662:.
6637:.
6629:.
6617:.
6590:.
6578:.
6572:.
6547:69
6545:.
6522:.
6512:54
6510:.
6485:51
6431:.
6423:.
6413:32
6411:.
6388:.
6376:.
6308:^
6272:^
6252:.
6240:^
6216:;
6192:.
6184:.
6157:.
6131:^
6063:^
5957:,
5953:,
5826:CH
5740:,
5704:.
5653:,
5633:,
5614:,
5610:,
5606:,
5602:,
5545:,
5535:,
5525:,
5515:,
5468:.
5452:.
5402:.
5274:.
5195:)
5177:)
5159:)
5141:)
5124:)
5107:)
4944:.
4924:.
4609:O
4496:T
4455:12
4429:6h
4376:12
4347:3h
4294:12
4265:6v
4212:12
4119:6h
4046:6
4037:3h
3964:6
3896:12
3867:3d
3794:6
3785:3v
3712:6
3630:6
3622:)
3618:(S
3616:3i
3573:3
3489:4h
3416:8
3408:)
3404:(V
3402:2d
3329:8
3320:4v
3247:8
3183:8
3174:4h
3131:4
3079:4
3014:8
3006:)
3002:(V
3000:2h
2931:4
2922:2v
2853:4
2772:4
2763:2h
2718:2
2710:)
2708:1h
2706:(C
2661:2
2601:2
2593:)
2589:(S
2546:1
2468:.
2438:.
2430:,
2418:,
2414:,
2329:6h
2302:3d
2261:4h
2207:2h
2169:2h
2109:)
673:.
484::
464::
427::
404:,
271:,
245:,
172:,
165:,
150:,
146:,
123:.
115:,
85:.
54:,
46:,
8067:e
8060:t
8053:v
7133:e
7126:t
7119:v
6972:e
6965:t
6958:v
6878:.
6853:.
6828:.
6816::
6808::
6785:.
6779::
6771::
6744:.
6732::
6724::
6714::
6690:.
6678::
6670::
6645:.
6633::
6625::
6600:.
6594::
6586::
6557:.
6553::
6530:.
6526::
6518::
6495:.
6491::
6464:.
6439:.
6427::
6419::
6396:.
6392::
6384::
6361:.
6334:.
6302:.
6266:.
6234:.
6202:.
6188::
6169:.
6116:.
6091:.
5965:.
5924:)
5922:2
5895:2
5880:2
5852:5
5850:H
5848:2
5846:C
5834:O
5832:2
5828:3
5702:2
5698:4
5694:4
5686:2
5622:.
5596:2
5552:4
5548:3
5542:4
5538:1
5532:3
5528:2
5522:3
5518:1
5512:2
5508:1
5379:)
5375:(
5323:)
5319:(
5235:)
5231:(
5103:(
5063:V
5055:e
5052:l
5049:c
5046:i
5043:t
5040:r
5037:a
5034:p
5029:V
5022:e
5019:l
5016:c
5013:i
5010:t
5007:r
5004:a
5001:p
4996:N
4989:=
4985:F
4982:P
4979:A
4753:2
4748:Z
4738:4
4733:S
4712:h
4710:O
4689:4
4684:S
4660:d
4658:T
4637:4
4632:S
4588:2
4583:Z
4573:4
4568:A
4547:h
4545:T
4524:4
4519:A
4470:2
4465:Z
4450:D
4427:D
4406:2
4401:Z
4391:6
4386:Z
4381:=
4371:D
4345:D
4324:2
4319:Z
4309:6
4304:Z
4299:=
4289:D
4263:C
4242:2
4237:Z
4227:6
4222:Z
4217:=
4207:D
4183:6
4181:D
4160:2
4155:Z
4145:6
4140:Z
4117:C
4096:2
4091:Z
4081:3
4076:Z
4071:=
4066:6
4061:Z
4035:C
4014:2
4009:Z
3999:3
3994:Z
3989:=
3984:6
3979:Z
3952:6
3950:C
3926:2
3921:Z
3911:6
3906:Z
3901:=
3891:D
3865:D
3844:2
3839:Z
3829:3
3824:Z
3819:=
3814:6
3809:D
3783:C
3762:2
3757:Z
3747:3
3742:Z
3737:=
3732:6
3727:D
3703:3
3701:D
3680:2
3675:Z
3665:3
3660:Z
3655:=
3650:6
3645:Z
3620:6
3614:C
3593:3
3588:Z
3561:3
3559:C
3530:2
3525:Z
3515:8
3510:D
3487:D
3466:2
3461:Z
3451:4
3446:Z
3441:=
3436:8
3431:D
3406:d
3400:D
3379:2
3374:Z
3364:4
3359:Z
3354:=
3349:8
3344:D
3318:C
3297:2
3292:Z
3282:4
3277:Z
3272:=
3267:8
3262:D
3238:4
3236:D
3215:2
3210:Z
3200:4
3195:Z
3172:C
3151:4
3146:Z
3122:4
3120:S
3099:4
3094:Z
3067:4
3065:C
3039:2
3034:Z
3025:V
3004:h
2998:D
2974:2
2969:Z
2959:2
2954:Z
2949:=
2945:V
2920:C
2896:2
2891:Z
2881:2
2876:Z
2871:=
2867:V
2843:2
2841:D
2815:2
2810:Z
2800:2
2795:Z
2790:=
2786:V
2761:C
2738:2
2733:Z
2704:s
2702:C
2681:2
2676:Z
2649:2
2647:C
2621:2
2616:Z
2591:2
2587:i
2585:C
2564:1
2559:Z
2534:1
2532:C
2359:h
2357:O
2327:D
2300:D
2259:D
2205:D
2167:C
2139:i
2137:C
2105:(
2012:2
2009:+
1998:2
1979:2
1960:2
1949:1
1944:)
1911:(
1905:b
1902:a
1897:k
1894:h
1891:2
1885:+
1882:)
1849:(
1843:c
1840:a
1832:h
1829:2
1823:+
1820:)
1787:(
1781:c
1778:b
1770:k
1767:2
1761:+
1750:2
1738:2
1734:c
1728:2
1718:+
1707:2
1695:2
1691:b
1685:2
1681:k
1675:+
1664:2
1652:2
1648:a
1642:2
1638:h
1629:=
1622:2
1618:d
1614:1
1578:2
1569:)
1562:c
1559:a
1542:h
1539:2
1526:2
1522:c
1516:2
1506:+
1499:2
1495:b
1482:2
1472:2
1468:k
1461:+
1454:2
1450:a
1444:2
1440:h
1433:(
1429:=
1422:2
1418:d
1414:1
1382:2
1378:c
1372:2
1362:+
1355:2
1351:b
1345:2
1341:k
1335:+
1328:2
1324:a
1318:2
1314:h
1308:=
1301:2
1297:d
1293:1
1262:)
1251:3
1243:2
1240:+
1229:2
1221:3
1215:1
1212:(
1207:2
1203:a
1197:)
1174:2
1166:(
1163:)
1157:h
1154:+
1148:k
1145:+
1142:k
1139:h
1136:(
1133:2
1130:+
1119:2
1111:)
1106:2
1098:+
1093:2
1089:k
1085:+
1080:2
1076:h
1072:(
1066:=
1059:2
1055:d
1051:1
1015:2
1011:c
1005:2
995:+
991:)
984:2
980:a
973:2
969:k
965:+
962:k
959:h
956:+
951:2
947:h
940:(
934:3
931:4
926:=
919:2
915:d
911:1
879:2
875:c
869:2
859:+
852:2
848:a
841:2
837:k
833:+
828:2
824:h
817:=
810:2
806:d
802:1
770:2
766:a
759:2
751:+
746:2
742:k
738:+
733:2
729:h
722:=
715:2
711:d
707:1
684:d
606:2
602:n
598:+
593:2
589:m
585:+
580:2
571:a
566:=
561:n
558:m
551:d
537:d
533:a
518:a
372:|
363:k
360:h
355:g
349:|
340:2
334:=
331:d
314:d
299:2
291:ℓ
289:/
287:3
284:a
280:k
278:/
276:2
273:a
269:h
267:/
265:1
262:a
251:ℓ
247:k
243:h
176:i
174:z
169:i
167:y
162:i
160:x
158:(
152:c
148:b
144:a
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.