491:
example, Sharpless provided evidence for the reaction proceeding via a step-wise mechanism. Additionally both
Sharpless and Corey showed that the active catalyst possesses a U-shaped chiral binding pocket. Corey also showed that the catalyst obeys Michaelis-Menten kinetics and acts like an enzyme pocket with a pre-equilibrium. In the February 1997 issue of the Journal of the American Chemical Society Sharpless published the results of a study (a Hammett analysis) which he claimed supported a cyclization over a . In the October issue of the same year, however, Sharpless also published the results of another study conducted in collaboration with Ken Houk and Singleton which provided conclusive evidence for the mechanism. Thus Sharpless was forced to concede the decade-long debate.
483:
438:
426:
197:
500:
643:
626:
614:
434:
dihydroxylate another alkene. Dihydroxylations resulting from this secondary pathway generally suffer lower enantioselectivities than those resulting from the primary pathway. A schematic showing this secondary catalytic pathway is shown below. This secondary pathway may be suppressed by using a higher molar concentration of ligand.
621:
In this example SAD gives the diol of the alkene closest to the (electron-withdrawing) para-methoxybenzoyl group, albeit in low yield. This is likely due to the ability of the aryl ring to interact favorably with the active site of the catalyst via π-stacking. In this manner the aryl substituent can
594:
Citric acid: Osmium tetroxide is an electrophilic oxidant and as such reacts slowly with electron-deficient olefins. It has been found that the rate of oxidation of electron-deficient olefins can be accelerated by maintaining the pH of the reaction slightly acidic. On the other hand, a high pH can
490:
The next ten years saw numerous publications by both Corey and
Sharpless, each supporting their own version of the mechanism. While these studies were not able to distinguish between the two proposed cyclization pathways, they were successful in shedding light on the mechanism in other ways. For
638:
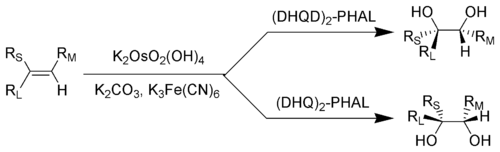
The diastereoselectivity of SAD is set primarily by the choice of ligand (i.e. AD-mix-α versus AD-mix-β), however factors such as pre-existing chirality in the substrate or neighboring functional groups may also play a role. In the example shown below, the para-methoxybenzoyl substituent serves
1240:
Corey, E. J.; DaSilva
Jardine, Paul; Virgil, Scott; Yuen, Po Wai; Connell, Richard D. (December 1989). "Enantioselective vicinal hydroxylation of terminal and E-1,2-disubstituted olefins by a chiral complex of osmium tetroxide. An effective controller system and a rational mechanistic model".
1396:
Sharpless, K. B.; Gypser, Andreas; Ho, Pui Tong; Kolb, Hartmuth C.; Kondo, Teruyuki; Kwong, Hoi-Lun; McGrath, Dominic V.; Rubin, A. Erik; Norrby, Per-Ola; Gable, Kevin P.; Sharpless, K. Barry (1997). "Toward an
Understanding of the High Enantioselectivity in the Osmium-Catalyzed Asymmetric
433:
The mechanism of the
Sharpless asymmetric dihydroxylation has been extensively studied and a potential secondary catalytic cycle has been identified (see below). If the osmylate ester intermediate is oxidized before it dissociates, then an osmium(VIII)-diol complex is formed which may then
478:
with the alkene to directly generate the osmylate ester. Corey's suggestion was based on a previous computational study done by
Jorgensen and Hoffmann which determined the reaction pathway to be the lower energy pathway. In addition Corey reasoned that steric repulsions in the octahedral
1456:
Corey, E. J.; Guzman-Perez, Angel; Noe, Mark C. (November 1995). "The application of a mechanistic model leads to the extension of the
Sharpless asymmetric dihydroxylation to allylic 4-methoxybenzoates and conformationally related amine and homoallylic alcohol derivatives".
1334:
Corey, E. J.; Noe, Mark C.; Sarshar, Sepehr (1994). "X-ray crystallographic studies provide additional evidence that an enzyme-like binding pocket is crucial to the enantioselective dihydroxylation of olefins by OsO4-bis-cinchona alkaloid complexes".
1367:
Corey, E. J.; Noe, M. C. (17 January 1996). "Kinetic
Investigations Provide Additional Evidence That an Enzyme-like Binding Pocket Is Crucial for High Enantioselectivity in the Bis-Cinchona Alkaloid Catalyzed Asymmetric Dihydroxylation of Olefins".
473:
to form an osmylate ester which after hydrolysis would give the corresponding diol. In 1989 E. J. Corey published a slightly different variant of this reaction and suggested that the reaction most likely proceeded via a cycloaddition of
1131:
Ogino, Y.; Chen, H.; Kwong, H.-L.; Sharpless, K. B. (1991). "On the timing of hydrolysis / reoxidation in the osmium-catalyzed asymmetric dihydroxylation of olefins using potassium ferricyanide as the reoxidant".
308:) are expensive and extremely toxic, it has become desirable to develop catalytic variants of this reaction. Some stoichiometric terminal oxidants that have been employed in these catalytic reactions include
1159:
Wai, J. S. M.; Marko, I.; Svendsen, J. N.; Finn, M. G.; Jacobsen, E. N.; Sharpless, K. Barry (1989). "A mechanistic insight leads to a greatly improved osmium-catalyzed asymmetric dihydroxylation process".
1103:; Singleton, D. A.; Strassner, T.; Thomas, A. A. (1997). "Experimental and Theoretical Kinetic Isotope Effects for Asymmetric Dihydroxylation. Evidence Supporting a Rate-Limiting "(3 + 2)" Cycloaddition".
414:) has been identified as a catalyst to accelerate this step of the catalytic cycle and if frequently used as an additive to allow non-terminal alkene substrates to react efficiently at 0 °C. Finally, the
1064:; Noe, M. C.; Grogan, M. J. (1996). "Experimental test of the - and -cycloaddition pathways for the bis-cinchona alkaloid-OsO4 catalyzed dihydroxylation of olefins by means of kinetic isotope effects".
1278:
Corey, E. J.; Noe, Mark C. (December 1993). "Rigid and highly enantioselective catalyst for the dihydroxylation of olefins using osmium tetraoxide clarifies the origin of enantiospecificity".
1306:
Kolb, H. C.; Anderson, P. G.; Sharpless, K. B. (February 1994). "Toward an
Understanding of the High Enantioselectivity in the Osmium-Catalyzed Asymmetric Dihydroxylation (AD). 1. Kinetics".
1195:
Sundermeier, U., Dobler, C., Beller, M. Recent developments in the osmium-catalyzed dihydroxylation of olefins. Modern
Oxidation Methods. 2004 WILEY-VCH Verlag GmbH & Co. KGaA,Weinheim.
193:(DHQ) as the ligand. Asymmetric dihydroxylation reactions are also highly site selective, providing products derived from reaction of the most electron-rich double bond in the substrate.
482:
519:
Crystallographic evidence has shown that the active catalyst possesses a pentacoordinate osmium species held in a U-shaped binding pocket. The nitrogenous ligand holds OsO
437:
859:
Minato, M.; Yamamoto, K.; Tsuji, J. (1990). "Osmium tetraoxide catalyzed vicinal hydroxylation of higher olefins by using hexacyanoferrate(III) ion as a cooxidant".
531:
Numerous catalytic systems and modifications have been developed for the SAD. Given below is a brief overview of the various components of the catalytic system:
370:
are combined with a stoichiometric ferricyanide oxidant in the presence of chiral nitrogenous ligands to create an asymmetric environment around the oxidant.
1033:; Amberg, Willi; Bennani, Youssef L.; et al. (1992). "The osmium-catalyzed asymmetric dihydroxylation: A new ligand class and a process improvement".
425:
586:) is the most commonly used stoichiometric oxidant for the reaction, and is the oxidant that comes in the commercially available AD-mix preparations.
132:
218:
It is common practice to perform this reaction using a catalytic amount of osmium tetroxide, which after reaction is regenerated with
1212:
Hentges, Steven G.; Sharpless, K. Barry (June 1980). "Asymmetric induction in the reaction of osmium tetroxide with olefins".
378:
The reaction mechanism of the Sharpless dihydroxylation begins with the formation of the osmium tetroxide – ligand complex (
1200:
1495:
1425:
Xu, D.; Crispino, G. A.; Sharpless, K. B. (September 1992). "Selective asymmetric dihydroxylation (AD) of dienes".
117:
758:
736:
Noe, Mark C.; Letavic, Michael A.; Snow, Sheri L. (15 December 2005). "Asymmetric Dihydroxylation of Alkenes".
696:
610:
In general Sharpless asymmetric dihydroxylation favors oxidation of the more electron-rich alkene (scheme 1).
523:
in a chiral environment making approach of one side of the olefin sterically hindered while the other is not.
716:
711:
1490:
639:
primarily as a source of steric bulk to allow the catalyst to differentiate the two faces of the alkene.
539:, however certain additives can coordinate to the osmium(VIII) and modify its electronic properties. OsO
469:
onto the alkene to give an osmaoxetane intermediate (see below). This intermediate would then undergo a
366:
alkene dihydroxylation, referred to as the Sharpless asymmetric dihydroxylation (SAD). Low levels of OsO
296:
is an old and extremely useful method for the functionalization of olefins. However, since osmium(VIII)
321:
227:
125:
241:
and very expensive osmium tetroxide needed. These four reagents are commercially available premixed ("
564:
Peroxides were among the first stoichiometric oxidants to be used in this catalytic cycle; see the
336:
281:
1500:
706:
572:
347:
332:
277:
223:
165:
359:
30:
8:
1337:
701:
596:
565:
470:
317:
175:
1100:
1030:
976:
888:
775:
665:
309:
273:
181:. The reaction has been applied to alkenes of virtually every substitution, often high
1350:
961:
654:-disubstituted alkenes when both ends of the olefin have similar steric environments.
1459:
1427:
1399:
1370:
1308:
1280:
1243:
1214:
1196:
1145:
1105:
1078:
1066:
1061:
993:
949:
905:
832:
754:
670:
313:
269:
257:
153:
40:
642:
625:
613:
1467:
1435:
1407:
1378:
1346:
1316:
1288:
1251:
1222:
1169:
1141:
1113:
1074:
1043:
1002:
957:
914:
869:
841:
788:
746:
678:
508:
504:
499:
363:
301:
293:
196:
186:
182:
161:
980:
892:
807:
750:
415:
190:
1484:
1035:
1006:
918:
861:
845:
462:
1096:
939:
VanRheenen, V.; Kelly, R. C.; Cha, D. Y. (1976). "An improved catalytic OsO
1397:
Dihydroxylation. 4. Electronic Effects in Amine-Accelerated Osmylations".
595:
increase the rate of oxidation of internal olefins, and also increase the
738:
1471:
1439:
1320:
1292:
1255:
1226:
1173:
1047:
873:
792:
682:
461:
In his original report Sharpless suggested the reaction proceeded via a
780:
668:(1988). "Asymmetric dihydroxylation via ligand-accelerated catalysis".
391:
1411:
1382:
1117:
719:- olefin to diol, followed by oxidative cleavage to form two aldehydes
503:
Allyl benzoate bound within the U-shaped binding pocket of the active
219:
828:-2-Phenylcyclohexanol Via Sharpless Asymmetric Dihydroxylation (AD)"
265:
261:
185:
are realized, with the chiral outcome controlled by the choice of
806:
Gonzalez, Javier; Aurigemma, Christine; Truesdale, Larry (2004).
558:
Chiral Auxiliary: This is usually some kind of cinchona alkaloid.
297:
168:
486:
Osmium tetroxide dihydroxylation proposed and correct mechanism
242:
171:
157:
1239:
1029:
663:
650:
It is often difficult to obtain high diastereoselectivity on
238:
1094:
773:
664:
Jacobsen, E. N.; Marko, I.; Mungall, W. S.; Schroeder, G.;
441:
Catalytic cycle of the Sharpless asymmetric dihydroxylation
418:
oxidant regenerates the osmium tetroxide – ligand complex (
249:-PHAL is called AD-mix-α, and the mixture containing (DHQD)
178:
95:
947:-1,2-glycols using tertiary amine oxides as the oxidant".
805:
568:. Drawbacks of peroxides include chemoselectivity issues.
1130:
429:
The reaction mechanism of the Sharpless dihydroxylation
1362:
1360:
974:
1395:
1305:
237:. This dramatically reduces the amount of the highly
1455:
1158:
1424:
1357:
858:
938:
1268:Thomas, G.; Sharpless, K. B. ACIEE 1993, 32, 1329
571:Trialkylammonium N-oxides, such as NMO—as in the
1482:
1211:
778:(1994). "Catalytic Asymmetric Dihydroxylation".
1333:
280:, for which he was awarded a share of the 2001
260:. The introduction of chirality into nonchiral
1060:
989:)-1,2-Diphenyl-1,2-ethanediol (Stilbene diol)"
735:
272:. This reaction was developed principally by
1191:
1189:
1187:
1185:
1183:
1090:
1088:
599:(e.e.) for the oxidation of terminal olefins.
362:was the first to develop a general, reliable
886:
555:(an Os(VI) species) due to safety concerns.
1180:
1085:
479:intermediate would disfavor the pathway.
1366:
1277:
498:
195:
51:
897:)-1,2-Dihydroxyethyl]-1,5-Dihydro-3
1483:
382:). A -cycloaddition with the alkene (
276:building on the already known racemic
1451:
1449:
799:
535:Catalytic Oxidant: This is always OsO
494:
373:
22:Sharpless asymmetric dihydroxylation
1025:
1023:
774:Kolb, H. C.; Van Nieuwenhze, M. S.;
633:
526:
146:Sharpless asymmetric dihydroxylation
767:
605:
256:Such chiral diols are important in
13:
1446:
657:
641:
624:
612:
481:
436:
424:
14:
1512:
1020:
729:
543:is often generated in situ from K
386:) gives the cyclic intermediate
245:"). The mixture containing (DHQ)
1418:
1389:
1327:
1299:
1271:
1262:
1233:
1205:
1152:
1124:
975:McKee, B. H.; Gilheany, D. G.;
1054:
968:
932:
880:
852:
697:Asymmetric catalytic oxidation
445:
200:The Sharpless dihydroxylation.
1:
1351:10.1016/s0040-4039(00)76644-5
1095:DelMonte, A. J.; Haller, J.;
962:10.1016/s0040-4039(00)78093-2
723:
287:
210:= Medium-sized substituent; R
1146:10.1016/0040-4039(91)80601-2
1079:10.1016/0040-4039(96)01005-2
712:Sharpless aminohydroxylation
575:—and trimethylamine N-oxide.
7:
751:10.1002/0471264180.or066.02
690:
268:is an important concept in
10:
1517:
1015:, vol. 9, p. 383
927:, vol. 9, p. 251
622:act as a directing group.
402:). Methanesulfonamide (CH
398:) and the reduced osmate (
292:Alkene dihydroxylation by
253:-PHAL is called AD-mix-β.
150:Sharpless bishydroxylation
717:Lemieux–Johnson oxidation
578:Potassium ferricyanide (K
139:
118:sharpless-dihydroxylation
113:Organic Chemistry Portal
107:
92:
83:
74:
62:
55:
46:
21:
1007:10.15227/orgsyn.070.0047
943:oxidation of olefins to
919:10.15227/orgsyn.073.0001
846:10.15227/orgsyn.079.0093
561:Stoichiometric Oxidant:
471:1,1- migratory insertion
282:Nobel Prize in Chemistry
264:through usage of chiral
206:= Largest substituent; R
1496:Organic redox reactions
707:Upjohn dihydroxylation
647:
630:
618:
516:
487:
442:
430:
348:potassium ferricyanide
333:Upjohn dihydroxylation
278:Upjohn dihydroxylation
224:potassium ferricyanide
215:
214:= Smallest substituent
645:
628:
616:
511:interacting with the
502:
485:
440:
428:
199:
164:in the presence of a
78:Chiral quinine ligand
901:-2,4-Benzodioxepine"
808:"Synthesis of (+)-(1
394:liberates the diol (
340:-butyl hydroperoxide
183:enantioselectivities
31:Karl Barry Sharpless
1472:10.1021/ja00149a003
1466:(44): 10805–10816.
1440:10.1021/ja00045a043
1338:Tetrahedron Letters
1321:10.1021/ja00083a014
1293:10.1021/ja00079a045
1256:10.1021/ja00208a025
1227:10.1021/ja00532a050
1174:10.1021/ja00185a050
1048:10.1021/jo00036a003
874:10.1021/jo00289a066
793:10.1021/cr00032a009
702:Milas hydroxylation
683:10.1021/ja00214a053
597:enantiomeric excess
566:Milas hydroxylation
318:Milas hydroxylation
1491:Addition reactions
648:
631:
619:
517:
495:Catalyst structure
488:
443:
431:
374:Reaction mechanism
360:K. Barry Sharpless
325:-Methylmorpholine
310:potassium chlorate
274:K. Barry Sharpless
231:-methylmorpholine
216:
1460:J. Am. Chem. Soc.
1434:(19): 7570–7571.
1428:J. Am. Chem. Soc.
1412:10.1021/ja961464t
1400:J. Am. Chem. Soc.
1383:10.1021/ja952567z
1371:J. Am. Chem. Soc.
1309:J. Am. Chem. Soc.
1281:J. Am. Chem. Soc.
1244:J. Am. Chem. Soc.
1215:J. Am. Chem. Soc.
1118:10.1021/ja971650e
1112:(41): 9907–9908.
1106:J. Am. Chem. Soc.
1073:(28): 4899–4902.
1067:Tetrahedron Lett.
1042:(10): 2768–2771.
1013:Collected Volumes
994:Organic Syntheses
956:(23): 1973–1976.
950:Tetrahedron Lett.
925:Collected Volumes
906:Organic Syntheses
833:Organic Syntheses
671:J. Am. Chem. Soc.
634:Stereoselectivity
527:Catalytic systems
314:hydrogen peroxide
270:organic synthesis
258:organic synthesis
154:chemical reaction
148:(also called the
143:
142:
103:
102:
41:Addition reaction
16:Chemical reaction
1508:
1476:
1475:
1453:
1444:
1443:
1422:
1416:
1415:
1393:
1387:
1386:
1364:
1355:
1354:
1331:
1325:
1324:
1303:
1297:
1296:
1275:
1269:
1266:
1260:
1259:
1237:
1231:
1230:
1209:
1203:
1193:
1178:
1177:
1162:J. Am. Chem. Soc
1156:
1150:
1149:
1140:(2): 3965–3968.
1134:Tetrahedron Lett
1128:
1122:
1121:
1101:Sharpless, K. B.
1092:
1083:
1082:
1058:
1052:
1051:
1031:Sharpless, K. B.
1027:
1018:
1016:
1009:
977:Sharpless, K. B.
972:
966:
965:
936:
930:
928:
921:
889:Sharpless, K. B.
884:
878:
877:
856:
850:
848:
803:
797:
796:
787:(8): 2483–2547.
776:Sharpless, K. B.
771:
765:
764:
745:(109): 109–625.
733:
686:
677:(6): 1968–1970.
666:Sharpless, K. B.
606:Regioselectivity
509:osmium tetroxide
505:dihydroquinidine
458:
457:
453:
450:vs debate": -->
364:enantioselective
302:osmium tetroxide
294:osmium tetroxide
187:dihydroquinidine
162:osmium tetroxide
135:
120:
87:
53:
52:
19:
18:
1516:
1515:
1511:
1510:
1509:
1507:
1506:
1505:
1481:
1480:
1479:
1454:
1447:
1423:
1419:
1394:
1390:
1365:
1358:
1332:
1328:
1304:
1300:
1276:
1272:
1267:
1263:
1238:
1234:
1210:
1206:
1194:
1181:
1157:
1153:
1129:
1125:
1093:
1086:
1059:
1055:
1028:
1021:
1011:
973:
969:
942:
937:
933:
923:
885:
881:
857:
853:
804:
800:
772:
768:
761:
734:
730:
726:
693:
660:
658:Further reading
636:
608:
585:
581:
573:Upjohn Reaction
554:
550:
546:
542:
538:
529:
522:
497:
477:
468:
459:
455:
451:
449:
448:
413:
409:
405:
376:
369:
357:
353:
307:
290:
252:
248:
213:
209:
205:
201:
131:
116:
85:
76:
69:
64:
17:
12:
11:
5:
1514:
1504:
1503:
1501:Name reactions
1498:
1493:
1478:
1477:
1445:
1417:
1388:
1356:
1326:
1315:(1278): 1278.
1298:
1287:(115): 12579.
1270:
1261:
1232:
1204:
1179:
1151:
1123:
1084:
1053:
1019:
967:
940:
931:
879:
868:(2): 766–768.
851:
798:
766:
759:
727:
725:
722:
721:
720:
714:
709:
704:
699:
692:
689:
688:
687:
659:
656:
635:
632:
607:
604:
603:
602:
601:
600:
589:
588:
587:
583:
579:
576:
569:
559:
556:
552:
548:
544:
540:
536:
528:
525:
520:
496:
493:
475:
466:
447:
444:
416:stoichiometric
411:
407:
403:
375:
372:
367:
355:
351:
305:
289:
286:
250:
246:
211:
207:
203:
191:dihydroquinine
141:
140:
137:
136:
129:
122:
121:
114:
110:
109:
105:
104:
101:
100:
98:(main product)
90:
89:
81:
80:
72:
71:
67:
60:
59:
49:
48:
44:
43:
38:
37:Reaction type
34:
33:
28:
24:
23:
15:
9:
6:
4:
3:
2:
1513:
1502:
1499:
1497:
1494:
1492:
1489:
1488:
1486:
1473:
1469:
1465:
1462:
1461:
1452:
1450:
1441:
1437:
1433:
1430:
1429:
1421:
1413:
1409:
1405:
1402:
1401:
1392:
1384:
1380:
1376:
1373:
1372:
1363:
1361:
1352:
1348:
1344:
1340:
1339:
1330:
1322:
1318:
1314:
1311:
1310:
1302:
1294:
1290:
1286:
1283:
1282:
1274:
1265:
1257:
1253:
1249:
1246:
1245:
1236:
1228:
1224:
1220:
1217:
1216:
1208:
1202:
1201:3-527-30642-0
1198:
1192:
1190:
1188:
1186:
1184:
1175:
1171:
1167:
1163:
1155:
1147:
1143:
1139:
1135:
1127:
1119:
1115:
1111:
1108:
1107:
1102:
1098:
1091:
1089:
1080:
1076:
1072:
1069:
1068:
1063:
1057:
1049:
1045:
1041:
1038:
1037:
1036:J. Org. Chem.
1032:
1026:
1024:
1014:
1008:
1004:
1000:
996:
995:
990:
988:
984:
978:
971:
963:
959:
955:
952:
951:
946:
935:
926:
920:
916:
912:
908:
907:
902:
900:
896:
890:
883:
875:
871:
867:
864:
863:
862:J. Org. Chem.
855:
847:
843:
839:
835:
834:
829:
827:
823:
819:
816:)- and (−)-(1
815:
811:
802:
794:
790:
786:
783:
782:
777:
770:
762:
756:
752:
748:
744:
741:
740:
732:
728:
718:
715:
713:
710:
708:
705:
703:
700:
698:
695:
694:
684:
680:
676:
673:
672:
667:
662:
661:
655:
653:
644:
640:
627:
623:
615:
611:
598:
593:
592:
590:
577:
574:
570:
567:
563:
562:
560:
557:
534:
533:
532:
524:
514:
510:
506:
501:
492:
484:
480:
472:
464:
463:cycloaddition
454:
439:
435:
427:
423:
421:
417:
401:
397:
393:
389:
385:
381:
371:
365:
361:
349:
345:
341:
339:
334:
330:
328:
324:
319:
315:
311:
303:
299:
295:
285:
283:
279:
275:
271:
267:
263:
259:
254:
244:
240:
236:
234:
230:
225:
221:
198:
194:
192:
188:
184:
180:
177:
173:
170:
167:
163:
159:
155:
151:
147:
138:
134:
130:
127:
124:
123:
119:
115:
112:
111:
106:
99:
97:
91:
88:
82:
79:
73:
70:
61:
58:
54:
50:
45:
42:
39:
36:
35:
32:
29:
26:
25:
20:
1463:
1458:
1431:
1426:
1420:
1403:
1398:
1391:
1374:
1369:
1345:(18): 2861.
1342:
1336:
1329:
1312:
1307:
1301:
1284:
1279:
1273:
1264:
1250:(26): 9243.
1247:
1242:
1235:
1221:(12): 4263.
1218:
1213:
1207:
1165:
1161:
1154:
1137:
1133:
1126:
1109:
1104:
1070:
1065:
1056:
1039:
1034:
1012:
998:
992:
986:
982:
970:
953:
948:
944:
934:
924:
910:
904:
898:
894:
882:
865:
860:
854:
837:
831:
825:
821:
817:
813:
809:
801:
784:
779:
769:
742:
737:
731:
674:
669:
651:
649:
646:SAD scheme 3
637:
629:SAD scheme 2
620:
617:SAD scheme 1
609:
530:
518:
512:
489:
460:
432:
419:
399:
395:
387:
383:
379:
377:
343:
337:
326:
322:
291:
255:
232:
228:
217:
149:
145:
144:
133:RXNO:0000142
128:ontology ID
108:Identifiers
93:
84:
77:
65:
56:
27:Named after
1406:(8): 1840.
1168:(3): 1123.
1097:Houk, K. N.
1062:Corey, E.J.
739:Org. React.
1485:Categories
1377:(2): 319.
893:"3-[(1
781:Chem. Rev.
760:0471264180
724:References
591:Additive:
507:catalyst,
446:vs debate
392:hydrolysis
346:BHP), and
288:Background
220:reoxidants
189:(DHQD) vs
174:to form a
390:. Basic
266:catalysts
262:reactants
152:) is the
47:Reaction
979:(1992).
891:(1996).
887:Oi, R.;
691:See also
298:reagents
222:such as
176:vicinal
169:quinine
1199:
1001:: 47.
840:: 93.
757:
582:Fe(CN)
465:of OsO
354:Fe(CN)
331:(NMO,
329:-oxide
243:AD-mix
235:-oxide
172:ligand
166:chiral
158:alkene
156:of an
57:Alkene
913:: 1.
826:trans
515:face.
300:like
239:toxic
160:with
1197:ISBN
755:ISBN
551:(OH)
452:edit
338:tert
304:(OsO
179:diol
96:diol
94:1,2-
1468:doi
1464:117
1436:doi
1432:114
1408:doi
1404:119
1379:doi
1375:118
1347:doi
1317:doi
1313:116
1289:doi
1252:doi
1248:111
1223:doi
1219:102
1170:doi
1166:111
1142:doi
1114:doi
1110:119
1075:doi
1044:doi
1003:doi
958:doi
945:cis
915:doi
870:doi
842:doi
789:doi
747:doi
679:doi
675:110
652:cis
547:OsO
474:OsO
422:).
358:).
335:),
320:),
226:or
126:RSC
66:OsO
1487::
1448:^
1359:^
1343:35
1341:.
1285:26
1182:^
1164:.
1136:.
1099:;
1087:^
1071:37
1040:57
1022:^
1010:;
999:70
997:.
991:.
981:"(
954:17
922:;
911:73
909:.
903:.
866:55
838:79
836:.
830:.
824:)-
820:,2
812:,2
785:94
753:.
743:66
513:Re
410:NH
406:SO
350:(K
312:,
284:.
1474:.
1470::
1442:.
1438::
1414:.
1410::
1385:.
1381::
1353:.
1349::
1323:.
1319::
1295:.
1291::
1258:.
1254::
1229:.
1225::
1176:.
1172::
1148:.
1144::
1138:3
1120:.
1116::
1081:.
1077::
1050:.
1046::
1017:.
1005::
987:R
985:,
983:R
964:.
960::
941:4
929:.
917::
899:H
895:S
876:.
872::
849:.
844::
822:S
818:R
814:R
810:S
795:.
791::
763:.
749::
685:.
681::
584:6
580:3
553:4
549:2
545:2
541:4
537:4
521:4
476:4
467:4
456:]
420:2
412:2
408:2
404:3
400:6
396:5
388:4
384:3
380:2
368:4
356:6
352:3
344:t
342:(
327:N
323:N
316:(
306:4
251:2
247:2
233:N
229:N
212:S
208:M
204:L
202:R
86:↓
75:+
68:4
63:+
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.