2885:) is that lowering the energy of the oxygen atom's nonbonding hybrid orbitals (by assigning them more s character and less p character) and correspondingly raising the energy of the oxygen atom's hybrid orbitals bonded to the hydrogen atoms (by assigning them more p character and less s character) has the net effect of lowering the energy of the occupied molecular orbitals because the energy of the oxygen atom's nonbonding hybrid orbitals contributes completely to the energy of the oxygen atom's lone pairs while the energy of the oxygen atom's other two hybrid orbitals contributes only partially to the energy of the bonding orbitals (the remainder of the contribution coming from the hydrogen atoms' 1s orbitals).
2548:
2359:
1786:
105:
1731:
2795:
2536:
52:
4350:
75:
1669:
1860:. This denser saltwater sinks by convection and the replacing seawater is subject to the same process. This produces essentially freshwater ice at −1.9 °C on the surface. The increased density of the seawater beneath the forming ice causes it to sink towards the bottom. On a large scale, the process of brine rejection and sinking cold salty water results in ocean currents forming to transport such water away from the Poles, leading to a global system of currents called the
1811:
2029:
65:
1958:
2462:
3724:). In the inner Solar System, planets, asteroids, and moons formed almost entirely of metals and silicates. Water has since been delivered to the inner Solar System via an as-yet unknown mechanism, theorized to be the impacts of asteroids or comets carrying water from the outer Solar System, where bodies contain much more water ice. The difference between planetary bodies located inside and outside the frost line can be stark. Earth's mass is 0.000023% water, while
1134:
9113:
2286:). Because of autoionization, at ambient temperatures pure liquid water has a similar intrinsic charge carrier concentration to the semiconductor germanium and an intrinsic charge carrier concentration three orders of magnitude greater than the semiconductor silicon, hence, based on charge carrier concentration, water can not be considered to be a completely dielectric material or electrical insulator but to be a limited conductor of ionic charge.
9102:
9135:
8830:
2582:
1873:
4959:
9124:
2507:
1307:
7491:
1794:
bottom up, and all life in it would be killed. Furthermore, given that water is a good thermal insulator (due to its heat capacity), some frozen lakes might not completely thaw in summer. As it is, the inversion of the density curve leads to a stable layering for surface temperatures below 4 °C, and with the layer of ice that floats on top insulating the water below, even e.g.,
3609:, other than the simple difference in specific mass, involve properties that are affected by hydrogen bonding, such as freezing and boiling, and other kinetic effects. This is because the nucleus of deuterium is twice as heavy as protium, and this causes noticeable differences in bonding energies. The difference in boiling points allows the isotopologues to be separated. The
1137:
1707:(more commonly known as latent heat) of water is 333.55 kJ/kg at 0 °C: the same amount of energy is required to melt ice as to warm ice from −160 °C up to its melting point or to heat the same amount of water by about 80 °C. Of common substances, only that of ammonia is higher. This property confers resistance to melting on the ice of
7509:
5470:
1139:
1939:. The reverse process accounts for the fog burning off in the morning. If the humidity is increased at room temperature, for example, by running a hot shower or a bath, and the temperature stays about the same, the vapor soon reaches the pressure for phase change and then condenses out as minute water droplets, commonly referred to as steam.
1569:. The molecules of water are constantly moving concerning each other, and the hydrogen bonds are continually breaking and reforming at timescales faster than 200 femtoseconds (2 × 10 seconds). However, these bonds are strong enough to create many of the peculiar properties of water, some of which make it integral to life.
1943:
the air is small, relative humidity, the ratio of the partial pressure due to the water vapor to the saturated partial vapor pressure, is much more useful. Vapor pressure above 100% relative humidity is called supersaturated and can occur if the air is rapidly cooled, for example, by rising suddenly in an updraft.
5456:
7522:
Notice that the bond angles decrease as the number of nonbonding electron pairs increases. A bonding pair of electrons is attracted by both nuclei of the bonded atoms, but a nonbonding pair is attracted primarily by only one nucleus. Because a nonbonding pair experiences less nuclear attraction, its
3668:
may affect biochemical processes—ingestion of large amounts impairs kidney and central nervous system function. Small quantities can be consumed without any ill-effects; humans are generally unaware of taste differences, but sometimes report a burning sensation or sweet flavor. Very large amounts of
1942:
A saturated gas or one with 100% relative humidity is when the vapor pressure of water in the air is at equilibrium with vapor pressure due to (liquid) water; water (or ice, if cool enough) will fail to lose mass through evaporation when exposed to saturated air. Because the amount of water vapor in
4723:
The gases produced bubble to the surface, where they can be collected or ignited with a flame above the water if this was the intention. The required potential for the electrolysis of pure water is 1.23 V at 25 °C. The operating potential is actually 1.48 V or higher in practical electrolysis.
2834:
structures form due to the four hydrogen bonds, thereby forming an open structure and a three-dimensional bonding network, resulting in the anomalous decrease in density when cooled below 4 °C. This repeated, constantly reorganizing unit defines a three-dimensional network extending throughout
2502:
observed a strong repulsive force between hydrophilic surfaces. To dehydrate hydrophilic surfaces—to remove the strongly held layers of water of hydration—requires doing substantial work against these forces, called hydration forces. These forces are very large but decrease rapidly over a nanometer
1759:
0–4 °C allows for a denser molecular packing in which some of the lattice cavities are filled by water molecules. Above 4 °C, however, thermal expansion becomes the dominant effect, and water near the boiling point (100 °C) is about 4% less dense than water at 4 °C (39 °F).
6037:
Ocean warming dominates the global energy change inventory. Warming of the ocean accounts for about 93% of the increase in the Earth's energy inventory between 1971 and 2010 (high confidence), with the warming of the upper (0 to 700 m) ocean accounting for about 64% of the total. Melting ice
1758:
These peculiar effects are due to the highly directional bonding of water molecules via the hydrogen bonds: ice and liquid water at low temperature have comparatively low-density, low-energy open lattice structures. The breaking of hydrogen bonds on melting with increasing temperature in the range
2479:), due to the collective action of hydrogen bonds between water molecules. These hydrogen bonds are constantly breaking, with new bonds being formed with different water molecules; but at any given time in a sample of liquid water, a large portion of the molecules are held together by such bonds.
1742:
of water is about 1 gram per cubic centimetre (62 lb/cu ft): this relationship was originally used to define the gram. The density varies with temperature, but not linearly: as the temperature increases, the density rises to a peak at 3.98 °C (39.16 °F) and then decreases; the
1793:
The unusual density curve and lower density of ice than of water is essential for much of the life on earth—if water were most dense at the freezing point, then in winter the cooling at the surface would lead to convective mixing. Once 0 °C are reached, the water body would freeze from the
2217:
point of about 231 K (−42 °C; −44 °F). The melting point of ordinary hexagonal ice falls slightly under moderately high pressures, by 0.0073 °C (0.0131 °F)/atm or about 0.5 °C (0.90 °F)/70 atm as the stabilization energy of hydrogen bonding is exceeded by
8574:
Weingärtner, Hermann; Teermann, Ilka; Borchers, Ulrich; Balsaa, Peter; Lutze, Holger V.; Schmidt, Torsten C.; Franck, Ernst Ulrich; Wiegand, Gabriele; Dahmen, Nicolaus; Schwedt, Georg; Frimmel, Fritz H.; Gordalla, Birgit C. (2016). "Water, 1. Properties, Analysis, and
Hydrological Cycle".
2013:
The bulk modulus of water ice ranges from 11.3 GPa at 0 K up to 8.6 GPa at 273 K. The large change in the compressibility of ice as a function of temperature is the result of its relatively large thermal expansion coefficient compared to other common solids.
7523:
electron domain is spread out more in space than is the electron domain for a bonding pair (Figure 9.7). Nonbonding electron pairs, therefore, take up more space than bonding pairs; in essence, they act as large and fatter balloons in our analogy of Figure 9.5. As a result,
2312:
and deionized ultra-pure water systems used, for instance, in semiconductor manufacturing plants. A salt or acid contaminant level exceeding even 100 parts per trillion (ppt) in otherwise ultra-pure water begins to noticeably lower its resistivity by up to several kΩ·m.
1821:
The density of saltwater depends on the dissolved salt content as well as the temperature. Ice still floats in the oceans, otherwise, they would freeze from the bottom up. However, the salt content of oceans lowers the freezing point by about 1.9 °C (due to
3224:
2650:
are relatively soluble in water, and nonpolar substances such as fats and oils are not. Nonpolar molecules stay together in water because it is energetically more favorable for the water molecules to hydrogen bond to each other than to engage in
2412:
Although hydrogen bonding is a relatively weak attraction compared to the covalent bonds within the water molecule itself, it is responsible for several of the water's physical properties. These properties include its relatively high
2405:, points from a region between the two hydrogen atoms to the oxygen atom. The charge differences cause water molecules to aggregate (the relatively positive areas being attracted to the relatively negative areas). This attraction,
1659:
and the critical pressure is caused by the weight of the ocean at the extreme depths where the vents are located. This pressure is reached at a depth of about 2200 meters: much less than the mean depth of the ocean (3800 meters).
1403:
in salts and bond to other polar substances such as alcohols and acids, thus dissolving them. Its hydrogen bonding causes its many unique properties, such as having a solid form less dense than its liquid form, a relatively high
5779:
2005:
of water is about 2.2 GPa. The low compressibility of non-gasses, and of water in particular, leads to their often being assumed as incompressible. The low compressibility of water means that even in the deep
2728:. Unlike previously reported tunneling motions in water, this involved the concerted breaking of two hydrogen bonds. Later in the same year, the discovery of the quantum tunneling of water molecules was reported.
4393:) and an acidic proton are added to the two carbon atoms bonded together in the carbon-carbon double bond, resulting in an alcohol. When the addition of water to an organic molecule cleaves the molecule in two,
1830:
blocked by an expansion of water as it becomes colder near the freezing point. The oceans' cold water near the freezing point continues to sink. So creatures that live at the bottom of cold oceans like the
2835:
the liquid. This view is based upon neutron scattering studies and computer simulations, and it makes sense in the light of the unambiguously tetrahedral arrangement of water molecules in ice structures.
2842:
suggested that water molecules in the liquid state typically bind not to four but only two others; thus forming chains and rings. The term "string theory of water" (which is not to be confused with the
5635:
Ramires, Maria L. V.; Castro, Carlos A. Nieto de; Nagasaka, Yuchi; Nagashima, Akira; Assael, Marc J.; Wakeham, William A. (1995-05-01). "Standard
Reference Data for the Thermal Conductivity of Water".
1747:
so that the density only decreases as a function of temperature. The increase observed for water from 0 °C (32 °F) to 3.98 °C (39.16 °F) and for a few other liquids is described as
1908:. Water being a relatively polar compound will tend to be miscible with liquids of high polarity such as ethanol and acetone, whereas compounds with low polarity will tend to be immiscible and poorly
1550:
at wavelengths of around 750 nm which cause it to appear to have a blue color. This can easily be observed in a water-filled bath or wash-basin whose lining is white. Large ice crystals, as in
7213:
Richardson, Jeremy O.; Pérez, Cristóbal; Lobsiger, Simon; Reid, Adam A.; Temelso, Berhane; Shields, George C.; Kisiel, Zbigniew; Wales, David J.; Pate, Brooks H.; Althorpe, Stuart C. (2016-03-18).
4828:, or its equivalent in different languages, although there are other systematic names which can be used to describe the molecule. Oxidane is only intended to be used as the name of the mononuclear
2624:
from the water. Contrary to the common misconception, water and hydrophobic substances do not "repel", and the hydration of a hydrophobic surface is energetically, but not entropically, favorable.
1802:
freezes only to about 1 m thickness in winter. In general, for deep enough lakes, the temperature at the bottom stays constant at about 4 °C (39 °F) throughout the year (see diagram).
7905:
2567:
Because water has strong cohesive and adhesive forces, it exhibits capillary action. Strong cohesion from hydrogen bonding and adhesion allows trees to transport water more than 100 m upward.
5096:(VSMOW), used for calibration, melts at 273.1500089(10) K (0.000089(10) °C, and boils at 373.1339 K (99.9839 °C). Other isotopic compositions melt or boil at slightly different temperatures.
3338:
3770:) donor and a base is a proton acceptor. When reacting with a stronger acid, water acts as a base; when reacting with a stronger base, it acts as an acid. For instance, water receives an
4614:
Water can be split into its constituent elements, hydrogen, and oxygen, by passing an electric current through it. This process is called electrolysis. The cathode half reaction is:
7599:
Hardy, Edme H.; Zygar, Astrid; Zeidler, Manfred D.; Holz, Manfred; Sacher, Frank D. (2001). "Isotope effect on the translational and rotational motion in liquid water and ammonia".
1138:
2620:
that water molecules generate between other water molecules. If a substance has properties that do not allow it to overcome these strong intermolecular forces, the molecules are
1850:, 3.5%) the ice that forms is essentially salt-free, with about the same density as freshwater ice. This ice floats on the surface, and the salt that is "frozen out" adds to the
5771:
3639:. Because water molecules exchange hydrogen atoms with one another, hydrogen deuterium oxide (DOH) is much more common in low-purity heavy water than pure dideuterium monoxide
1826:) and lowers the temperature of the density maximum of water to the former freezing point at 0 °C. This is why, in ocean water, the downward convection of colder water is
3563:
exists in nature only in minute quantities, being produced primarily via cosmic ray-induced nuclear reactions in the atmosphere. Water with one protium and one deuterium atom
2490:
the water may form a thin film because the molecular forces between glass and water molecules (adhesive forces) are stronger than the cohesive forces. In biological cells and
2344:
10 S/cm, but this conductivity is now thought to be almost entirely from surface defects, and without those, ice is an insulator with an immeasurably small conductivity.
6315:; Salzmann, Christoph; Kohl, Ingrid; Mayer, Erwin; Hallbrucker, Andreas (2001-01-01). "A second distinct structural "state" of high-density amorphous ice at 77 K and 1 bar".
2332:
into oxygen and hydrogen gases but in the absence of dissolved ions this is a very slow process, as very little current is conducted. In ice, the primary charge carriers are
3107:
5428:
Tanaka, M; Girard, G; Davis, R; Peuto, A; Bignell, N (August 2001). "Recommended table for the density of water between 0 C and 40 C based on recent experimental reports".
2826:, or structural properties like those observed in water because none of them can form four hydrogen bonds: either they cannot donate or accept hydrogen atoms, or there are
8750:
3058:
1923:
that is thermodynamically stable with the liquid (or solid) at a given temperature is relatively low compared with total atmospheric pressure. For example, if the vapor's
4844:
for the –OH group. The name oxane is explicitly mentioned by the IUPAC as being unsuitable for this purpose, since it is already the name of a cyclic ether also known as
5579:
3716:, where the Sun's radiation is too weak to vaporize solid and liquid water (as well as other elements and chemical compounds with relatively low melting points, such as
1648:. In nature, this only rarely occurs in extremely hostile conditions. A likely example of naturally occurring supercritical water is in the hottest parts of deep water
14961:
3355:, physical processes that convert solid rocks and minerals into soil and sediment, but under some conditions chemical reactions with water occur as well, resulting in
1140:
14970:
14254:
14226:
7789:
5671:
17803:
17476:
14938:
14264:
14245:
14217:
2297:. If water has even a tiny amount of such an impurity, then the ions can carry charges back and forth, allowing the water to conduct electricity far more readily.
17795:
17572:
17540:
16777:
16753:
16741:
15156:
14929:
10942:
5399:
2877:
explanation is that the oxygen atom's lone pairs are physically larger and therefore take up more space than the oxygen atom's bonds to the hydrogen atoms. The
2543:
is under the water level, which has risen gently and smoothly. Surface tension prevents the clip from submerging and the water from overflowing the glass edges.
17787:
17731:
17564:
17532:
17484:
17444:
17221:
17200:
16813:
16801:
16789:
16765:
10354:
8419:
6939:
Hoy, AR; Bunker, PR (1979). "A precise solution of the rotation bending Schrödinger equation for a triatomic molecule with application to the water molecule".
17779:
17755:
17723:
17681:
17644:
17516:
17436:
17428:
17208:
17195:
17179:
17139:
17123:
17115:
8202:
2635:. The partially negative dipole ends of the water are attracted to positively charged components of the solute, and vice versa for the positive dipole ends.
17665:
17636:
17620:
17588:
17556:
17508:
17226:
17131:
17107:
15161:
5291:
2616:("water-fearing") substances. The ability of a substance to dissolve in water is determined by whether or not the substance can match or better the strong
5455:
Lemmon, Eric W.; Bell, Ian H.; Huber, Marcia L.; McLinden, Mark O. "Thermophysical
Properties of Fluid Systems". In Linstrom, P.J.; Mallard, W.G. (eds.).
3669:
heavy water must be consumed for any toxicity to become apparent. Rats, however, are able to avoid heavy water by smell, and it is toxic to many animals.
17657:
17612:
17604:
17580:
17548:
17468:
17213:
10227:
1823:
1148:
2559:
of 71.99 mN/m at 25 °C which is caused by the strength of the hydrogen bonding between water molecules. This allows insects to walk on water.
17596:
17460:
17320:
10516:
6022:
3448:
is the current international standard for water isotopes. Naturally occurring water is almost completely composed of the neutron-less hydrogen isotope
2847:
of physics) was coined. These observations were based upon X-ray absorption spectroscopy that probed the local environment of individual oxygen atoms.
1320:
17673:
17284:
17187:
14512:
10763:
8576:
8377:
7893:
4876:
6038:(including Arctic sea ice, ice sheets, and glaciers) and warming of the continents and atmosphere account for the remainder of the change in energy.
2078:
in Göttingen produced data on several other triple points in the early 20th century. Kamb and others documented further triple points in the 1960s.
17771:
16438:
16344:
16320:
16308:
8023:
6874:
Light, Truman S.; Licht, Stuart; Bevilacqua, Anthony C.; Morash, Kenneth R. (2005-01-01). "The
Fundamental Conductivity and Resistivity of Water".
5312:
Sometimes these compounds have generic or common names (e.g., H2O is "water") and they also have systematic names (e.g., H2O, dihydrogen monoxide).
2447:, despite hydrogen sulfide having nearly twice the molar mass of water. The extra bonding between water molecules also gives liquid water a large
16380:
16368:
16356:
16332:
8052:
2374:
of electrons. One effect usually ascribed to the lone pairs is that the H–O–H gas-phase bend angle is 104.48°, which is smaller than the typical
2213:
well below that temperature without freezing if the liquid is not mechanically disturbed. It can remain in a fluid state down to its homogeneous
1162:
7821:
2698:. The water dipoles make hydrogen bonds with the polar regions of the sugar molecule (OH groups) and allow it to be carried away into solution.
16426:
15942:
15930:
14085:
9026:
6558:
8159:
1927:
is 2% of atmospheric pressure and the air is cooled from 25 °C, starting at about 22 °C, water will start to condense, defining the
15765:
8400:
2074:(forms) of ice, water has other triple points, which have either three polymorphs of ice or two polymorphs of ice and liquid in equilibrium.
6612:
5514:
2503:
or less. They are important in biology, particularly when cells are dehydrated by exposure to dry atmospheres or to extracellular freezing.
2417:
and boiling point temperatures: more energy is required to break the hydrogen bonds between water molecules. In contrast, hydrogen sulfide (
1722:
The specific heat capacity of ice at −10 °C is 2030 J/(kg·K) and the heat capacity of steam at 100 °C is 2080 J/(kg·K).
16392:
16268:
15979:
15918:
15801:
15789:
15777:
15510:
15475:
9184:
7525:
electron domains for nonbonding electron pairs exert greater repulsive forces on adjacent electron domains and tend to compress bond angles
479:
1990:
around 45 °C before increasing again with increasing temperature. As the pressure is increased, the compressibility decreases, being
16153:
16129:
16117:
15065:
6224:
104:
6124:
5600:
Silverstein, Todd P.; Heller, Stephen T. (17 April 2017). "pKa Values in the
Undergraduate Curriculum: What Is the Real pKa of Water?".
3978:, although it can also react with Lewis bases, forming hydrogen bonds between the electron pair donors and the hydrogen atoms of water.
2869:, molecular structure, allowing it to be polar. The hydrogen–oxygen–hydrogen angle is 104.45°, which is less than the 109.47° for ideal
2716:
dynamics in water was reported as early as 1992. At that time it was known that there are motions which destroy and regenerate the weak
16987:
16189:
16177:
16165:
16141:
15890:
10093:
7192:
6914:
6861:
C. S. Fuller "Defect
Interactions in Semiconductors" Chapter 5 pp. 192-221 in "Semiconductors" N. B. Hannay Ed. Reinhold, New York 1959
3416:
Rain is generally mildly acidic, with a pH between 5.2 and 5.8 if not having any acid stronger than carbon dioxide. If high amounts of
2778:
take advantage of water's opacity to microwave radiation to heat the water inside of foods. Water's light blue color is caused by weak
6638:
15856:
4817:
1680:
of 4184 J/(kg·K) at 20 °C (4182 J/(kg·K) at 25 °C) —the second-highest among all the heteroatomic species (after
2382:
to the hydrogens, so they require more space. The increased repulsion of the lone pairs forces the O–H bonds closer to each other.
6533:
6074:
8731:
6910:
6395:
8739:
8677:
Release on the IAPWS Formulation 1995 for the
Thermodynamic Properties of Ordinary Water Substance for General and Scientific Use
6467:
7466:
O, is similar. It has two electron pairs with nothing attached to them. They, too, must be taken into account. Molecules like NH
51:
7067:
6094:, le poids absolu d'un volume d'eau pure égal au cube de la centième partie du mètre, et à la température de la glace fondante.
5332:
2779:
2075:
1315:
5571:
4533:
to emit oxygen gas, but very few oxidants react with water even if their reduction potential is greater than the potential of
3982:
describes water as both a weak hard acid and a weak hard base, meaning that it reacts preferentially with other hard species:
8617:
8564:
8432:
8394:
8367:
8341:
8309:
8288:
8265:
7503:
5862:
4735:
showed that water was composed of oxygen and hydrogen in 1781. The first decomposition of water into hydrogen and oxygen, by
1036:
8487:
6562:
1904:
are immiscible, usually forming layers according to increasing density from the top. This can be predicted by comparing the
14011:
6838:
5743:
4868:
6055:
5901:
Smith, Jared D.; Christopher D. Cappa; Kevin R. Wilson; Ronald C. Cohen; Phillip L. Geissler; Richard J. Saykally (2005).
5868:
2401:
points from each H to the O, making the oxygen partially negative and each hydrogen partially positive. A large molecular
8727:
7774:
6791:
5018:
4988:
3689:
Water is the most abundant substance on Earth's surface and also the third most abundant molecule in the universe, after
2737:
1063:
2631:). The relatively small size of water molecules (~ 3 angstroms) allows many water molecules to surround one molecule of
1974:
of water is a function of pressure and temperature. At 0 °C, at the limit of zero pressure, the compressibility is
8676:
8594:
8534:
8511:
8481:
8315:
8244:
5680:, 8—Concentrative Properties of Aqueous Solutions: Density, Refractive Index, Freezing Point Depression, and Viscosity.
3382:, with a variety of small molecules that can be embedded in its spacious crystal lattice. The most notable of these is
2209:
The melting point of ice is 0 °C (32 °F; 273 K) at standard pressure; however, pure liquid water can be
8502:
Reece, Jane B.; Urry, Lisa A.; Cain, Michael L.; Wasserman, Steven A.; Minorsky, Peter V.; Jackson, Robert B. (2013).
7575:
5360:
5168:. Also, fairly pure silicon has a negative coefficient of thermal expansion for temperatures between about 18 and 120
3240:
8735:
8709:
7455:
6218:
1558:
454:
8446:
6263:
74:
64:
11213:
9177:
8192:
6296:
5093:
4902:, using acid and base names. None of these exotic names are used widely. The polarized form of the water molecule,
3677:
refers to deuterium-depleted water (DDW), water in which the deuterium content has been reduced below the standard
3445:
2810:
because it can accept two bonds using the lone pairs on oxygen and donate two hydrogen atoms. Other molecules like
2523:
2071:
1763:
566:
6647:
1327:
17911:
15058:
8781:
8661:
5575:
1887:
1779:
8683:
Online calculator using the IAPWS Supplementary
Release on Properties of Liquid Water at 0.1 MPa, September 2008
5299:
1688:(40.65 kJ/mol or 2257 kJ/kg at the normal boiling point), both of which are a result of the extensive
14979:
11167:
10086:
7309:
6738:
5708:
2375:
1090:
17:
4788:
scales were, or currently are, defined by the freezing and boiling points of water. The less common scales of
17851:
9001:
8155:
7088:
5540:
4530:
2617:
129:
6015:
17916:
17866:
15074:
10054:
5960:
Deguchi, Shigeru; Tsujii, Kaoru (2007-06-19). "Supercritical water: a fascinating medium for soft matter".
2856:
2724:. On 18 March 2016, it was reported that the hydrogen bond can be broken by quantum tunneling in the water
2300:
It is known that the theoretical maximum electrical resistivity for water is approximately 18.2 MΩ·cm (182
431:
5528:
4365:
crystallizes with one molecule of "lattice" water, which interacts with the sulfate and with the centers.
289:
17886:
17881:
17861:
14772:
9170:
8936:
4740:
8016:
7330:
Pope; Fry (1996). "Absorption spectrum (380-700nm) of pure water. II. Integrating cavity measurements".
6416:
Fine, R.A.; Millero, F.J. (1973). "Compressibility of water as a function of temperature and pressure".
2838:
However, there is an alternative theory for the structure of water. In 2004, a controversial paper from
15051:
11423:
4978:
4884:
3219:{\displaystyle K_{\rm {eq}}={\frac {a_{\rm {H_{3}O^{+}}}\cdot a_{\rm {OH^{-}}}}{a_{\rm {H_{2}O}}^{2}}}}
2707:
1775:
1748:
824:
806:
6828:"Revised Release on the Pressure along the Melting and Sublimation Curves of Ordinary Water Substance"
17926:
17871:
17083:
14851:
14831:
14792:
10079:
8973:
8745:
8044:
7813:
4973:
4833:
3713:
3091:
2866:
2621:
340:
8147:
17896:
14996:
14891:
14782:
14442:
14004:
8958:
8887:
6463:
5485:
4315:
3672:
2972:
2905:
2899:
2878:
2252:
1952:
1861:
1698:
Most of the additional energy stored in the climate system since 1970 has accumulated in the oceans
1496:
1458:
8381:
6043:
2978:
2547:
17856:
17699:
14906:
14821:
14802:
14615:
14560:
14426:
14366:
14283:
14273:
14207:
12653:
11389:
9878:
7447:
6673:
6599:
5510:
5245:). Using the standard definition of atmosphere, 1,013,250 dynes/cm, it works out to 0.0073°C/atm.
5058:
4748:
4516:
2870:
2690:, each being surrounded by water molecules. The ions are then easily transported away from their
2608:
due to its high dielectric constant. Substances that mix well and dissolve in water are known as
2367:
2317:
2060:
2023:
1755:
is also less dense than liquid water—upon freezing, the density of water decreases by about 9%.
17921:
17500:
14881:
14861:
14841:
14812:
14455:
14356:
14235:
13224:
13210:
12398:
12296:
11139:
11125:
9825:
9127:
9069:
9011:
8556:
4983:
4769:
4752:
3098:
2771:
2448:
2402:
2358:
1677:
1012:
9036:
6206:
4527:
is a reaction between iron and oxygen that is dissolved in water, not between iron and water.
3580:
in far lower amounts (0.000003%) and any such molecules are temporary as the atoms recombine.
3424:
oxides are present in the air, they too will dissolve into the cloud and raindrops, producing
2226:, i.e., reaching 355 K (82 °C) at 2.216 GPa (21,870 atm) (triple point of
2010:
at 4 km depth, where pressures are 40 MPa, there is only a 1.8% decrease in volume.
17906:
17628:
17492:
17452:
17163:
16610:
16593:
15146:
15141:
14916:
14871:
14753:
14611:
14601:
14483:
13825:
13561:
12670:
12449:
12381:
12005:
11457:
11153:
11069:
10553:
9957:
9562:
8978:
8359:
8255:
7692:
7665:
6181:
4609:
2767:
2248:
1785:
1656:
9051:
7168:
6651:
2451:. This high heat capacity makes water a good heat storage medium (coolant) and heat shield.
1919:
As a gas, water vapor is completely miscible with air. On the other hand, the maximum water
1487:
is a constant, so their respective concentrations are inversely proportional to each other.
17763:
17707:
17239:
17155:
16892:
15655:
14762:
14336:
14322:
14312:
13774:
13544:
13510:
13308:
13025:
12840:
12602:
12585:
12228:
12177:
11338:
11321:
11287:
11206:
11111:
11055:
11000:
10988:
10902:
10807:
10775:
10472:
10443:
10419:
10145:
9654:
9074:
8907:
8082:
7639:
7608:
7598:
7394:
7339:
7283:
7226:
7023:
6948:
6775:
6712:
6608:
6509:
6425:
6324:
6149:
5969:
5914:
5903:"Unified description of temperature-dependent hydrogen bond rearrangements in liquid water"
5644:
5609:
5375:
4758:
4450:
4426:
4422:
2968:
2839:
2515:
2476:
2055:
of water. Since 1954, this point had been used to define the base unit of temperature, the
1685:
1633:
1625:). Visible steam and clouds are formed from minute droplets of water suspended in the air.
880:
440:
117:
31:
9134:
6701:"Review of the vapour pressures of ice and supercooled water for atmospheric applications"
6640:
Impact of High
Pressure — Low Temperature Processes on Cellular Materials Related to Foods
213:
8:
17876:
17824:
17715:
17400:
17234:
16856:
15876:
15650:
15151:
14743:
14591:
14404:
14332:
14302:
14190:
14140:
14130:
14055:
13997:
13881:
13791:
13637:
13252:
13182:
13126:
12927:
12899:
12823:
12806:
12789:
12755:
12704:
12636:
12568:
12432:
12364:
12330:
12279:
12143:
11985:
11975:
11935:
11738:
11571:
11491:
11474:
11440:
11304:
11097:
11080:
10934:
10914:
10889:
10860:
10577:
10536:
10492:
10431:
10407:
10386:
10279:
10267:
10113:
9732:
9696:
9499:
9242:
9232:
9079:
8774:
7718:
Miller, Inglis J. Jr.; Mooser, Gregory (Jul 1979). "Taste
Responses to Deuterium Oxide".
7440:
3375:
2874:
2652:
2643:
2519:
1629:
1543:
776:
309:
203:
155:
8086:
7643:
7612:
7398:
7343:
7287:
7230:
7027:
6952:
6779:
6716:
6513:
6489:
6429:
6328:
6153:
6078:
5973:
5918:
5648:
5613:
5379:
4204:
When a salt of a weak acid or of a weak base is dissolved in water, water can partially
1384:
on Earth's surface. It is also the third most abundant molecule in the universe (behind
269:
17901:
17388:
17383:
17292:
17276:
16883:
16717:
16687:
16623:
14665:
14634:
14625:
14399:
14293:
14170:
14094:
14075:
13808:
13707:
13578:
13459:
13420:
13392:
12997:
12857:
12772:
12738:
12687:
12500:
12466:
12245:
12102:
12092:
11885:
11875:
11858:
11828:
11758:
11519:
11406:
11355:
11236:
10926:
10877:
10815:
10722:
10639:
10627:
10606:
10504:
10255:
10215:
9990:
9845:
9758:
9714:
9504:
9193:
8829:
8715:
8549:
8523:
8277:
7743:
7672:. Vol. 126, no. 4. New York: Popular Science Publishing. Apr 1935. p. 17
7420:
7363:
7044:
7011:
6918:
6730:
6525:
6173:
6139:
5937:
5902:
5900:
5008:
4922:
4379:
4362:
4282:
3759:
3379:
2398:
2064:
1704:
1697:
1649:
1502:
1432:
1223:
1113:
959:
944:
762:
249:
6455:
6387:
2612:("water-loving") substances, while those that do not mix well with water are known as
1581:
is the most common and is the form that is generally denoted by the word "water". The
17747:
17739:
17305:
17268:
17171:
17062:
17047:
17042:
16847:
16639:
16066:
15467:
15397:
15257:
15205:
15189:
15181:
15102:
14644:
14542:
14502:
14493:
14473:
14416:
14195:
14180:
14150:
14115:
14050:
13909:
13853:
13721:
13679:
13651:
13623:
13595:
13527:
13493:
13476:
13434:
13336:
13322:
13238:
13112:
13084:
13042:
12885:
12619:
12517:
12415:
12347:
12313:
12194:
12112:
11995:
11949:
11945:
11818:
11688:
11627:
11558:
11372:
11087:
11012:
10976:
10894:
10799:
10787:
10708:
10700:
10688:
10644:
10565:
10528:
10484:
10460:
10395:
10366:
10235:
10169:
10137:
9910:
9892:
9772:
9598:
9416:
9411:
9397:
9309:
9304:
9237:
9101:
9086:
8869:
8809:
8719:
8705:
8613:
8590:
8560:
8530:
8507:
8477:
8438:
8428:
8390:
8363:
8337:
8305:
8284:
8261:
8240:
7735:
7731:
7563:"Guideline on the Use of Fundamental Physical Constants and Basic Constants of Water"
7499:
7451:
7412:
7355:
7301:
7252:
7244:
7184:
7082:
7049:
6960:
6891:
6734:
6529:
6340:
6292:
6214:
6165:
5993:
5985:
5942:
5858:
5660:
5391:
5238:
5220:
5043:
5028:
4856:
4208:
the salt, producing the corresponding base or acid, which gives aqueous solutions of
3782:
3705:. 0.23 ppm of the earth's mass is water and 97.39% of the global water volume of 1.38
3548:
3453:
3383:
3360:
2861:
The repulsive effects of the two lone pairs on the oxygen atom cause water to have a
2811:
2755:
2713:
2589:
2394:
2353:
1905:
1814:
1744:
1653:
1641:
1598:
1462:
1368:. It is by far the most studied chemical compound and is described as the "universal
1353:
1289:
1269:
1233:
1228:
1218:
1190:
1184:
930:
754:
17336:
7747:
7367:
3754:
2535:
1692:
between its molecules. These two unusual properties allow water to moderate Earth's
237:
17524:
17260:
17247:
17147:
17091:
17052:
16978:
16965:
16919:
16708:
15586:
15230:
15107:
15006:
14947:
14670:
14547:
14468:
14389:
14160:
14125:
13867:
13735:
13665:
13378:
13266:
13196:
13168:
13140:
13011:
12983:
12969:
12211:
12072:
12055:
12025:
11955:
11848:
11838:
11778:
11623:
11584:
11545:
11041:
10959:
10848:
10751:
10739:
10713:
10618:
10548:
10455:
10378:
10346:
10333:
10157:
10102:
10043:
9747:
9668:
9544:
9461:
9344:
9269:
9264:
9253:
9006:
8804:
8701:
8697:
8643:
8609:
8582:
8351:
8232:
8090:
7727:
7647:
7634:
Urey, Harold C.; et al. (15 Mar 1935). "Concerning the Taste of Heavy Water".
7616:
7424:
7402:
7347:
7296:
7291:
7234:
7176:
7039:
7031:
6956:
6883:
6783:
6720:
6517:
6433:
6332:
6177:
6157:
5977:
5932:
5922:
5850:
5652:
5617:
5462:
5441:
5437:
5383:
5324:
4860:
4744:
4318:, water can be either a ligand or simply lodged in the framework, or both. Thus,
4278:
3750:
2862:
2794:
2783:
2747:
2647:
2627:
When an ionic or polar compound enters water, it is surrounded by water molecules (
2576:
2444:
2337:
2294:
1924:
1566:
1506:
1428:
1357:
1213:
965:
891:
502:
17409:
17356:
16943:
8657:
8586:
7651:
7215:"Concerted hydrogen-bond breaking by quantum tunneling in the water hexamer prism"
4797:
2774:
utilize the portion of the light spectrum that is transmitted well through water.
2378:
angle of 109.47°. The lone pairs are closer to the oxygen atom than the electrons
17891:
17375:
17255:
17099:
17057:
17033:
17019:
17005:
15671:
15322:
15197:
15112:
15092:
14532:
13980:
13976:
13895:
13693:
13406:
13350:
13280:
13070:
12955:
12913:
12551:
12534:
12483:
12160:
12045:
12035:
11925:
11905:
11808:
11768:
11649:
11597:
11532:
11270:
11199:
11180:
11046:
11029:
10872:
10827:
10734:
10670:
10594:
10316:
10303:
10247:
10210:
10181:
10125:
9939:
9840:
9682:
9426:
9421:
9334:
8968:
8963:
8902:
8892:
8841:
8689:
8544:
8467:
8331:
7699:. Vol. 130, no. 6. New York: Popular Science Publishing. pp. 22–23
7095:
6554:
6312:
5747:
4864:
4845:
4836:. These derivatives commonly have other recommended names. For example, the name
4732:
4515:, are oxidized by water as well, but their oxides adhere to the metal and form a
4438:
4406:
4320:
3712:
Water is far more prevalent in the outer Solar System, beyond a point called the
3544:
3368:
2775:
2691:
2659:
2556:
2511:
2499:
2309:
2305:
1971:
1856:
1843:
1547:
1389:
1372:" and the "solvent of life". It is the most abundant substance on the surface of
973:
780:
758:
742:
359:
17068:
15122:
15082:
8236:
6827:
6123:
Shell, Scott M.; Debenedetti, Pablo G.; Panagiotopoulos, Athanassios Z. (2002).
5740:
5621:
2223:
1730:
404:
17350:
17300:
16571:
15663:
15370:
15346:
15334:
15310:
15213:
15097:
15043:
14346:
13749:
13294:
13154:
13056:
12941:
12721:
12262:
11915:
11895:
11662:
11253:
10589:
10308:
10193:
10001:
9921:
9850:
9514:
9294:
9116:
8767:
8635:
6161:
5842:
4993:
4829:
4398:
4307:
3610:
3233:
O or OH is approximated by its concentration, and the activity of the solvent H
2882:
2819:
2759:
2593:
2390:
2321:
2051:
at which ordinary solid, liquid, and gaseous water coexist in equilibrium is a
1920:
1396:
1365:
1298:
863:
842:
796:
539:
16549:
16023:
6760:
4801:
2340:). Ice was previously thought to have a small but measurable conductivity of 1
2222:) above 209.9 MPa (2,072 atm), the melting point increases markedly
1431:, depending on the pH of the solution that it is in; it readily produces both
1399:
with each other and are strongly polar. This polarity allows it to dissociate
17845:
14522:
14376:
13364:
12082:
11798:
11788:
11636:
11610:
10967:
10839:
10341:
10328:
10015:
9864:
9783:
9573:
9485:
9471:
9451:
9383:
9349:
9314:
9204:
9041:
9021:
7248:
7010:
Chiavazzo, Eliodoro; Fasano, Matteo; Asinari, Pietro; Decuzzi, Paolo (2014).
6895:
6344:
6064:, 6. Properties of Water and Steam as a Function of Temperature and Pressure.
5989:
5664:
5395:
5225:
5186:
5165:
5038:
4789:
4781:
4410:
3725:
3364:
3352:
2844:
2807:
2799:
2743:
2717:
2430:), has much weaker hydrogen bonding due to sulfur's lower electronegativity.
2414:
2406:
2251:, but not even "deionized" water is completely free of ions. Water undergoes
1716:
1689:
1668:
1645:
1614:
1535:
1413:
1405:
993:
784:
772:
662:
652:
538:
Almost colorless or white crystalline solid, almost colorless liquid, with a
15531:
9149:
8442:
8299:
7239:
7214:
5927:
5854:
2510:
Rain water flux from a canopy. Among the forces that govern drop formation:
2409:, explains many of the properties of water, such as its solvent properties.
17816:
17367:
16970:
16675:
16663:
16406:
16296:
16215:
13565:
13228:
13214:
13098:
12657:
12015:
12009:
11965:
11748:
11701:
11675:
11427:
11020:
10954:
10205:
10029:
9975:
9811:
9797:
9640:
9626:
9524:
9354:
9324:
9274:
9218:
9105:
8985:
8951:
8682:
8473:
7562:
7416:
7359:
7305:
7256:
7053:
6169:
6011:
5997:
5946:
5077:
A commonly quoted value of 15.7 used mainly in organic chemistry for the pK
5033:
4964:
4805:
4793:
4736:
4446:
4414:
3490:
or T), which has two neutrons. Oxygen also has three stable isotopes, with
3441:
3356:
2597:
2370:
for the two hydrogens from the oxygen vertex. The oxygen atom also has two
2329:
2052:
2002:
1913:
1835:
generally live in water 4 °C colder than at the bottom of frozen-over
1832:
1810:
1578:
916:
766:
750:
6255:
2304:·m) at 25 °C. This figure agrees well with what is typically seen on
1957:
1762:
Under increasing pressure, ice undergoes a number of transitions to other
1161:
17010:
16825:
16700:
16414:
16284:
15741:
15729:
15628:
15623:
15618:
15595:
15298:
15286:
15274:
13964:
12931:
12402:
12300:
10679:
10661:
9534:
9431:
9284:
9046:
9016:
8941:
8931:
8897:
8859:
8814:
7739:
7638:. Vol. 81, no. 2098. New York: The Science Press. p. 273.
7351:
6144:
5264:
5053:
5048:
5003:
4762:
4755:
showed that water is composed of two parts hydrogen and one part oxygen.
4375:
4343:
4213:
3979:
3552:
3540:
3472:
3347:
The action of water on rock over long periods of time typically leads to
2831:
2613:
2609:
2495:
2461:
2218:
intermolecular repulsion, but as ice transforms into its polymorphs (see
2210:
2044:
2028:
1836:
1795:
1618:
1381:
1174:
738:
726:
696:
7272:"Quantum Tunneling of Water in Beryl: A New State of the Water Molecule"
3471:
or D), a hydrogen isotope with one neutron, and fewer than 20 parts per
1147:
329:
16729:
16651:
16525:
15753:
15358:
14105:
14065:
13959:
13829:
13641:
13548:
13312:
12674:
12640:
12453:
12385:
12122:
11989:
9612:
9441:
7035:
5242:
4998:
4935:
O) when one does not wish to specify whether one is speaking of liquid
4785:
4441:+1 and oxygen in the oxidation state −2. It oxidizes chemicals such as
4394:
4205:
3975:
3971:
3742:
3449:
3348:
2823:
2540:
2469:
2379:
2371:
2366:
An important feature of water is its polar nature. The structure has a
2325:
2255:
in the liquid state when two water molecules form one hydroxide anion (
2214:
1982:. At the zero-pressure limit, the compressibility reaches a minimum of
1565:, which are generally gaseous. This unique property of water is due to
1420:
1409:
1154:
678:
672:
525:
341:
300:
9138:
8173:
8106:
8104:
8094:
8073:
Lewis, G. N.; MacDonald, R. T. (1933). "Concentration of H2 Isotope".
7620:
7438:
Gonick, Larry; Criddle, Craig (2005-05-03). "Chapter 3 Togetherness".
7188:
7012:"Scaling behaviour for the water transport in nanoconfined geometries"
6887:
6787:
6521:
6437:
5387:
4768:
The properties of water have historically been used to define various
1561:, water is primarily a liquid, unlike other analogous hydrides of the
16584:
16276:
16247:
16093:
15963:
15906:
15717:
15633:
15440:
14020:
13934:
13885:
13795:
13711:
13438:
13326:
13256:
13186:
13130:
13116:
13046:
12903:
12889:
12844:
12606:
12589:
12555:
12232:
12181:
12164:
12039:
11979:
11939:
11929:
11889:
11772:
11762:
11742:
11653:
11523:
11393:
9162:
8946:
8912:
8421:
Principles of chemical nomenclature: a guide to IUPAC recommendations
6725:
6336:
5981:
5656:
5194:
4512:
4508:
4402:
3967:
3567:
occur naturally in ordinary water in low concentrations (~0.03%) and
3556:
3457:
3425:
3065:
2913:
2909:
2763:
2628:
2491:
2386:
1928:
1712:
1663:
1594:
1562:
1445:
1108:
910:
868:
847:
746:
682:
7999:
7997:
7407:
7382:
7271:
7180:
6700:
5882:
5718:, National Institute of Standards and Technology, Gaithersburg (MD)
5466:
4349:
2581:
1872:
1297:
Except where otherwise noted, data are given for materials in their
17000:
16579:
16475:
16235:
16105:
16006:
15971:
15864:
15835:
15705:
15502:
15262:
13954:
13944:
13939:
13778:
13725:
13683:
13655:
13627:
13599:
13582:
13514:
13463:
13424:
13396:
13382:
13270:
13200:
13172:
13144:
13029:
13001:
12987:
12827:
12810:
12793:
12759:
12725:
12708:
12572:
12436:
12368:
12334:
12198:
12147:
12106:
12096:
11999:
11969:
11879:
11862:
11832:
11822:
11718:
11692:
11666:
11640:
11575:
11342:
11308:
11291:
11240:
10071:
9519:
8128:
8101:
7919:
7901:
6106:
6104:
6102:
5237:
The source gives it as 0.0072°C/atm. However the author defines an
4837:
4556:
4442:
4342:
consists of centers and one "lattice water". Water is typically a
3417:
3237:
O is approximated by 1, so that we obtain the simple ionic product
2815:
2721:
2483:
2048:
2039:
and water vapor in the lower left portion of a water phase diagram.
1893:
1881:
1851:
1847:
1846:
of saltwater begins to freeze (at −1.9 °C for normal salinity
1590:
1532:
1526:
1522:
1385:
1264:
1120:
1104:
734:
722:
714:
700:
686:
379:
320:
6582:
6580:
6209:. In O'Mara, William C.; Herring, Robert B.; Hunt, Lee P. (eds.).
5813:
5811:
5809:
5458:
NIST Chemistry WebBook, NIST Standard Reference Database Number 69
5197:(melting point of 1,211 K (938 °C; 1,720 °F)), and
2818:
can also form hydrogen bonds. However, they do not show anomalous
2494:, water is in contact with membrane and protein surfaces that are
1719:, ice was and still is in common use for retarding food spoilage.
1672:
Heat of vaporization of water from melting to critical temperature
236:
16839:
16502:
16223:
16085:
15898:
15827:
15813:
15689:
15432:
15405:
15241:
13949:
13913:
13857:
13812:
13739:
13669:
13531:
13497:
13480:
13410:
13354:
13340:
13284:
13242:
13088:
13074:
12917:
12861:
12776:
12742:
12691:
12504:
12470:
12283:
12249:
12215:
12116:
12076:
12029:
11802:
11792:
11782:
11614:
11588:
11549:
11495:
11478:
11461:
8197:
7994:
7984:
7982:
7124:
5549:, Chapter 8: Dissociation Constants of Inorganic Acids and Bases.
5198:
5190:
5182:
4958:
4777:
4520:
4311:
3862:
3721:
3717:
3476:
3437:
3229:
However, for dilute solutions, the activity of a solute such as H
2751:
2725:
2695:
2605:
2586:
2333:
2227:
2121:
1909:
1897:
1854:
and density of the seawater just below it, in a process known as
1799:
1771:
1739:
1708:
1693:
1681:
1551:
1369:
1274:
1259:
1254:
1245:
1155:
788:
730:
718:
704:
557:
391:
8525:
Organic Solvents Physical Properties and Methods of Purification
7852:
7850:
7754:
7136:
6979:
6351:
6099:
5752:
5721:
5552:
3440:
of both hydrogen and oxygen exist, giving rise to several known
2289:
Because water is such a good solvent, it almost always has some
2219:
1610:
16467:
16459:
15697:
15613:
15608:
15603:
15249:
13871:
13697:
13298:
13158:
13060:
13015:
12973:
12959:
12945:
12623:
12521:
12419:
12351:
12317:
12059:
12049:
12019:
11959:
11909:
11852:
11842:
11812:
11752:
11705:
11679:
11601:
11562:
11536:
11444:
11410:
11325:
11274:
8383:
Nomenclature of Inorganic Chemistry: IUPAC Recommendations 2005
7169:"Vibration-Rotation-Tunneling Dynamics in Small Water Clusters"
6577:
6122:
5806:
5169:
4883:, which is a rarely used name of water, and mostly used in the
4773:
4453:
metals. One example of an alkali metal reacting with water is:
4274:
3421:
2827:
2673:
2632:
2301:
2290:
2167:
2149:
2099:
2056:
2033:
1767:
1752:
1637:
1606:
1539:
1361:
708:
280:
8298:
Campbell, Neil A.; Williamson, Brad; Heyden, Robin J. (2006).
8227:
Boyd, Claude E. (2000). "pH, Carbon Dioxide, and Alkalinity".
7979:
7955:
7943:
7931:
5634:
16201:
16077:
13989:
13899:
13753:
12266:
11257:
11222:
8923:
8864:
8849:
8790:
7874:
7862:
7847:
7835:
7570:
7148:
7114:
7112:
7110:
7009:
6967:
6834:
6363:
5778:(website). United States of America: USGS. October 22, 2019.
5711:
in Linstrom, Peter J.; Mallard, William G. (eds.);
5023:
4940:
4936:
2806:
A single water molecule can participate in a maximum of four
2687:
2506:
2487:
2135:
2007:
1622:
1582:
1373:
980:
690:
415:
260:
226:
38:
4346:
ligand, i.e., it forms only one bond with the central atom.
2498:; that is, surfaces that have a strong attraction to water.
2316:
In pure water, sensitive equipment can detect a very slight
13102:
12538:
12487:
12126:
11919:
11899:
11376:
11359:
8754:
7212:
6991:
6213:. Park Ridge, New Jersey: Noyes Publications. p. 431.
6125:"Molecular structural order and anomalies in liquid silica"
4524:
4209:
3746:
2639:
2551:
Temperature dependence of the surface tension of pure water
1602:
1529:
1424:
1400:
547:
370:
7107:
6873:
6490:"Elastic Constants, Bulk Modulus, and Compressibility of H
5823:
5794:
5713:
5537:, Vapor Pressure of Water From 0 to 370 °C in Sec. 6.
3408:, naturally found in large quantities on the ocean floor.
1824:
freezing-point depression of a solvent containing a solute
1652:, in which water is heated to the critical temperature by
13368:
12086:
11191:
8854:
8690:"Structure and Properties of Water in its Various States"
6382:
6380:
6378:
6311:
6291:(2nd ed.). Pearson Prentice-Hall. pp. 162–163.
5013:
4944:
4397:
is said to occur. Notable examples of hydrolysis are the
3363:, a type of chemical alteration of a rock which produces
2486:
properties because of its polar nature. On clean, smooth
2465:
2244:
1936:
1932:
1901:
1743:
initial increase is unusual because most liquids undergo
1586:
1377:
1204:
936:
8759:
8297:
5888:
5201:(melting point of 545 K (272 °C; 521 °F))
3097:
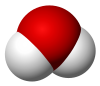
The thermodynamic equilibrium constant is a quotient of
1457:
ions. Related to its amphoteric character, it undergoes
7967:
5494:, Properties of Ice and Supercooled Water in Section 6.
5263:
Both acid and base names exist for water because it is
5193:(melting point of 303 K (30 °C; 86 °F),
4808:
of water is a more commonly used standard point today.
3061:
1789:
Temperature distribution in a lake in summer and winter
7666:"Experimenter Drinks 'Heavy Water' at $ 5,000 a Quart"
6375:
5427:
4310:, which contains two water molecules coordinated to a
3728:, a moon of Saturn, is almost entirely made of water.
1589:
and commonly takes the structure of hard, amalgamated
801:
3.1690 kilopascals or 0.031276 atm at 25 °C
16022:
8418:
Leigh, G. J.; Favre, H. A; Metanomski, W. V. (1998).
8231:. Boston, Massachusetts: Springer. pp. 105–122.
8045:"Joseph Louis Gay-Lussac, French chemist (1778–1850)"
6705:
Quarterly Journal of the Royal Meteorological Society
6680:
6052:, Chapter 6: Properties of Ice and Supercooled Water.
5683:
3243:
3110:
3090:), with a value close to 10 mol L at 25 °C. See
2981:
2032:
The solid/liquid/vapor triple point of liquid water,
1734:
Density of ice and water as a function of temperature
15530:
8417:
8179:
8134:
8116:
8110:
5359:
Braun, Charles L.; Smirnov, Sergei N. (1993-08-01).
4954:
3060:, has a value of about 10 at 25 °C. At neutral
2830:
effects in bulky residues. In water, intermolecular
7543:
7531:
6808:
6394:. National Oceanic and Atmospheric Administration.
5454:
4875:, but this is not among the names published by the
1998:at 0 °C and 100 megapascals (1,000 bar).
8573:
8548:
8522:
8276:
7760:
7439:
6758:
6586:
6287:Housecroft, Catherine E.; Sharpe, Alan G. (2005).
5817:
5558:
5461:. National Institute of Standards and Technology.
4374:As a hard base, water reacts readily with organic
3332:
3218:
3052:
2694:into solution. An example of a nonionic solute is
1664:Heat capacity and heats of vaporization and fusion
8551:Living Ice: Understanding Glaciers and Glaciation
8378:International Union of Pure and Applied Chemistry
8350:
8201:. National Center for Biotechnology Information.
8003:
7489:
7175:. Lawrence Berkeley Lab., CA (United States): 6.
7142:
6357:
6204:
6110:
5758:
5599:
5189:of 1,687 K (1,414 °C; 2,577 °F)),
4877:International Union of Pure and Applied Chemistry
4855:. This is analogous to related compounds such as
17843:
15073:
6609:International Committee for Weights and Measures
6286:
5354:
5352:
5350:
3333:{\displaystyle K_{\rm {eq}}\approx K_{\rm {w}}=}
2347:
1696:by buffering large fluctuations in temperature.
403:
8640:Molecular Theory of Water and Aqueous Solutions
8529:. Techniques of Chemistry. Wiley-Interscience.
8253:
7077:. Archived from the original on August 7, 2007.
6985:
6502:Journal of Physical and Chemical Reference Data
5637:Journal of Physical and Chemical Reference Data
5164:Negative thermal expansion is also observed in
4867:(heavy water). Using chemical nomenclature for
4273:Water's Lewis base character makes it a common
3101:of all products and reactants including water:
2720:by internal rotations of the substituent water
2638:In general, ionic and polar substances such as
1867:
1805:
1766:with higher density than liquid water, such as
1577:Within the Earth's atmosphere and surface, the
1542:atom. Water is a tasteless, odorless liquid at
1423:, meaning that it can exhibit properties of an
1136:
212:
8604:Zumdahl, Steven S.; Zumdahl, Susan A. (2013).
8603:
8578:Ullmann's Encyclopedia of Industrial Chemistry
8072:
7988:
7961:
7949:
7937:
7925:
7880:
7868:
7856:
7841:
7592:
7154:
7130:
6973:
6911:"Lecture 12: Proton Conduction, Stoichiometry"
6559:National Institute of Standards and Technology
6369:
3626:at 25 °C is 23% higher than the value of
3583:The most notable physical differences between
3079:) equals that of the (solvated) hydrogen ion (
667:99.98 °C (211.96 °F; 373.13 K)
27:Physical and chemical properties of pure water
15059:
14005:
11207:
10087:
9178:
9154:
8775:
8274:
8254:Campbell, Mary K.; Farrell, Shawn O. (2007).
7437:
6997:
6869:
6867:
6282:
6280:
5959:
5347:
5267:(able to react both as an acid or an alkali).
5181:Other substances that expand on freezing are
3543:because of its higher density. It is used in
2731:
657:0.00 °C (32.00 °F; 273.15 K)
8696:. Wiley Online Library 2019. pp. 1–19.
8501:
7717:
7380:
7118:
6211:Handbook of semiconductor silicon technology
6010:
5829:
5800:
5716:, NIST Standard Reference Database Number 69
5358:
3974:, or electron-pair donor, in reactions with
2662:; the sodium chloride, NaCl, separates into
2362:Water molecule - structure and dipole moment
1715:. Before and since the advent of mechanical
1376:and the only common substance to exist as a
606:(temperature of maximum density, often 4 °C)
16935:
16699:
16622:
15594:
15134:
6759:Debenedetti, P. G.; Stanley, H. E. (2003).
6415:
5504:
5502:
5500:
4559:. An example of the oxidation of water is:
4432:
1725:
16518:
15066:
15052:
14012:
13998:
11214:
11200:
10094:
10080:
9185:
9171:
8782:
8768:
8275:Campbell, Neil A.; Reece, Jane B. (2009).
7633:
7269:
6915:University of Illinois at Urbana-Champaign
6908:
6864:
6277:
6249:
6247:
6245:
6243:
6241:
5835:
4947:, or a component in a mixture or mineral.
2475:Water molecules stay close to each other (
2238:
1465:, or approximately, the concentrations of
308:
30:"HOH" redirects here. For other uses, see
16541:
16405:
16267:
15174:
8506:(10th ed.). Boston, Mass.: Pearson.
8457:Lewis, William C.M.; Rice, James (1922).
8456:
7693:"Is 'Heavy Water' the Fountain of Youth?"
7406:
7295:
7238:
7068:"Physical Forces Organizing Biomolecules"
7043:
6938:
6724:
6686:
6630:
6143:
5936:
5926:
4890:Other systematic names for water include
4851:The simplest systematic name of water is
4425:reactions; the latter is then known as a
3966:Because the oxygen atom in water has two
3745:: it has the ability to act as either an
2067:, rather than the triple point of water.
2024:Triple point § Triple point of water
439:
15999:
15962:
15495:
15460:
8634:
8581:. Wiley-VCH Verlag GmbH & Co. KGaA.
7772:
7684:
7658:
7446:(1st ed.). HarperResource. p.
7166:
6636:
6487:
5847:IUPAC Compendium of Chemical Terminology
5508:
5497:
5423:
5421:
5419:
5089:
5087:
4832:used for naming derivatives of water by
4348:
3753:in chemical reactions. According to the
2793:
2580:
2546:
2534:
2505:
2460:
2454:
2357:
2233:
2027:
1956:
1871:
1809:
1784:
1729:
1667:
1572:
68:Ball-and-stick model of a water molecule
37:For broader coverage of this topic, see
16876:
16495:
15889:
15425:
8687:
8520:
8329:
7973:
7802:– via Applied Physics Laboratory.
7490:Theodore L. Brown; et al. (2015).
7329:
7270:Kolesnikov, Alexander I. (2016-04-22).
6876:Electrochemical and Solid-State Letters
6667:
6449:
6447:
6238:
5704:
5702:
5700:
5698:
1364:, which is nearly colorless apart from
328:
78:Space filling model of a water molecule
14:
17844:
16912:
16838:
16458:
15855:
15826:
15688:
15240:
14040:
9192:
8555:. Cambridge University Press. p.
7690:
6698:
6067:
6021:. IPCC WGI AR5 (Report). p. 257.
5889:Campbell, Williamson & Heyden 2006
5254:Using the fact that 0.5/0.0073 = 68.5.
5248:
4739:, was done in 1800 by English chemist
4555:. Almost all such reactions require a
4353:Some hydrogen-bonding contacts in FeSO
3736:
2888:
2857:Molecular orbital diagram § Water
2850:
2592:from high concentrations of dissolved
2076:Gustav Heinrich Johann Apollon Tammann
2063:, the kelvin is now defined using the
1490:
16214:
16076:
15390:
15047:
14732:
13993:
11195:
10075:
9166:
9153:
8763:
8543:
8469:CRC Handbook of Chemistry and Physics
8376:
8122:
7627:
7498:(13 ed.). Pearson. p. 351.
6820:
6814:
6498:for the Temperature Range 50 K-273 K"
5782:from the original on December 1, 2021
5416:
5231:
5084:
4281:complexes, examples of which include
2570:
466:Key: XLYOFNOQVPJJNP-UHFFFAOYSA-N
288:
268:
10101:
9123:
8465:
8226:
7549:
7537:
7512:from the original on 4 February 2024
7496:Chemistry : the central science
7104:, Surface Tension of Common Liquids.
7101:
6618:from the original on 27 January 2018
6453:
6444:
6061:
6049:
5727:
5695:
5689:
5677:
5546:
5534:
5491:
5473:from the original on 23 October 2023
4931:is a term used for hydrogen oxide (H
4507:Some other reactive metals, such as
4369:
3888:ion, and is thus acting as an acid:
3411:
2701:
2328:at 25.00 °C. Water can also be
8658:"Water Properties and Measurements"
8193:"Compound Summary for CID 22247451"
7711:
6565:from the original on 20 August 2018
6317:Physical Chemistry Chemical Physics
6253:
5774:. U.S. Department of the Interior.
5335:from the original on 13 August 2017
5210:(1-0.95865/1.00000) × 100% = 4.135%
5019:Optical properties of water and ice
4989:Electromagnetic absorption by water
4268:
2742:Water is relatively transparent to
2738:Electromagnetic absorption by water
2562:
2243:Pure water containing no exogenous
2082:The various triple points of water
1900:in all proportions. Water and most
394:
378:
24:
8828:
8628:
8260:(6th ed.). Cengage Learning.
8180:Leigh, Favre & Metanomski 1998
8135:Leigh, Favre & Metanomski 1998
8111:Leigh, Favre & Metanomski 1998
7782:Johns Hopkins APL Technical Digest
5099:
4761:isolated the first sample of pure
3316:
3312:
3293:
3283:
3268:
3253:
3250:
3203:
3194:
3174:
3170:
3149:
3139:
3120:
3117:
3094:for values at other temperatures.
3036:
3032:
3013:
3003:
2988:
2893:
2530:
1965:
1946:
1609:, other crystalline and amorphous
1132:
73:
63:
25:
17938:
8650:
8304:. Boston: Pearson Prentice Hall.
6941:Journal of Molecular Spectroscopy
4925:hydroxide by IUPAC nomenclature.
3757:definition, an acid is a proton (
2658:An example of an ionic solute is
9133:
9122:
9112:
9111:
9100:
8185:
8140:
8066:
8037:
8017:"Enterprise and electrolysis..."
8009:
7814:"Planetologie und Fernerkundung"
6601:Proceedings of the 106th meeting
6075:"Decree on weights and measures"
5511:"Properties of substance: water"
5257:
5155:and more complex ions that form.
5094:Vienna Standard Mean Ocean Water
4957:
4519:protective layer. Note that the
3446:Vienna Standard Mean Ocean Water
3431:
2971:for this reaction, known as the
2204:
1961:Vapor pressure diagrams of water
1607:common hexagonal crystalline ice
1544:ambient temperature and pressure
1305:
530:18.01528(33) g/mol
103:
50:
8751:Why does ice float in my drink?
8662:United States Geological Survey
8490:from the original on 2024-02-04
8406:from the original on 2019-12-12
8318:from the original on 2014-11-02
8219:
8205:from the original on 2014-08-27
8162:from the original on 2016-08-16
8075:The Journal of Chemical Physics
8055:from the original on 2023-05-29
8026:from the original on 2016-03-03
7908:from the original on 2016-06-22
7886:
7824:from the original on 2023-04-11
7806:
7795:from the original on 2023-04-11
7766:
7691:Müller, Grover C. (June 1937).
7581:from the original on 2017-01-28
7555:
7483:
7431:
7374:
7323:
7312:from the original on 2020-11-18
7263:
7206:
7195:from the original on 2020-08-01
7160:
7060:
7003:
6932:
6902:
6855:
6844:from the original on 2014-03-02
6797:from the original on 2018-11-01
6752:
6741:from the original on 2020-08-18
6692:
6661:
6637:Schlüter, Oliver (2003-07-28).
6592:
6555:"Base unit definitions: Kelvin"
6547:
6536:from the original on 2021-11-28
6481:
6470:from the original on 2007-10-28
6409:
6398:from the original on 2020-07-06
6305:
6266:from the original on 2016-06-25
6227:from the original on 2024-02-04
6198:
6116:
6077:. April 7, 1795. Archived from
6028:from the original on 2020-10-16
6004:
5953:
5894:
5871:from the original on 2019-04-29
5764:
5733:
5628:
5593:
5582:from the original on 2016-02-14
5576:University of California, Davis
5564:
5517:from the original on 2014-06-02
5405:from the original on 2019-12-01
5213:
5204:
5175:
5158:
5071:
4811:
4603:
4437:Water contains hydrogen in the
3342:
2017:
1888:List of water-miscible solvents
1780:very-high-density amorphous ice
1301:(at 25 °C , 100 kPa).
14019:
8702:10.1002/9781119300762.wsts0002
8459:A System of Physical Chemistry
8389:. Royal Society of Chemistry.
8333:Qualitative Inorganic Analysis
7442:The cartoon guide to chemistry
7297:10.1103/PhysRevLett.116.167802
6761:"Supercooled and Glassy Water"
6668:Tammann, Gustav H.J.A (1925).
6611:. Sèvres. 16–20 October 2017.
5772:"Water, the Universal Solvent"
5448:
5317:
5284:
3709:10 km is found in the oceans.
3327:
3307:
3304:
3277:
3047:
3027:
3024:
2997:
2904:In liquid water there is some
2802:(1) between molecules of water
1356:inorganic compound that is at
1091:Occupational safety and health
149:Hydrogen hydroxide (HH or HOH)
13:
1:
9002:Extraterrestrial liquid water
8587:10.1002/14356007.a28_001.pub3
8427:. Oxford: Blackwell Science.
8156:National Institutes of Health
8004:Greenwood & Earnshaw 1997
7652:10.1126/science.81.2098.273-a
7143:Greenwood & Earnshaw 1997
6648:Technische Universität Berlin
6358:Greenwood & Earnshaw 1997
6260:The USGS Water Science School
6111:Greenwood & Earnshaw 1997
5759:Greenwood & Earnshaw 1997
5602:Journal of Chemical Education
5368:Journal of Chemical Education
5273:
4869:type I ionic binary compounds
3684:
3655:Consumption of pure isolated
2348:Polarity and hydrogen bonding
2087:Phases in stable equilibrium
2070:Due to the existence of many
1896:with many liquids, including
1006:75.385 ± 0.05 J/(mol·K)
15075:Binary compounds of hydrogen
8476:(84th ed.). CRC Press.
7773:Prockter, Louise M. (2005).
7732:10.1016/0031-9384(79)90124-0
6961:10.1016/0022-2852(79)90019-5
5509:Anatolievich, Kiper Ruslan.
5292:"naming molecular compounds"
5065:
4871:, water would take the name
4804:were defined similarly. The
4662:The anode half reaction is:
3731:
3523:in 0.2% of water molecules.
3053:{\displaystyle K_{\rm {w}}=}
2789:
2555:Water has an unusually high
2524:Plateau–Rayleigh instability
2266:) and one hydronium cation (
1868:Miscibility and condensation
1806:Density of saltwater and ice
1030:69.95 ± 0.03 J/(mol·K)
7:
8237:10.1007/978-1-4615-4485-2_7
7383:"Water—an enduring mystery"
7167:Pugliano, N. (1992-11-01).
6986:Campbell & Farrell 2007
6418:Journal of Chemical Physics
5622:10.1021/acs.jchemed.6b00623
4950:
3064:, the concentration of the
2754:light, but it absorbs most
2393:. Due to the difference in
2385:Another consequence of its
2247:is an excellent electronic
2181:ice II, ice III, and ice V
2134:liquid water, ice III, and
1057:−285.83 ± 0.04 kJ/mol
59:
10:
17943:
14734:
11424:Praseodymium(III,IV) oxide
11221:
8020:Royal Society of Chemistry
7989:Zumdahl & Zumdahl 2013
7962:Zumdahl & Zumdahl 2013
7950:Zumdahl & Zumdahl 2013
7938:Zumdahl & Zumdahl 2013
7926:Zumdahl & Zumdahl 2013
7881:Zumdahl & Zumdahl 2013
7869:Zumdahl & Zumdahl 2013
7857:Zumdahl & Zumdahl 2013
7842:Zumdahl & Zumdahl 2013
7155:Zumdahl & Zumdahl 2013
7131:Zumdahl & Zumdahl 2013
6974:Zumdahl & Zumdahl 2013
6370:Zumdahl & Zumdahl 2013
6205:Bullis, W. Murray (1990).
6162:10.1103/PhysRevE.66.011202
5907:Proc. Natl. Acad. Sci. USA
5572:"What is the pKa of Water"
4979:Dihydrogen monoxide parody
4885:dihydrogen monoxide parody
4727:
4607:
2897:
2854:
2735:
2732:Electromagnetic absorption
2708:Quantum tunneling of water
2705:
2655:with non-polar molecules.
2653:van der Waals interactions
2574:
2351:
2192:ice II, ice V, and ice VI
2021:
1950:
1885:
1879:
1776:high-density amorphous ice
1749:negative thermal expansion
1494:
36:
29:
17815:
17656:
17427:
17084:Transition metal hydrides
17082:
17032:
16609:
16065:
15646:
15585:
15229:
15121:
15081:
14042:
14027:
13973:
13927:
13846:
13767:
13616:
13452:
12878:
12136:
11731:
11512:
11229:
10109:
9200:
9160:
9155:Links to related articles
9095:
9062:
8994:
8921:
8880:
8840:
8826:
8797:
8789:
8461:. Longmans, Green and Co.
8356:Chemistry of the Elements
8354:; Earnshaw, Alan (1997).
8283:(8th ed.). Pearson.
8198:Pubchem Compound Database
7820:(in German). 2006-02-28.
7775:"Ice in the Solar System"
7720:Physiology & Behavior
7087:: CS1 maint: unfit URL (
6998:Campbell & Reece 2009
6670:The States Of Aggregation
6456:"Bulk Elastic Properties"
5843:"Autoprotolysis constant"
5241:as 1,000,000 dynes/cm (a
4974:Chemical bonding of water
4879:(IUPAC). Another name is
2293:dissolved in it, often a
2220:crystalline states of ice
2148:liquid water, ice V, and
1876:Red line shows saturation
1597:, or loosely accumulated
1360:a tasteless and odorless
1295:
1288:
1283:
1196:
1088:
1083:
986:
923:
495:
475:
450:
196:
140:
128:
116:
111:
102:
58:
49:
8746:Water Density Calculator
8736:dynamic liquid viscosity
8688:Chaplin, Martin (2019).
6464:Georgia State University
6016:"3: Observations: Ocean"
6014:; Rintoul, S.R. (2013).
5442:10.1088/0026-1394/38/4/3
5278:
4834:substituent nomenclature
4433:Water in redox reactions
3970:, water often acts as a
3099:thermodynamic activities
2900:Self-ionization of water
2879:molecular orbital theory
2111:273.16 K (0.01 °C)
1953:Vapour pressure of water
1862:thermohaline circulation
1726:Density of water and ice
1546:. Liquid water has weak
1497:Water chemistry analysis
1366:an inherent hint of blue
1284:Supplementary data page
771:Partially miscible with
12654:Praseodymium(III) oxide
11390:Manganese(II,III) oxide
8466:Lide, David R. (2003).
8301:Biology: Exploring Life
7761:Weingärtner et al. 2016
7697:Popular Science Monthly
7670:Popular Science Monthly
7276:Physical Review Letters
7240:10.1126/science.aae0012
6587:Weingärtner et al. 2016
6488:Neumeier, J.J. (2018).
6388:"Can the ocean freeze?"
5928:10.1073/pnas.0506899102
5855:10.1351/goldbook.A00532
5818:Weingärtner et al. 2016
5559:Weingärtner et al. 2016
5059:Water thread experiment
4749:Joseph Louis Gay-Lussac
2782:in the red part of the
2772:photosynthetic pigments
2368:bent molecular geometry
2318:electrical conductivity
2239:Electrical conductivity
1408:of 100 °C for its
695:Improved solubility in
17912:Extraterrestrial water
17817:Exotic matter hydrides
13225:Protactinium(IV) oxide
13211:Praseodymium(IV) oxide
12399:Einsteinium(III) oxide
12297:Californium(III) oxide
11715:Water (hydrogen oxide)
11230:Mixed oxidation states
9012:Planetary oceanography
8834:
8521:Riddick, John (1970).
6699:Murphy, D. M. (2005).
6392:National Ocean Service
5714:NIST Chemistry WebBook
5325:"Definition of Hydrol"
5081:of water is incorrect.
5034:Viscosity § Water
4984:Double distilled water
4753:Alexander von Humboldt
4409:. Water can also be a
4366:
3367:. It also occurs when
3334:
3220:
3054:
2803:
2604:Water is an excellent
2601:
2552:
2544:
2527:
2472:
2449:specific heat capacity
2363:
2040:
1962:
1877:
1818:
1790:
1735:
1678:specific heat capacity
1676:Water has a very high
1673:
1143:
79:
69:
13975:Oxides are sorted by
13826:Technetium(VII) oxide
13562:Protactinium(V) oxide
12671:Promethium(III) oxide
12450:Gadolinium(III) oxide
12382:Dysprosium(III) oxide
12006:Protactinium monoxide
11458:Terbium(III,IV) oxide
8832:
8694:Encyclopedia of Water
8679:(simpler formulation)
8545:Sharp, Robert Phillip
8360:Butterworth-Heinemann
7492:"9.2 The Vsepr Model"
7381:Ball, Philip (2008).
7016:Nature Communications
6687:Lewis & Rice 1922
6674:Constable And Company
4610:Electrolysis of water
4531:Water can be oxidized
4352:
3861:In the reaction with
3335:
3221:
3055:
2797:
2584:
2550:
2538:
2509:
2464:
2455:Cohesion and adhesion
2361:
2234:Electrical properties
2031:
1960:
1875:
1813:
1788:
1733:
1684:), as well as a high
1671:
1617:of water is known as
1585:of water is known as
1573:Water, ice, and vapor
1461:. The product of the
1395:Water molecules form
1142:
182:κ-Hydroxylhydrogen(0)
130:Systematic IUPAC name
77:
67:
17852:Amphoteric compounds
13775:Dichlorine heptoxide
13545:Phosphorus pentoxide
13511:Dinitrogen pentoxide
13309:Technetium(IV) oxide
13026:Dinitrogen tetroxide
12841:Ytterbium(III) oxide
12603:Neodymium(III) oxide
12586:Manganese(III) oxide
12229:Berkelium(III) oxide
12178:Americium(III) oxide
11339:Dichlorine pentoxide
11322:Cobalt(II,III) oxide
11288:Chlorine perchlorate
9075:Ocean stratification
8908:Doubly labeled water
8352:Greenwood, Norman N.
8330:Charlot, G. (2007).
7928:, pp. 932, 936.
7818:www.geo.fu-berlin.de
7352:10.1364/ao.36.008710
6081:on February 25, 2013
5361:"Why is water blue?"
5302:on 24 September 2018
4840:is recommended over
4759:Gilbert Newton Lewis
4427:dehydration reaction
4382:, a hydroxyl group (
4283:metal aquo complexes
3241:
3108:
2979:
2969:equilibrium constant
2840:Stockholm University
2516:Cohesion (chemistry)
2482:Water also has high
2468:drops adhering to a
2320:of 0.05501 ± 0.0001
2129:251 K (−22 °C)
1686:heat of vaporization
1634:critical temperature
1554:, also appear blue.
1125:(fire diamond)
1079:−237.24 kJ/mol
905:1.3330 (20 °C)
881:Thermal conductivity
158:(DHMO) (parody name)
118:Preferred IUPAC name
32:HOH (disambiguation)
17917:Transport phenomena
17867:Triatomic molecules
17429:Lanthanide hydrides
17036:(Group 17 hydrides)
16615:(Group 16 hydrides)
16069:(Group 15) hydrides
13882:Ruthenium tetroxide
13792:Manganese heptoxide
13638:Molybdenum trioxide
13253:Ruthenium(IV) oxide
13183:Plutonium(IV) oxide
13127:Neptunium(IV) oxide
12928:Californium dioxide
12900:Berkelium(IV) oxide
12824:Vanadium(III) oxide
12807:Tungsten(III) oxide
12790:Titanium(III) oxide
12756:Thallium(III) oxide
12705:Samarium(III) oxide
12637:Phosphorus trioxide
12569:Lutetium(III) oxide
12433:Europium(III) oxide
12365:Dinitrogen trioxide
12331:Chromium(III) oxide
12280:Caesium sesquioxide
12144:Actinium(III) oxide
11986:Phosphorus monoxide
11976:Palladium(II) oxide
11936:Manganese(II) oxide
11739:Aluminium(II) oxide
11572:Dichlorine monoxide
11492:Triuranium octoxide
11475:Tribromine octoxide
11441:Silver(I,III) oxide
11305:Chloryl perchlorate
9080:Lake stratification
9063:Physical parameters
8087:1933JChPh...1..341L
7644:1935Sci....81..273U
7613:2001JChPh.114.3174H
7399:2008Natur.452..291B
7344:1997ApOpt..36.8710P
7288:2016PhRvL.116p7802K
7231:2016Sci...351.1310R
7225:(6279): 1310–1313.
7173:UNT Digital Library
7133:, pp. 458–459.
7075:Biophysical Society
7028:2014NatCo...5.4565C
6953:1979JMoSp..74....1H
6909:Crofts, A. (1996).
6780:2003PhT....56f..40D
6717:2005QJRMS.131.1539M
6514:2018JPCRD..47c3101N
6430:1973JChPh..59.5529F
6329:2001PCCP....3.5355L
6289:Inorganic Chemistry
6154:2002PhRvE..66a1202S
5974:2007SMat....3..797D
5919:2005PNAS..10214171S
5913:(40): 14171–14174.
5730:, 9—Dipole Moments.
5649:1995JPCRD..24.1377R
5614:2017JChEd..94..690S
5380:1993JChEd..70..612B
5224:resulting from the
4881:dihydrogen monoxide
4378:; for example in a
3737:Acid–base reactions
3539:, is also known as
3501:present in 99.76%,
3376:clathrate compounds
3374:Water ice can form
3213:
2889:Chemical properties
2875:valence bond theory
2851:Molecular structure
2692:crystalline lattice
2596:turns the water of
2520:Van der Waals force
2389:is that water is a
2116:liquid water, ice I
2083:
1630:supercritical fluid
1628:Water also forms a
1559:standard conditions
1491:Physical properties
915:0.890 mPa·s (0.890
777:methyl ethyl ketone
644:0.9167 g/mL at 0 °C
250:Beilstein Reference
156:Dihydrogen monoxide
46:
17887:Neutron moderators
17882:Inorganic solvents
17862:Hydrogen compounds
15085:(Group 1) hydrides
13847:+8 oxidation state
13809:Rhenium(VII) oxide
13768:+7 oxidation state
13708:Tellurium trioxide
13617:+6 oxidation state
13579:Tantalum pentoxide
13460:Antimony pentoxide
13453:+5 oxidation state
13421:Vanadium(IV) oxide
13393:Tungsten(IV) oxide
12998:Chromium(IV) oxide
12879:+4 oxidation state
12858:Yttrium(III) oxide
12773:Thulium(III) oxide
12739:Terbium(III) oxide
12688:Rhodium(III) oxide
12501:Holmium(III) oxide
12467:Gallium(III) oxide
12246:Bismuth(III) oxide
12137:+3 oxidation state
12103:Vanadium(II) oxide
12093:Titanium(II) oxide
11886:Germanium monoxide
11876:Europium(II) oxide
11859:Dinitrogen dioxide
11829:Chromium(II) oxide
11759:Berkelium monoxide
11732:+2 oxidation state
11520:Aluminium(I) oxide
11513:+1 oxidation state
11407:Mellitic anhydride
11356:Iron(II,III) oxide
11237:Antimony tetroxide
9194:Hydrogen compounds
9027:List of Candidates
8888:Deuterium-depleted
8835:
8051:. Footnote 122-1.
7904:. 2 October 2013.
7036:10.1038/ncomms4565
6837:. September 2011.
6711:(608): 1539–1565.
5746:2023-07-28 at the
5009:Hydrogen polyoxide
4900:hydrogen hydroxide
4770:temperature scales
4380:hydration reaction
4367:
4363:metal aquo complex
4314:center. In solid
3877:, water donates a
3781:ion from HCl when
3555:, decaying with a
3380:clathrate hydrates
3330:
3216:
3187:
3050:
2804:
2602:
2571:Water as a solvent
2553:
2545:
2528:
2473:
2399:bond dipole moment
2364:
2105:, and water vapor
2081:
2065:Boltzmann constant
2041:
1963:
1878:
1839:lakes and rivers.
1819:
1791:
1736:
1705:enthalpy of fusion
1674:
1650:hydrothermal vents
1503:chemical substance
1386:molecular hydrogen
1328:Infobox references
1224:Hydrogen telluride
1197:Related compounds
1144:
1114:Water intoxication
763:dimethyl sulfoxide
677:Poorly soluble in
167:Hydrohydroxic acid
80:
70:
44:
17839:
17838:
17833:
17832:
17658:Actinide hydrides
17034:Hydrogen halides
16996:
16961:
16960:
16931:
16930:
16908:
16907:
16872:
16871:
16834:
16833:
16695:
16694:
16601:
16567:
16566:
16537:
16536:
16514:
16513:
16491:
16490:
16454:
16453:
16401:
16400:
16263:
16262:
16210:
16209:
16061:
16060:
16018:
16017:
15995:
15994:
15958:
15957:
15885:
15884:
15851:
15850:
15822:
15821:
15684:
15683:
15642:
15641:
15587:Group 14 hydrides
15581:
15580:
15526:
15525:
15491:
15490:
15456:
15455:
15421:
15420:
15386:
15385:
15225:
15224:
15170:
15169:
15041:
15040:
15035:
15034:
13987:
13986:
13910:Hassium tetroxide
13854:Iridium tetroxide
13722:Tungsten trioxide
13680:Selenium trioxide
13652:Polonium trioxide
13624:Chromium trioxide
13596:Vanadium(V) oxide
13528:Niobium pentoxide
13494:Bismuth pentoxide
13477:Arsenic pentoxide
13435:Zirconium dioxide
13337:Terbium(IV) oxide
13323:Tellurium dioxide
13239:Rhodium(IV) oxide
13113:Manganese dioxide
13085:Hafnium(IV) oxide
13043:Germanium dioxide
12886:Americium dioxide
12620:Nickel(III) oxide
12518:Indium(III) oxide
12416:Erbium(III) oxide
12348:Cobalt(III) oxide
12314:Cerium(III) oxide
12195:Antimony trioxide
12113:Yttrium(II) oxide
11996:Polonium monoxide
11946:Mercury(II) oxide
11819:Chlorine monoxide
11689:Thallium(I) oxide
11559:Dicarbon monoxide
11373:Lead(II,IV) oxide
11189:
11188:
10069:
10068:
9147:
9146:
9087:Ocean temperature
8619:978-1-13-361109-7
8566:978-0-521-33009-1
8434:978-0-86542-685-6
8396:978-0-85404-438-2
8369:978-0-08-037941-8
8343:978-1-4067-4789-8
8311:978-0-13-250882-7
8290:978-0-8053-6844-4
8267:978-0-495-39041-1
8182:, pp. 27–28.
8148:"Tetrahydropyran"
8095:10.1063/1.1749300
8049:1902 Encyclopedia
7621:10.1063/1.1340584
7505:978-0-321-91041-7
7393:(7185): 291–292.
7119:Reece et al. 2013
6988:, pp. 37–38.
6888:10.1149/1.1836121
6788:10.1063/1.1595053
6522:10.1063/1.5030640
6438:10.1063/1.1679903
6323:(24): 5355–5357.
6254:Perlman, Howard.
5864:978-0-9678550-9-7
5830:Reece et al. 2013
5801:Reece et al. 2013
5388:10.1021/ed070p612
5221:Adiabatic cooling
5044:Water (data page)
5029:Superheated water
4921:, is also called
4873:hydrogen monoxide
4857:hydrogen peroxide
4820:name of water is
4741:William Nicholson
4588:→ 4 AgF + 4 HF +
4449:metals, and some
4370:Organic chemistry
3783:hydrochloric acid
3549:neutron moderator
3526:Deuterium oxide,
3412:Acidity in nature
3384:methane clathrate
3361:mineral hydration
3214:
2812:hydrogen fluoride
2756:ultraviolet light
2714:quantum tunneling
2702:Quantum tunneling
2618:attractive forces
2590:calcium carbonate
2395:electronegativity
2354:Chemical polarity
2202:
2201:
1745:thermal expansion
1642:critical pressure
1525:of water has two
1336:Chemical compound
1334:
1333:
1290:Water (data page)
1270:Hydrogen fluoride
1234:Hydrogen peroxide
1229:Hydrogen polonide
1219:Hydrogen selenide
1185:Safety data sheet
1064:Gibbs free energy
931:Crystal structure
755:dimethylformamide
463:InChI=1S/H2O/h1H2
238:Interactive image
179:μ-Oxidodihydrogen
98:
97:
92: Hydrogen, H
16:(Redirected from
17934:
17927:Greenhouse gases
17872:Oxygen compounds
16994:
16933:
16932:
16910:
16909:
16874:
16873:
16836:
16835:
16697:
16696:
16620:
16619:
16599:
16539:
16538:
16516:
16515:
16493:
16492:
16456:
16455:
16403:
16402:
16265:
16264:
16212:
16211:
16074:
16073:
16020:
16019:
15997:
15996:
15960:
15959:
15887:
15886:
15853:
15852:
15824:
15823:
15686:
15685:
15644:
15643:
15592:
15591:
15528:
15527:
15493:
15492:
15458:
15457:
15423:
15422:
15388:
15387:
15238:
15237:
15172:
15171:
15132:
15131:
15068:
15061:
15054:
15045:
15044:
15012:
15002:
14989:
14976:
14967:
14957:
14944:
14935:
14922:
14912:
14897:
14887:
14877:
14867:
14857:
14847:
14837:
14827:
14818:
14808:
14798:
14788:
14778:
14768:
14759:
14749:
14676:
14650:
14640:
14631:
14621:
14607:
14597:
14566:
14553:
14528:
14518:
14508:
14499:
14489:
14479:
14461:
14448:
14432:
14422:
14410:
14382:
14372:
14362:
14352:
14342:
14328:
14318:
14308:
14299:
14289:
14279:
14270:
14260:
14251:
14241:
14232:
14223:
14213:
14201:
14176:
14166:
14156:
14146:
14136:
14111:
14101:
14091:
14081:
14071:
14061:
14031:
14030:
14014:
14007:
14000:
13991:
13990:
13868:Osmium tetroxide
13736:Uranium trioxide
13666:Rhenium trioxide
13379:Titanium dioxide
13267:Selenium dioxide
13197:Polonium dioxide
13169:Platinum dioxide
13141:Nitrogen dioxide
13012:Curium(IV) oxide
12984:Chlorine dioxide
12970:Cerium(IV) oxide
12212:Arsenic trioxide
12073:Thorium monoxide
12056:Disulfur dioxide
12026:Silicon monoxide
11956:Nickel(II) oxide
11849:Copper(II) oxide
11839:Cobalt(II) oxide
11779:Bromine monoxide
11624:Mercury(I) oxide
11585:Gallium(I) oxide
11546:Caesium monoxide
11216:
11209:
11202:
11193:
11192:
11177:
11163:
11149:
11135:
11121:
11107:
11093:
11083:
11076:
11065:
10299:
10298:
10297:
10289:
10288:
10103:Oxygen compounds
10096:
10089:
10082:
10073:
10072:
10061:
10050:
10039:
10025:
10011:
9997:
9986:
9971:
9953:
9935:
9917:
9906:
9888:
9874:
9860:
9836:
9821:
9807:
9793:
9779:
9768:
9754:
9743:
9728:
9710:
9692:
9678:
9664:
9650:
9636:
9622:
9608:
9594:
9583:
9569:
9558:
9540:
9530:
9510:
9495:
9481:
9467:
9457:
9447:
9437:
9407:
9393:
9379:
9364:
9340:
9330:
9320:
9300:
9290:
9280:
9260:
9249:
9228:
9214:
9187:
9180:
9173:
9164:
9163:
9151:
9150:
9137:
9126:
9125:
9115:
9114:
9104:
9007:Asteroidal water
8995:Extraterrestrial
8784:
8777:
8770:
8761:
8760:
8723:
8673:
8671:
8669:
8646:
8644:World Scientific
8623:
8610:Cengage Learning
8608:(9th ed.).
8600:
8570:
8554:
8540:
8528:
8517:
8504:Campbell Biology
8498:
8496:
8495:
8462:
8453:
8451:
8445:. Archived from
8426:
8414:
8412:
8411:
8405:
8388:
8373:
8358:(2nd ed.).
8347:
8326:
8324:
8323:
8294:
8282:
8271:
8250:
8214:
8213:
8211:
8210:
8189:
8183:
8177:
8171:
8170:
8168:
8167:
8144:
8138:
8132:
8126:
8120:
8114:
8108:
8099:
8098:
8070:
8064:
8063:
8061:
8060:
8041:
8035:
8034:
8032:
8031:
8013:
8007:
8001:
7992:
7986:
7977:
7971:
7965:
7959:
7953:
7947:
7941:
7935:
7929:
7923:
7917:
7916:
7914:
7913:
7890:
7884:
7878:
7872:
7866:
7860:
7854:
7845:
7839:
7833:
7832:
7830:
7829:
7810:
7804:
7803:
7801:
7800:
7794:
7779:
7770:
7764:
7758:
7752:
7751:
7715:
7709:
7708:
7706:
7704:
7688:
7682:
7681:
7679:
7677:
7662:
7656:
7655:
7631:
7625:
7624:
7607:(7): 3174–3181.
7596:
7590:
7589:
7587:
7586:
7580:
7567:
7559:
7553:
7547:
7541:
7535:
7529:
7528:
7519:
7517:
7487:
7481:
7480:
7445:
7435:
7429:
7428:
7410:
7378:
7372:
7371:
7327:
7321:
7320:
7318:
7317:
7299:
7267:
7261:
7260:
7242:
7210:
7204:
7203:
7201:
7200:
7164:
7158:
7152:
7146:
7140:
7134:
7128:
7122:
7116:
7105:
7099:
7093:
7092:
7086:
7078:
7072:
7064:
7058:
7057:
7047:
7007:
7001:
6995:
6989:
6983:
6977:
6971:
6965:
6964:
6936:
6930:
6929:
6927:
6926:
6917:. Archived from
6906:
6900:
6899:
6871:
6862:
6859:
6853:
6852:
6850:
6849:
6843:
6832:
6824:
6818:
6812:
6806:
6805:
6803:
6802:
6796:
6765:
6756:
6750:
6749:
6747:
6746:
6728:
6726:10.1256/qj.04.94
6696:
6690:
6684:
6678:
6677:
6665:
6659:
6658:
6656:
6650:. Archived from
6645:
6634:
6628:
6627:
6625:
6623:
6617:
6606:
6596:
6590:
6584:
6575:
6574:
6572:
6570:
6551:
6545:
6544:
6542:
6541:
6485:
6479:
6478:
6476:
6475:
6451:
6442:
6441:
6413:
6407:
6406:
6404:
6403:
6384:
6373:
6367:
6361:
6355:
6349:
6348:
6337:10.1039/b108676f
6313:Loerting, Thomas
6309:
6303:
6302:
6284:
6275:
6274:
6272:
6271:
6251:
6236:
6235:
6233:
6232:
6202:
6196:
6195:
6193:
6192:
6186:
6180:. Archived from
6147:
6145:cond-mat/0203383
6129:
6120:
6114:
6108:
6097:
6096:
6088:
6086:
6071:
6065:
6059:
6053:
6047:
6041:
6040:
6034:
6033:
6027:
6020:
6008:
6002:
6001:
5982:10.1039/b611584e
5957:
5951:
5950:
5940:
5930:
5898:
5892:
5886:
5880:
5879:
5877:
5876:
5839:
5833:
5827:
5821:
5815:
5804:
5798:
5792:
5791:
5789:
5787:
5768:
5762:
5756:
5750:
5737:
5731:
5725:
5719:
5706:
5693:
5687:
5681:
5675:
5669:
5668:
5657:10.1063/1.555963
5643:(3): 1377–1381.
5632:
5626:
5625:
5597:
5591:
5590:
5588:
5587:
5568:
5562:
5556:
5550:
5544:
5538:
5532:
5526:
5525:
5523:
5522:
5506:
5495:
5489:
5483:
5482:
5480:
5478:
5452:
5446:
5445:
5425:
5414:
5413:
5411:
5410:
5404:
5365:
5356:
5345:
5344:
5342:
5340:
5321:
5315:
5314:
5309:
5307:
5298:. Archived from
5288:
5268:
5261:
5255:
5252:
5246:
5235:
5229:
5217:
5211:
5208:
5202:
5179:
5173:
5162:
5156:
5154:
5153:
5152:
5142:
5141:
5133:
5132:
5125:
5124:
5114:
5113:
5112:
5103:
5097:
5091:
5082:
5075:
4967:
4962:
4961:
4920:
4919:
4918:
4911:
4910:
4861:hydrogen sulfide
4745:Anthony Carlisle
4719:
4718:
4717:
4710:
4709:
4701:
4700:
4699:
4690:
4689:
4688:
4678:
4676:
4675:
4658:
4657:
4656:
4646:
4645:
4644:
4637:
4636:
4628:
4627:
4626:
4599:
4598:
4597:
4587:
4585:
4584:
4574:
4573:
4572:
4554:
4552:
4551:
4543:
4542:
4503:
4502:
4501:
4492:
4491:
4490:
4481:
4480:
4479:
4469:
4467:
4466:
4405:of proteins and
4401:of fats and the
4392:
4391:
4390:
4341:
4338:
4337:
4329:
4328:
4305:
4304:
4303:
4295:
4294:
4279:transition metal
4269:Ligand chemistry
4264:
4263:
4262:
4252:
4250:
4249:
4239:
4238:
4237:
4229:
4228:
4216:their basic pH:
4200:
4199:
4198:
4190:
4188:
4187:
4175:
4174:
4173:
4172:
4167:
4166:
4164:
4163:
4150:
4149:
4148:
4147:
4142:
4141:
4140:
4139:
4126:
4125:
4124:
4116:
4114:
4113:
4101:
4100:
4099:
4098:
4093:
4092:
4090:
4089:
4076:
4075:
4074:
4073:
4068:
4067:
4066:
4065:
4052:
4051:
4050:
4043:
4042:
4032:
4031:
4030:
4029:
4024:
4023:
4021:
4020:
4007:
4006:
4005:
4004:
3999:
3998:
3997:
3996:
3962:
3961:
3960:
3951:
3950:
3949:
3939:
3938:
3937:
3936:
3931:
3930:
3928:
3927:
3914:
3913:
3912:
3911:
3906:
3905:
3904:
3903:
3887:
3886:
3885:
3876:
3875:
3874:
3857:
3856:
3855:
3846:
3845:
3844:
3837:
3836:
3826:
3825:
3824:
3823:
3818:
3817:
3815:
3814:
3801:
3800:
3799:
3798:
3793:
3780:
3779:
3778:
3769:
3767:
3766:
3708:
3704:
3700:
3699:
3698:
3680:
3667:
3665:
3664:
3651:
3649:
3648:
3638:
3636:
3635:
3625:
3623:
3622:
3608:
3606:
3605:
3595:
3593:
3592:
3579:
3577:
3576:
3566:
3562:
3545:nuclear reactors
3538:
3536:
3535:
3522:
3520:
3519:
3511:
3509:
3508:
3500:
3498:
3497:
3489:
3487:
3486:
3470:
3468:
3467:
3407:
3405:
3404:
3396:
3395:
3339:
3337:
3336:
3331:
3326:
3325:
3324:
3323:
3303:
3302:
3301:
3300:
3291:
3290:
3273:
3272:
3271:
3258:
3257:
3256:
3225:
3223:
3222:
3217:
3215:
3212:
3207:
3206:
3202:
3201:
3186:
3185:
3184:
3183:
3182:
3181:
3160:
3159:
3158:
3157:
3156:
3147:
3146:
3130:
3125:
3124:
3123:
3089:
3088:
3087:
3078:
3077:
3076:
3059:
3057:
3056:
3051:
3046:
3045:
3044:
3043:
3023:
3022:
3021:
3020:
3011:
3010:
2993:
2992:
2991:
2963:
2962:
2961:
2952:
2951:
2950:
2943:
2942:
2932:
2930:
2929:
2871:sp hybridization
2784:visible spectrum
2748:near ultraviolet
2686:
2685:
2684:
2672:
2671:
2670:
2622:precipitated out
2577:Aqueous solution
2563:Capillary action
2445:room temperature
2442:
2440:
2439:
2429:
2427:
2426:
2407:hydrogen bonding
2343:
2338:proton conductor
2285:
2284:
2283:
2276:
2275:
2265:
2264:
2263:
2084:
2080:
2061:starting in 2019
1997:
1995:
1989:
1987:
1981:
1979:
1925:partial pressure
1690:hydrogen bonding
1567:hydrogen bonding
1548:absorption bands
1520:
1518:
1517:
1507:chemical formula
1486:
1485:
1484:
1475:
1474:
1473:
1455:
1454:
1453:
1442:
1441:
1440:
1358:room temperature
1350:
1318:
1312:
1309:
1308:
1214:Hydrogen sulfide
1164:
1157:
1150:
1135:
1075:
1053:
1026:
1002:
987:Thermochemistry
892:Refractive index
638:
634:
632:
629:
622:
618:
616:
613:
605:
603:
597:
595:
592:
585:
581:
579:
576:
542:, colorless gas
519:
517:
516:
503:Chemical formula
443:
407:
396:
382:
360:Gmelin Reference
343:
332:
312:
292:
272:
240:
216:
161:Dihydrogen oxide
107:
91:
85:
60:
54:
47:
43:
21:
17942:
17941:
17937:
17936:
17935:
17933:
17932:
17931:
17897:Water chemistry
17842:
17841:
17840:
17835:
17834:
17829:
17811:
17807:
17799:
17791:
17783:
17775:
17767:
17759:
17751:
17743:
17735:
17727:
17719:
17711:
17703:
17696:
17692:
17685:
17677:
17669:
17652:
17648:
17640:
17632:
17624:
17616:
17608:
17600:
17592:
17584:
17576:
17568:
17560:
17552:
17544:
17536:
17528:
17520:
17512:
17504:
17496:
17488:
17480:
17472:
17464:
17456:
17448:
17440:
17423:
17413:
17404:
17396:
17392:
17379:
17371:
17340:
17326:
17311:
17296:
17288:
17280:
17272:
17264:
17251:
17243:
17230:
17217:
17204:
17191:
17183:
17175:
17167:
17159:
17151:
17143:
17135:
17127:
17119:
17111:
17103:
17095:
17078:
17035:
17028:
17023:
17014:
16991:
16982:
16974:
16957:
16947:
16927:
16923:
16904:
16900:
16896:
16887:
16868:
16864:
16860:
16851:
16830:
16821:
16817:
16809:
16805:
16797:
16793:
16785:
16781:
16773:
16769:
16761:
16757:
16749:
16745:
16737:
16733:
16725:
16721:
16712:
16691:
16683:
16679:
16671:
16667:
16659:
16655:
16647:
16643:
16634:
16614:
16612:
16605:
16597:
16588:
16575:
16563:
16553:
16533:
16529:
16510:
16506:
16487:
16483:
16479:
16471:
16450:
16446:
16442:
16434:
16430:
16422:
16418:
16397:
16388:
16384:
16376:
16372:
16364:
16360:
16352:
16348:
16340:
16336:
16328:
16324:
16316:
16312:
16304:
16300:
16292:
16288:
16280:
16259:
16255:
16251:
16243:
16239:
16231:
16227:
16206:
16197:
16193:
16185:
16181:
16173:
16169:
16161:
16157:
16149:
16145:
16137:
16133:
16125:
16121:
16113:
16109:
16101:
16097:
16089:
16068:
16057:
16053:
16045:
16014:
16010:
15991:
15987:
15983:
15975:
15954:
15950:
15946:
15938:
15934:
15926:
15922:
15914:
15910:
15902:
15881:
15872:
15868:
15847:
15843:
15839:
15818:
15809:
15805:
15797:
15793:
15785:
15781:
15773:
15769:
15761:
15757:
15749:
15745:
15737:
15733:
15725:
15721:
15713:
15709:
15701:
15680:
15675:
15667:
15659:
15638:
15577:
15573:
15565:
15561:
15553:
15522:
15518:
15514:
15506:
15487:
15483:
15479:
15471:
15452:
15448:
15444:
15436:
15417:
15413:
15409:
15401:
15382:
15378:
15374:
15366:
15362:
15354:
15350:
15342:
15338:
15330:
15326:
15318:
15314:
15306:
15302:
15294:
15290:
15282:
15278:
15270:
15266:
15253:
15232:
15221:
15217:
15209:
15201:
15193:
15185:
15166:
15126:
15124:
15117:
15084:
15077:
15072:
15042:
15037:
15036:
15011:
15007:
15001:
14997:
14988:
14984:
14980:
14978:
14975:
14971:
14969:
14966:
14962:
14956:
14952:
14948:
14946:
14943:
14939:
14937:
14934:
14930:
14921:
14917:
14911:
14907:
14896:
14892:
14886:
14882:
14876:
14872:
14866:
14862:
14856:
14852:
14846:
14842:
14836:
14832:
14826:
14822:
14820:
14817:
14813:
14807:
14803:
14797:
14793:
14787:
14783:
14777:
14773:
14767:
14763:
14761:
14758:
14754:
14748:
14744:
14675:
14671:
14649:
14645:
14639:
14635:
14633:
14630:
14626:
14620:
14616:
14614:
14606:
14602:
14596:
14592:
14565:
14561:
14552:
14548:
14527:
14523:
14517:
14513:
14507:
14503:
14501:
14498:
14494:
14488:
14484:
14478:
14474:
14460:
14456:
14447:
14443:
14431:
14427:
14421:
14417:
14409:
14405:
14381:
14377:
14371:
14367:
14361:
14357:
14351:
14347:
14341:
14337:
14335:
14327:
14323:
14317:
14313:
14307:
14303:
14301:
14298:
14294:
14288:
14284:
14278:
14274:
14272:
14269:
14265:
14259:
14255:
14253:
14250:
14246:
14240:
14236:
14234:
14231:
14227:
14225:
14222:
14218:
14212:
14208:
14200:
14196:
14175:
14171:
14165:
14161:
14155:
14151:
14145:
14141:
14135:
14131:
14110:
14106:
14099:
14095:
14093:
14090:
14086:
14080:
14076:
14070:
14066:
14060:
14056:
14023:
14018:
13988:
13983:
13981:Category:Oxides
13977:oxidation state
13969:
13923:
13919:
13905:
13896:Xenon tetroxide
13891:
13877:
13863:
13842:
13838:
13834:
13821:
13817:
13804:
13800:
13787:
13783:
13763:
13759:
13745:
13731:
13717:
13703:
13694:Sulfur trioxide
13689:
13675:
13661:
13647:
13633:
13612:
13608:
13604:
13591:
13587:
13574:
13570:
13557:
13553:
13540:
13536:
13523:
13519:
13506:
13502:
13489:
13485:
13472:
13468:
13448:
13444:
13430:
13416:
13407:Uranium dioxide
13402:
13388:
13374:
13360:
13351:Thorium dioxide
13346:
13332:
13318:
13304:
13290:
13281:Silicon dioxide
13276:
13262:
13248:
13234:
13220:
13206:
13192:
13178:
13164:
13150:
13136:
13122:
13108:
13094:
13080:
13071:Iridium dioxide
13066:
13052:
13038:
13034:
13021:
13007:
12993:
12979:
12965:
12956:Carbon trioxide
12951:
12937:
12923:
12914:Bromine dioxide
12909:
12895:
12874:
12870:
12866:
12853:
12849:
12836:
12832:
12819:
12815:
12802:
12798:
12785:
12781:
12768:
12764:
12751:
12747:
12734:
12730:
12717:
12713:
12700:
12696:
12683:
12679:
12666:
12662:
12649:
12645:
12632:
12628:
12615:
12611:
12598:
12594:
12581:
12577:
12564:
12560:
12552:Lanthanum oxide
12547:
12543:
12535:Iron(III) oxide
12530:
12526:
12513:
12509:
12496:
12492:
12484:Gold(III) oxide
12479:
12475:
12462:
12458:
12445:
12441:
12428:
12424:
12411:
12407:
12394:
12390:
12377:
12373:
12360:
12356:
12343:
12339:
12326:
12322:
12309:
12305:
12292:
12288:
12275:
12271:
12258:
12254:
12241:
12237:
12224:
12220:
12207:
12203:
12190:
12186:
12173:
12169:
12161:Aluminium oxide
12156:
12152:
12132:
12068:
12064:
12046:Sulfur monoxide
12036:Strontium oxide
11926:Magnesium oxide
11906:Iodine monoxide
11871:
11867:
11809:Carbon monoxide
11769:Beryllium oxide
11727:
11723:
11710:
11697:
11684:
11671:
11658:
11650:Potassium oxide
11645:
11632:
11619:
11606:
11598:Iodine(I) oxide
11593:
11580:
11567:
11554:
11541:
11533:Copper(I) oxide
11528:
11508:
11504:
11500:
11487:
11483:
11470:
11466:
11453:
11449:
11436:
11432:
11419:
11415:
11402:
11398:
11385:
11381:
11368:
11364:
11351:
11347:
11334:
11330:
11317:
11313:
11300:
11296:
11283:
11279:
11271:Carbon suboxide
11266:
11262:
11249:
11245:
11225:
11220:
11190:
11185:
11175:
11171:
11166:
11161:
11157:
11152:
11147:
11143:
11138:
11133:
11129:
11124:
11119:
11115:
11110:
11105:
11101:
11096:
11091:
11086:
11079:
11073:
11068:
11063:
11059:
11054:
11050:
11037:
11033:
11024:
11016:
11008:
11004:
10996:
10992:
10984:
10980:
10971:
10963:
10950:
10946:
10938:
10930:
10922:
10918:
10910:
10906:
10898:
10885:
10881:
10868:
10864:
10856:
10852:
10843:
10835:
10831:
10823:
10819:
10811:
10803:
10795:
10791:
10783:
10779:
10771:
10767:
10759:
10755:
10747:
10743:
10730:
10726:
10717:
10704:
10696:
10692:
10683:
10674:
10665:
10656:
10648:
10635:
10631:
10622:
10614:
10610:
10602:
10598:
10585:
10581:
10573:
10569:
10561:
10557:
10544:
10540:
10532:
10524:
10520:
10512:
10508:
10500:
10496:
10488:
10480:
10476:
10468:
10464:
10451:
10447:
10439:
10435:
10427:
10423:
10415:
10411:
10403:
10399:
10390:
10382:
10374:
10370:
10362:
10358:
10350:
10337:
10324:
10320:
10312:
10296:
10293:
10292:
10291:
10287:
10284:
10283:
10282:
10280:
10275:
10271:
10263:
10259:
10251:
10243:
10239:
10231:
10223:
10219:
10201:
10197:
10189:
10185:
10177:
10173:
10165:
10161:
10153:
10149:
10141:
10133:
10129:
10121:
10117:
10105:
10100:
10070:
10065:
10059:
10055:
10048:
10044:
10038:
10034:
10030:
10024:
10020:
10016:
10010:
10006:
10002:
9995:
9991:
9984:
9980:
9976:
9970:
9966:
9962:
9958:
9952:
9948:
9944:
9940:
9934:
9930:
9926:
9922:
9915:
9911:
9905:
9901:
9897:
9893:
9887:
9883:
9879:
9873:
9869:
9865:
9859:
9855:
9851:
9834:
9830:
9826:
9820:
9816:
9812:
9806:
9802:
9798:
9792:
9788:
9784:
9777:
9773:
9767:
9763:
9759:
9752:
9748:
9741:
9737:
9733:
9727:
9723:
9719:
9715:
9709:
9705:
9701:
9697:
9691:
9687:
9683:
9677:
9673:
9669:
9663:
9659:
9655:
9649:
9645:
9641:
9635:
9631:
9627:
9621:
9617:
9613:
9607:
9603:
9599:
9592:
9588:
9582:
9578:
9574:
9567:
9563:
9557:
9553:
9549:
9545:
9539:
9535:
9529:
9525:
9509:
9505:
9494:
9490:
9486:
9480:
9476:
9472:
9466:
9462:
9456:
9452:
9446:
9442:
9436:
9432:
9406:
9402:
9398:
9392:
9388:
9384:
9378:
9374:
9370:
9366:
9363:
9359:
9355:
9339:
9335:
9329:
9325:
9319:
9315:
9299:
9295:
9289:
9285:
9279:
9275:
9258:
9254:
9247:
9243:
9227:
9223:
9219:
9213:
9209:
9205:
9196:
9191:
9156:
9148:
9143:
9091:
9058:
8990:
8917:
8876:
8836:
8824:
8793:
8788:
8740:surface tension
8726:Calculation of
8712:
8667:
8665:
8656:
8653:
8631:
8629:Further reading
8626:
8620:
8597:
8567:
8537:
8514:
8493:
8491:
8484:
8449:
8435:
8424:
8409:
8407:
8403:
8397:
8386:
8370:
8344:
8321:
8319:
8312:
8291:
8268:
8247:
8222:
8217:
8208:
8206:
8191:
8190:
8186:
8178:
8174:
8165:
8163:
8146:
8145:
8141:
8133:
8129:
8121:
8117:
8109:
8102:
8071:
8067:
8058:
8056:
8043:
8042:
8038:
8029:
8027:
8022:. August 2003.
8015:
8014:
8010:
8002:
7995:
7987:
7980:
7972:
7968:
7960:
7956:
7948:
7944:
7936:
7932:
7924:
7920:
7911:
7909:
7892:
7891:
7887:
7879:
7875:
7867:
7863:
7855:
7848:
7840:
7836:
7827:
7825:
7812:
7811:
7807:
7798:
7796:
7792:
7777:
7771:
7767:
7759:
7755:
7716:
7712:
7702:
7700:
7689:
7685:
7675:
7673:
7664:
7663:
7659:
7632:
7628:
7597:
7593:
7584:
7582:
7578:
7565:
7561:
7560:
7556:
7548:
7544:
7536:
7532:
7515:
7513:
7506:
7488:
7484:
7473:
7469:
7465:
7458:
7436:
7432:
7408:10.1038/452291a
7379:
7375:
7338:(33): 8710–23.
7328:
7324:
7315:
7313:
7268:
7264:
7211:
7207:
7198:
7196:
7181:10.2172/6642535
7165:
7161:
7153:
7149:
7141:
7137:
7129:
7125:
7117:
7108:
7100:
7096:
7080:
7079:
7070:
7066:
7065:
7061:
7008:
7004:
6996:
6992:
6984:
6980:
6972:
6968:
6937:
6933:
6924:
6922:
6907:
6903:
6872:
6865:
6860:
6856:
6847:
6845:
6841:
6830:
6826:
6825:
6821:
6813:
6809:
6800:
6798:
6794:
6763:
6757:
6753:
6744:
6742:
6697:
6693:
6685:
6681:
6666:
6662:
6654:
6643:
6635:
6631:
6621:
6619:
6615:
6604:
6598:
6597:
6593:
6585:
6578:
6568:
6566:
6553:
6552:
6548:
6539:
6537:
6493:
6486:
6482:
6473:
6471:
6452:
6445:
6414:
6410:
6401:
6399:
6386:
6385:
6376:
6368:
6364:
6356:
6352:
6310:
6306:
6299:
6285:
6278:
6269:
6267:
6256:"Water Density"
6252:
6239:
6230:
6228:
6221:
6203:
6199:
6190:
6188:
6184:
6127:
6121:
6117:
6109:
6100:
6084:
6082:
6073:
6072:
6068:
6060:
6056:
6048:
6044:
6031:
6029:
6025:
6018:
6009:
6005:
5958:
5954:
5899:
5895:
5887:
5883:
5874:
5872:
5865:
5849:. IUPAC. 2009.
5841:
5840:
5836:
5828:
5824:
5816:
5807:
5799:
5795:
5785:
5783:
5770:
5769:
5765:
5757:
5753:
5748:Wayback Machine
5738:
5734:
5726:
5722:
5707:
5696:
5688:
5684:
5676:
5672:
5633:
5629:
5598:
5594:
5585:
5583:
5570:
5569:
5565:
5557:
5553:
5545:
5541:
5533:
5529:
5520:
5518:
5507:
5498:
5490:
5486:
5476:
5474:
5467:10.18434/T4D303
5453:
5449:
5426:
5417:
5408:
5406:
5402:
5363:
5357:
5348:
5338:
5336:
5329:Merriam-Webster
5323:
5322:
5318:
5305:
5303:
5290:
5289:
5285:
5281:
5276:
5271:
5262:
5258:
5253:
5249:
5236:
5232:
5218:
5214:
5209:
5205:
5180:
5176:
5163:
5159:
5151:
5146:
5145:
5144:
5140:
5137:
5136:
5135:
5131:
5129:
5128:
5127:
5123:
5120:
5119:
5118:
5116:
5111:
5109:
5108:
5107:
5105:
5104:
5100:
5092:
5085:
5080:
5076:
5072:
5068:
5063:
4963:
4956:
4953:
4943:, some form of
4934:
4929:Water substance
4917:
4915:
4914:
4913:
4909:
4907:
4906:
4905:
4903:
4896:hydroxylic acid
4865:deuterium oxide
4846:tetrahydropyran
4814:
4772:. Notably, the
4733:Henry Cavendish
4730:
4716:
4714:
4713:
4712:
4708:
4706:
4705:
4704:
4703:
4698:
4696:
4695:
4694:
4692:
4687:
4684:
4683:
4682:
4680:
4674:
4671:
4670:
4669:
4667:
4655:
4652:
4651:
4650:
4648:
4643:
4641:
4640:
4639:
4635:
4633:
4632:
4631:
4630:
4625:
4623:
4622:
4621:
4619:
4612:
4606:
4596:
4593:
4592:
4591:
4589:
4583:
4580:
4579:
4578:
4576:
4571:
4568:
4567:
4566:
4564:
4550:
4547:
4546:
4545:
4541:
4538:
4537:
4536:
4534:
4500:
4498:
4497:
4496:
4494:
4489:
4487:
4486:
4485:
4483:
4478:
4475:
4474:
4473:
4471:
4465:
4462:
4461:
4460:
4458:
4439:oxidation state
4435:
4418:
4407:polysaccharides
4389:
4387:
4386:
4385:
4383:
4372:
4360:
4356:
4336:
4333:
4332:
4331:
4327:
4324:
4323:
4322:
4319:
4302:
4299:
4298:
4297:
4293:
4290:
4289:
4288:
4286:
4271:
4261:
4258:
4257:
4256:
4254:
4248:
4245:
4244:
4243:
4241:
4236:
4233:
4232:
4231:
4227:
4224:
4223:
4222:
4220:
4197:
4194:
4193:
4192:
4186:
4183:
4182:
4181:
4179:
4177:
4170:
4169:
4168:
4162:
4159:
4158:
4157:
4155:
4154:
4153:
4152:
4145:
4144:
4143:
4138:
4136:
4135:
4134:
4132:
4131:
4130:
4129:
4123:
4120:
4119:
4118:
4112:
4109:
4108:
4107:
4105:
4103:
4096:
4095:
4094:
4088:
4085:
4084:
4083:
4081:
4080:
4079:
4078:
4071:
4070:
4069:
4064:
4062:
4061:
4060:
4058:
4057:
4056:
4055:
4049:
4047:
4046:
4045:
4041:
4038:
4037:
4036:
4034:
4027:
4026:
4025:
4019:
4016:
4015:
4014:
4012:
4011:
4010:
4009:
4002:
4001:
4000:
3995:
3993:
3992:
3991:
3989:
3988:
3987:
3986:
3959:
3957:
3956:
3955:
3953:
3948:
3945:
3944:
3943:
3941:
3934:
3933:
3932:
3926:
3923:
3922:
3921:
3919:
3918:
3917:
3916:
3909:
3908:
3907:
3902:
3899:
3898:
3897:
3895:
3894:
3893:
3892:
3884:
3882:
3881:
3880:
3878:
3873:
3870:
3869:
3868:
3866:
3854:
3852:
3851:
3850:
3848:
3843:
3841:
3840:
3839:
3835:
3832:
3831:
3830:
3828:
3821:
3820:
3819:
3813:
3810:
3809:
3808:
3806:
3805:
3804:
3803:
3796:
3795:
3794:
3791:
3790:
3789:
3777:
3775:
3774:
3773:
3771:
3765:
3763:
3762:
3761:
3758:
3739:
3734:
3706:
3702:
3697:
3694:
3693:
3692:
3690:
3687:
3678:
3663:
3660:
3659:
3658:
3656:
3647:
3644:
3643:
3642:
3640:
3634:
3631:
3630:
3629:
3627:
3621:
3618:
3617:
3616:
3614:
3613:coefficient of
3604:
3601:
3600:
3599:
3597:
3591:
3588:
3587:
3586:
3584:
3575:
3572:
3571:
3570:
3568:
3564:
3560:
3534:
3531:
3530:
3529:
3527:
3518:
3516:
3515:
3514:
3513:
3507:
3505:
3504:
3503:
3502:
3496:
3494:
3493:
3492:
3491:
3485:
3483:
3482:
3481:
3480:
3466:
3464:
3463:
3462:
3461:
3434:
3414:
3403:
3400:
3399:
3398:
3394:
3391:
3390:
3389:
3387:
3369:Portland cement
3345:
3319:
3315:
3311:
3310:
3296:
3292:
3286:
3282:
3281:
3280:
3267:
3266:
3262:
3249:
3248:
3244:
3242:
3239:
3238:
3236:
3232:
3208:
3197:
3193:
3192:
3191:
3177:
3173:
3169:
3168:
3164:
3152:
3148:
3142:
3138:
3137:
3136:
3132:
3131:
3129:
3116:
3115:
3111:
3109:
3106:
3105:
3086:
3084:
3083:
3082:
3080:
3075:
3073:
3072:
3071:
3069:
3039:
3035:
3031:
3030:
3016:
3012:
3006:
3002:
3001:
3000:
2987:
2986:
2982:
2980:
2977:
2976:
2960:
2958:
2957:
2956:
2954:
2949:
2947:
2946:
2945:
2941:
2938:
2937:
2936:
2934:
2928:
2925:
2924:
2923:
2921:
2906:self-ionization
2902:
2896:
2894:Self-ionization
2891:
2859:
2853:
2814:, ammonia, and
2792:
2776:Microwave ovens
2740:
2734:
2710:
2704:
2683:
2681:
2680:
2679:
2677:
2669:
2667:
2666:
2665:
2663:
2579:
2573:
2565:
2557:surface tension
2533:
2531:Surface tension
2512:Surface tension
2500:Irving Langmuir
2457:
2438:
2435:
2434:
2433:
2431:
2425:
2422:
2421:
2420:
2418:
2356:
2350:
2341:
2306:reverse osmosis
2282:
2280:
2279:
2278:
2274:
2271:
2270:
2269:
2267:
2262:
2260:
2259:
2258:
2256:
2241:
2236:
2207:
2165:
2119:
2103:
2037:
2026:
2020:
1993:
1991:
1985:
1983:
1977:
1975:
1972:compressibility
1968:
1966:Compressibility
1955:
1949:
1947:Vapour pressure
1931:, and creating
1890:
1884:
1870:
1857:brine rejection
1817:surface density
1808:
1728:
1666:
1613:are known. The
1601:crystals, like
1575:
1516:
1513:
1512:
1511:
1509:
1499:
1493:
1483:
1481:
1480:
1479:
1477:
1472:
1470:
1469:
1468:
1466:
1459:self-ionization
1452:
1450:
1449:
1448:
1446:
1439:
1437:
1436:
1435:
1433:
1390:carbon monoxide
1348:
1344:
1337:
1330:
1325:
1324:
1323: ?)
1314:
1310:
1306:
1302:
1279:
1248:
1238:
1207:
1169:
1168:
1167:
1166:
1159:
1152:
1145:
1141:
1133:
1112:
1107:
1101:
1076:
1070:
1066:
1054:
1051:
1045:
1041:
1038:
1037:Std enthalpy of
1027:
1024:
1017:
1014:
1003:
996:
976:
962:
960:Molecular shape
953:
947:
933:
902:
900:
885:0.6065 W/(m·K)
874:
857:
853:
833:
815:
781:dichloromethane
770:
759:dimethoxyethane
743:tetrahydrofuran
712:
694:
647:
636:
630:
627:
625:
620:
614:
611:
609:
601:
599:
593:
590:
588:
583:
577:
574:
572:
565:Liquid (1 atm,
515:
512:
511:
510:
508:
505:
491:
488:
483:
482:
471:
468:
467:
464:
458:
457:
446:
426:
410:
397:
385:
362:
353:
335:
315:
295:
275:
252:
243:
230:
219:
206:
192:
191:
152:Hydroxylic acid
136:
135:
124:
123:
94:
93:
89:
87:
86: Oxygen, O
83:
42:
35:
28:
23:
22:
15:
12:
11:
5:
17940:
17930:
17929:
17924:
17919:
17914:
17909:
17904:
17899:
17894:
17889:
17884:
17879:
17874:
17869:
17864:
17859:
17857:Forms of water
17854:
17837:
17836:
17831:
17830:
17828:
17827:
17821:
17819:
17813:
17812:
17810:
17809:
17805:
17801:
17797:
17793:
17789:
17785:
17781:
17777:
17773:
17769:
17765:
17761:
17757:
17753:
17749:
17745:
17741:
17737:
17733:
17729:
17725:
17721:
17717:
17713:
17709:
17705:
17701:
17697:
17694:
17690:
17687:
17683:
17679:
17675:
17671:
17667:
17662:
17660:
17654:
17653:
17651:
17650:
17646:
17642:
17638:
17634:
17630:
17626:
17622:
17618:
17614:
17610:
17606:
17602:
17598:
17594:
17590:
17586:
17582:
17578:
17574:
17570:
17566:
17562:
17558:
17554:
17550:
17546:
17542:
17538:
17534:
17530:
17526:
17522:
17518:
17514:
17510:
17506:
17502:
17498:
17494:
17490:
17486:
17482:
17478:
17474:
17470:
17466:
17462:
17458:
17454:
17450:
17446:
17442:
17438:
17433:
17431:
17425:
17424:
17422:
17421:
17411:
17406:
17402:
17398:
17394:
17390:
17386:
17381:
17377:
17373:
17369:
17365:
17353:
17348:
17338:
17333:
17322:
17318:
17307:
17303:
17298:
17294:
17290:
17286:
17282:
17278:
17274:
17270:
17266:
17262:
17258:
17253:
17249:
17245:
17241:
17237:
17232:
17228:
17224:
17219:
17215:
17211:
17206:
17202:
17198:
17193:
17189:
17185:
17181:
17177:
17173:
17169:
17165:
17161:
17157:
17153:
17149:
17145:
17141:
17137:
17133:
17129:
17125:
17121:
17117:
17113:
17109:
17105:
17101:
17097:
17093:
17088:
17086:
17080:
17079:
17077:
17065:
17060:
17055:
17050:
17045:
17040:
17038:
17030:
17029:
17027:
17026:
17021:
17017:
17012:
17008:
17003:
16998:
16989:
16984:
16980:
16976:
16972:
16968:
16962:
16959:
16958:
16956:
16955:
16945:
16939:
16937:
16929:
16928:
16926:
16925:
16921:
16916:
16914:
16906:
16905:
16903:
16902:
16898:
16894:
16890:
16885:
16880:
16878:
16870:
16869:
16867:
16866:
16862:
16858:
16854:
16849:
16844:
16842:
16832:
16831:
16829:
16828:
16823:
16819:
16815:
16811:
16807:
16803:
16799:
16795:
16791:
16787:
16783:
16779:
16775:
16771:
16767:
16763:
16759:
16755:
16751:
16747:
16743:
16739:
16735:
16731:
16727:
16723:
16719:
16715:
16710:
16705:
16703:
16693:
16692:
16690:
16685:
16681:
16677:
16673:
16669:
16665:
16661:
16657:
16653:
16649:
16645:
16641:
16637:
16632:
16628:
16626:
16617:
16613:chalcogenides
16607:
16606:
16604:
16603:
16595:
16590:
16586:
16582:
16577:
16573:
16568:
16565:
16564:
16562:
16561:
16551:
16545:
16543:
16535:
16534:
16532:
16531:
16527:
16522:
16520:
16512:
16511:
16509:
16508:
16504:
16499:
16497:
16489:
16488:
16486:
16485:
16481:
16477:
16473:
16469:
16464:
16462:
16452:
16451:
16449:
16448:
16444:
16440:
16436:
16432:
16428:
16424:
16420:
16416:
16411:
16409:
16399:
16398:
16396:
16395:
16390:
16386:
16382:
16378:
16374:
16370:
16366:
16362:
16358:
16354:
16350:
16346:
16342:
16338:
16334:
16330:
16326:
16322:
16318:
16314:
16310:
16306:
16302:
16298:
16294:
16290:
16286:
16282:
16278:
16273:
16271:
16261:
16260:
16258:
16257:
16253:
16249:
16245:
16241:
16237:
16233:
16229:
16225:
16220:
16218:
16208:
16207:
16205:
16204:
16199:
16195:
16191:
16187:
16183:
16179:
16175:
16171:
16167:
16163:
16159:
16155:
16151:
16147:
16143:
16139:
16135:
16131:
16127:
16123:
16119:
16115:
16111:
16107:
16103:
16099:
16095:
16091:
16087:
16082:
16080:
16071:
16063:
16062:
16059:
16058:
16056:
16055:
16051:
16047:
16043:
16039:
16033:
16031:
16016:
16015:
16013:
16012:
16008:
16003:
16001:
15993:
15992:
15990:
15989:
15985:
15981:
15977:
15973:
15968:
15966:
15956:
15955:
15953:
15952:
15948:
15944:
15940:
15936:
15932:
15928:
15924:
15920:
15916:
15912:
15908:
15904:
15900:
15895:
15893:
15883:
15882:
15880:
15879:
15874:
15870:
15866:
15861:
15859:
15849:
15848:
15846:
15845:
15841:
15837:
15832:
15830:
15820:
15819:
15817:
15816:
15811:
15807:
15803:
15799:
15795:
15791:
15787:
15783:
15779:
15775:
15771:
15767:
15763:
15759:
15755:
15751:
15747:
15743:
15739:
15735:
15731:
15727:
15723:
15719:
15715:
15711:
15707:
15703:
15699:
15694:
15692:
15682:
15681:
15679:
15678:
15673:
15669:
15665:
15661:
15657:
15653:
15647:
15640:
15639:
15637:
15636:
15631:
15626:
15621:
15616:
15611:
15606:
15600:
15598:
15589:
15583:
15582:
15579:
15578:
15576:
15575:
15571:
15567:
15563:
15559:
15555:
15551:
15547:
15541:
15539:
15524:
15523:
15521:
15520:
15516:
15512:
15508:
15504:
15499:
15497:
15489:
15488:
15486:
15485:
15481:
15477:
15473:
15469:
15464:
15462:
15454:
15453:
15451:
15450:
15446:
15442:
15438:
15434:
15429:
15427:
15419:
15418:
15416:
15415:
15411:
15407:
15403:
15399:
15394:
15392:
15384:
15383:
15381:
15380:
15376:
15372:
15368:
15364:
15360:
15356:
15352:
15348:
15344:
15340:
15336:
15332:
15328:
15324:
15320:
15316:
15312:
15308:
15304:
15300:
15296:
15292:
15288:
15284:
15280:
15276:
15272:
15268:
15264:
15260:
15255:
15251:
15246:
15244:
15235:
15227:
15226:
15223:
15222:
15220:
15219:
15215:
15211:
15207:
15203:
15199:
15195:
15191:
15187:
15183:
15178:
15176:
15168:
15167:
15165:
15164:
15159:
15154:
15149:
15144:
15138:
15136:
15129:
15127:earth hydrides
15119:
15118:
15116:
15115:
15110:
15105:
15100:
15095:
15089:
15087:
15079:
15078:
15071:
15070:
15063:
15056:
15048:
15039:
15038:
15033:
15032:
15029:
15026:
15023:
15020:
15017:
15014:
15009:
15004:
14999:
14994:
14991:
14986:
14982:
14973:
14964:
14959:
14954:
14950:
14941:
14932:
14927:
14924:
14919:
14914:
14909:
14904:
14900:
14899:
14894:
14889:
14884:
14879:
14874:
14869:
14864:
14859:
14854:
14849:
14844:
14839:
14834:
14829:
14824:
14815:
14810:
14805:
14800:
14795:
14790:
14785:
14780:
14775:
14770:
14765:
14756:
14751:
14746:
14741:
14737:
14736:
14733:
14730:
14729:
14726:
14723:
14720:
14717:
14714:
14711:
14708:
14705:
14702:
14699:
14696:
14693:
14690:
14687:
14684:
14681:
14678:
14673:
14668:
14662:
14661:
14658:
14655:
14652:
14647:
14642:
14637:
14628:
14623:
14618:
14609:
14604:
14599:
14594:
14589:
14586:
14583:
14580:
14577:
14574:
14571:
14568:
14563:
14558:
14555:
14550:
14545:
14539:
14538:
14535:
14530:
14525:
14520:
14515:
14510:
14505:
14496:
14491:
14486:
14481:
14476:
14471:
14466:
14463:
14458:
14453:
14450:
14445:
14440:
14437:
14434:
14429:
14424:
14419:
14414:
14412:
14407:
14402:
14396:
14395:
14392:
14387:
14384:
14379:
14374:
14369:
14364:
14359:
14354:
14349:
14344:
14339:
14330:
14325:
14320:
14315:
14310:
14305:
14296:
14291:
14286:
14281:
14276:
14267:
14262:
14257:
14248:
14243:
14238:
14229:
14220:
14215:
14210:
14205:
14203:
14198:
14193:
14187:
14186:
14183:
14178:
14173:
14168:
14163:
14158:
14153:
14148:
14143:
14138:
14133:
14128:
14122:
14121:
14118:
14113:
14108:
14103:
14097:
14088:
14083:
14078:
14073:
14068:
14063:
14058:
14053:
14047:
14046:
14043:
14041:
14039:
14037:
14029:
14028:
14025:
14024:
14017:
14016:
14009:
14002:
13994:
13985:
13984:
13974:
13971:
13970:
13968:
13967:
13962:
13957:
13952:
13947:
13942:
13937:
13931:
13929:
13925:
13924:
13922:
13921:
13917:
13907:
13903:
13893:
13889:
13879:
13875:
13865:
13861:
13850:
13848:
13844:
13843:
13841:
13840:
13836:
13832:
13823:
13819:
13815:
13806:
13802:
13798:
13789:
13785:
13781:
13771:
13769:
13765:
13764:
13762:
13761:
13757:
13750:Xenon trioxide
13747:
13743:
13733:
13729:
13719:
13715:
13705:
13701:
13691:
13687:
13677:
13673:
13663:
13659:
13649:
13645:
13635:
13631:
13620:
13618:
13614:
13613:
13611:
13610:
13606:
13602:
13593:
13589:
13585:
13576:
13572:
13568:
13559:
13555:
13551:
13542:
13538:
13534:
13525:
13521:
13517:
13508:
13504:
13500:
13491:
13487:
13483:
13474:
13470:
13466:
13456:
13454:
13450:
13449:
13447:
13446:
13442:
13432:
13428:
13418:
13414:
13404:
13400:
13390:
13386:
13376:
13372:
13362:
13358:
13348:
13344:
13334:
13330:
13320:
13316:
13306:
13302:
13295:Sulfur dioxide
13292:
13288:
13278:
13274:
13264:
13260:
13250:
13246:
13236:
13232:
13222:
13218:
13208:
13204:
13194:
13190:
13180:
13176:
13166:
13162:
13155:Osmium dioxide
13152:
13148:
13138:
13134:
13124:
13120:
13110:
13106:
13096:
13092:
13082:
13078:
13068:
13064:
13057:Iodine dioxide
13054:
13050:
13040:
13036:
13032:
13023:
13019:
13009:
13005:
12995:
12991:
12981:
12977:
12967:
12963:
12953:
12949:
12942:Carbon dioxide
12939:
12935:
12925:
12921:
12911:
12907:
12897:
12893:
12882:
12880:
12876:
12875:
12873:
12872:
12868:
12864:
12855:
12851:
12847:
12838:
12834:
12830:
12821:
12817:
12813:
12804:
12800:
12796:
12787:
12783:
12779:
12770:
12766:
12762:
12753:
12749:
12745:
12736:
12732:
12728:
12722:Scandium oxide
12719:
12715:
12711:
12702:
12698:
12694:
12685:
12681:
12677:
12668:
12664:
12660:
12651:
12647:
12643:
12634:
12630:
12626:
12617:
12613:
12609:
12600:
12596:
12592:
12583:
12579:
12575:
12566:
12562:
12558:
12549:
12545:
12541:
12532:
12528:
12524:
12515:
12511:
12507:
12498:
12494:
12490:
12481:
12477:
12473:
12464:
12460:
12456:
12447:
12443:
12439:
12430:
12426:
12422:
12413:
12409:
12405:
12396:
12392:
12388:
12379:
12375:
12371:
12362:
12358:
12354:
12345:
12341:
12337:
12328:
12324:
12320:
12311:
12307:
12303:
12294:
12290:
12286:
12277:
12273:
12269:
12263:Boron trioxide
12260:
12256:
12252:
12243:
12239:
12235:
12226:
12222:
12218:
12209:
12205:
12201:
12192:
12188:
12184:
12175:
12171:
12167:
12158:
12154:
12150:
12140:
12138:
12134:
12133:
12131:
12130:
12120:
12110:
12100:
12090:
12080:
12070:
12066:
12062:
12053:
12043:
12033:
12023:
12013:
12003:
11993:
11983:
11973:
11963:
11953:
11943:
11933:
11923:
11916:Lead(II) oxide
11913:
11903:
11896:Iron(II) oxide
11893:
11883:
11873:
11869:
11865:
11856:
11846:
11836:
11826:
11816:
11806:
11796:
11786:
11776:
11766:
11756:
11746:
11735:
11733:
11729:
11728:
11726:
11725:
11721:
11712:
11708:
11699:
11695:
11686:
11682:
11673:
11669:
11663:Rubidium oxide
11660:
11656:
11647:
11643:
11634:
11630:
11621:
11617:
11608:
11604:
11595:
11591:
11582:
11578:
11569:
11565:
11556:
11552:
11543:
11539:
11530:
11526:
11516:
11514:
11510:
11509:
11507:
11506:
11502:
11498:
11489:
11485:
11481:
11472:
11468:
11464:
11455:
11451:
11447:
11438:
11434:
11430:
11421:
11417:
11413:
11404:
11400:
11396:
11387:
11383:
11379:
11370:
11366:
11362:
11353:
11349:
11345:
11336:
11332:
11328:
11319:
11315:
11311:
11302:
11298:
11294:
11285:
11281:
11277:
11268:
11264:
11260:
11254:Boron suboxide
11251:
11247:
11243:
11233:
11231:
11227:
11226:
11219:
11218:
11211:
11204:
11196:
11187:
11186:
11184:
11183:
11178:
11173:
11169:
11164:
11159:
11155:
11150:
11145:
11141:
11136:
11131:
11127:
11122:
11117:
11113:
11108:
11103:
11099:
11094:
11089:
11084:
11077:
11071:
11066:
11061:
11057:
11052:
11048:
11044:
11039:
11035:
11031:
11027:
11022:
11018:
11014:
11010:
11006:
11002:
10998:
10994:
10990:
10986:
10982:
10978:
10974:
10969:
10965:
10961:
10957:
10952:
10948:
10944:
10940:
10936:
10932:
10928:
10924:
10920:
10916:
10912:
10908:
10904:
10900:
10896:
10892:
10887:
10883:
10879:
10875:
10870:
10866:
10862:
10858:
10854:
10850:
10846:
10841:
10837:
10833:
10829:
10825:
10821:
10817:
10813:
10809:
10805:
10801:
10797:
10793:
10789:
10785:
10781:
10777:
10773:
10769:
10765:
10761:
10757:
10753:
10749:
10745:
10741:
10737:
10732:
10728:
10724:
10720:
10715:
10711:
10706:
10702:
10698:
10694:
10690:
10686:
10681:
10677:
10672:
10668:
10663:
10659:
10654:
10650:
10646:
10642:
10637:
10633:
10629:
10625:
10620:
10616:
10612:
10608:
10604:
10600:
10596:
10592:
10587:
10583:
10579:
10575:
10571:
10567:
10563:
10559:
10555:
10551:
10546:
10542:
10538:
10534:
10530:
10526:
10522:
10518:
10514:
10510:
10506:
10502:
10498:
10494:
10490:
10486:
10482:
10478:
10474:
10470:
10466:
10462:
10458:
10453:
10449:
10445:
10441:
10437:
10433:
10429:
10425:
10421:
10417:
10413:
10409:
10405:
10401:
10397:
10393:
10388:
10384:
10380:
10376:
10372:
10368:
10364:
10360:
10356:
10352:
10348:
10344:
10339:
10335:
10331:
10326:
10322:
10318:
10314:
10310:
10306:
10301:
10294:
10285:
10277:
10273:
10269:
10265:
10261:
10257:
10253:
10249:
10245:
10241:
10237:
10233:
10229:
10225:
10221:
10217:
10213:
10208:
10203:
10199:
10195:
10191:
10187:
10183:
10179:
10175:
10171:
10167:
10163:
10159:
10155:
10151:
10147:
10143:
10139:
10135:
10131:
10127:
10123:
10119:
10115:
10110:
10107:
10106:
10099:
10098:
10091:
10084:
10076:
10067:
10066:
10064:
10063:
10057:
10052:
10046:
10041:
10036:
10032:
10027:
10022:
10018:
10013:
10008:
10004:
9999:
9993:
9988:
9982:
9978:
9973:
9968:
9964:
9960:
9955:
9950:
9946:
9942:
9937:
9932:
9928:
9924:
9919:
9913:
9908:
9903:
9899:
9895:
9890:
9885:
9881:
9876:
9871:
9867:
9862:
9857:
9853:
9848:
9843:
9838:
9832:
9828:
9823:
9818:
9814:
9809:
9804:
9800:
9795:
9790:
9786:
9781:
9775:
9770:
9765:
9761:
9756:
9750:
9745:
9739:
9735:
9730:
9725:
9721:
9717:
9712:
9707:
9703:
9699:
9694:
9689:
9685:
9680:
9675:
9671:
9666:
9661:
9657:
9652:
9647:
9643:
9638:
9633:
9629:
9624:
9619:
9615:
9610:
9605:
9601:
9596:
9590:
9585:
9580:
9576:
9571:
9565:
9560:
9555:
9551:
9547:
9542:
9537:
9532:
9527:
9522:
9517:
9512:
9507:
9502:
9497:
9492:
9488:
9483:
9478:
9474:
9469:
9464:
9459:
9454:
9449:
9444:
9439:
9434:
9429:
9424:
9419:
9414:
9409:
9404:
9400:
9395:
9390:
9386:
9381:
9376:
9372:
9368:
9361:
9357:
9352:
9347:
9342:
9337:
9332:
9327:
9322:
9317:
9312:
9307:
9302:
9297:
9292:
9287:
9282:
9277:
9272:
9267:
9262:
9256:
9251:
9245:
9240:
9235:
9230:
9225:
9221:
9216:
9211:
9207:
9201:
9198:
9197:
9190:
9189:
9182:
9175:
9167:
9161:
9158:
9157:
9145:
9144:
9142:
9141:
9130:
9119:
9108:
9096:
9093:
9092:
9090:
9089:
9084:
9083:
9082:
9077:
9070:Stratification
9066:
9064:
9060:
9059:
9057:
9056:
9055:
9054:
9049:
9044:
9039:
9031:
9030:
9029:
9024:
9019:
9014:
9009:
8998:
8996:
8992:
8991:
8989:
8988:
8983:
8982:
8981:
8976:
8966:
8961:
8956:
8955:
8954:
8949:
8939:
8934:
8928:
8926:
8919:
8918:
8916:
8915:
8910:
8905:
8900:
8895:
8890:
8884:
8882:
8878:
8877:
8875:
8874:
8873:
8872:
8862:
8857:
8852:
8846:
8844:
8838:
8837:
8827:
8825:
8823:
8822:
8817:
8812:
8807:
8801:
8799:
8795:
8794:
8787:
8786:
8779:
8772:
8764:
8758:
8757:
8748:
8743:
8732:liquid density
8728:vapor pressure
8724:
8710:
8685:
8680:
8674:
8652:
8651:External links
8649:
8648:
8647:
8630:
8627:
8625:
8624:
8618:
8601:
8596:978-3527306732
8595:
8571:
8565:
8541:
8536:978-0471927266
8535:
8518:
8513:978-0321775658
8512:
8499:
8483:978-0849304842
8482:
8463:
8454:
8452:on 2011-07-26.
8433:
8415:
8395:
8374:
8368:
8348:
8342:
8336:. Read Books.
8327:
8310:
8295:
8289:
8272:
8266:
8251:
8246:978-1461544852
8245:
8223:
8221:
8218:
8216:
8215:
8184:
8172:
8139:
8127:
8115:
8100:
8065:
8036:
8008:
8006:, p. 601.
7993:
7991:, p. 866.
7978:
7976:, p. 275.
7966:
7964:, p. 981.
7954:
7952:, p. 862.
7942:
7940:, p. 338.
7930:
7918:
7885:
7883:, p. 171.
7873:
7871:, p. 984.
7861:
7859:, p. 654.
7846:
7844:, p. 659.
7834:
7805:
7788:(2): 175–188.
7765:
7753:
7710:
7683:
7657:
7626:
7591:
7554:
7552:, p. 106.
7542:
7540:, p. 105.
7530:
7504:
7482:
7471:
7467:
7463:
7456:
7430:
7373:
7332:Applied Optics
7322:
7282:(16): 167802.
7262:
7205:
7159:
7157:, p. 518.
7147:
7145:, p. 627.
7135:
7123:
7106:
7094:
7059:
7002:
6990:
6978:
6976:, p. 393.
6966:
6931:
6901:
6882:(1): E16–E19.
6863:
6854:
6819:
6807:
6751:
6691:
6679:
6660:
6657:on 2008-03-09.
6629:
6591:
6576:
6546:
6491:
6480:
6443:
6408:
6374:
6372:, p. 493.
6362:
6360:, p. 624.
6350:
6304:
6297:
6276:
6237:
6219:
6197:
6115:
6113:, p. 625.
6098:
6066:
6054:
6042:
6003:
5968:(7): 797–803.
5952:
5893:
5881:
5863:
5834:
5822:
5805:
5793:
5763:
5761:, p. 620.
5751:
5732:
5720:
5694:
5682:
5670:
5627:
5608:(6): 690–695.
5592:
5578:. 2015-08-09.
5563:
5551:
5539:
5527:
5496:
5484:
5447:
5436:(4): 301–309.
5415:
5346:
5316:
5282:
5280:
5277:
5275:
5272:
5270:
5269:
5256:
5247:
5230:
5212:
5203:
5174:
5157:
5147:
5138:
5130:
5121:
5110:
5098:
5083:
5078:
5069:
5067:
5064:
5062:
5061:
5056:
5051:
5046:
5041:
5036:
5031:
5026:
5021:
5016:
5011:
5006:
5001:
4996:
4994:Fluid dynamics
4991:
4986:
4981:
4976:
4970:
4969:
4968:
4952:
4949:
4932:
4916:
4908:
4853:hydrogen oxide
4830:parent hydride
4813:
4810:
4729:
4726:
4721:
4720:
4715:
4707:
4697:
4685:
4672:
4660:
4659:
4653:
4642:
4634:
4624:
4608:Main article:
4605:
4602:
4601:
4600:
4594:
4581:
4569:
4548:
4539:
4505:
4504:
4499:
4488:
4476:
4463:
4451:alkaline earth
4434:
4431:
4423:E2 elimination
4419:2 substitution
4416:
4399:saponification
4388:
4371:
4368:
4358:
4354:
4334:
4325:
4308:perrhenic acid
4300:
4291:
4270:
4267:
4266:
4265:
4259:
4246:
4234:
4225:
4202:
4201:
4195:
4184:
4160:
4137:
4127:
4121:
4110:
4086:
4063:
4053:
4048:
4039:
4017:
3994:
3964:
3963:
3958:
3946:
3924:
3900:
3883:
3871:
3859:
3858:
3853:
3842:
3833:
3811:
3776:
3764:
3755:Brønsted-Lowry
3738:
3735:
3733:
3730:
3695:
3686:
3683:
3661:
3645:
3632:
3619:
3611:self-diffusion
3602:
3589:
3573:
3559:of 4500 days;
3532:
3517:
3512:in 0.04%, and
3506:
3495:
3484:
3465:
3433:
3430:
3413:
3410:
3401:
3392:
3344:
3341:
3329:
3322:
3318:
3314:
3309:
3306:
3299:
3295:
3289:
3285:
3279:
3276:
3270:
3265:
3261:
3255:
3252:
3247:
3234:
3230:
3227:
3226:
3211:
3205:
3200:
3196:
3190:
3180:
3176:
3172:
3167:
3163:
3155:
3151:
3145:
3141:
3135:
3128:
3122:
3119:
3114:
3085:
3074:
3049:
3042:
3038:
3034:
3029:
3026:
3019:
3015:
3009:
3005:
2999:
2996:
2990:
2985:
2965:
2964:
2959:
2948:
2939:
2926:
2898:Main article:
2895:
2892:
2890:
2887:
2852:
2849:
2808:hydrogen bonds
2800:hydrogen bonds
2791:
2788:
2768:photoreceptors
2760:infrared light
2736:Main article:
2733:
2730:
2706:Main article:
2703:
2700:
2682:
2668:
2575:Main article:
2572:
2569:
2564:
2561:
2532:
2529:
2456:
2453:
2436:
2423:
2391:polar molecule
2349:
2346:
2310:ultra-filtered
2281:
2272:
2261:
2253:autoionization
2240:
2237:
2235:
2232:
2206:
2203:
2200:
2199:
2196:
2193:
2189:
2188:
2185:
2182:
2178:
2177:
2174:
2171:
2170:, and ice III
2163:
2159:
2158:
2155:
2152:
2145:
2144:
2143:−17.0 °C
2141:
2138:
2131:
2130:
2127:
2124:
2117:
2113:
2112:
2109:
2106:
2101:
2098:liquid water,
2095:
2094:
2091:
2088:
2035:
2022:Main article:
2019:
2016:
1967:
1964:
1951:Main article:
1948:
1945:
1921:vapor pressure
1880:Main article:
1869:
1866:
1807:
1804:
1727:
1724:
1665:
1662:
1574:
1571:
1514:
1492:
1489:
1482:
1471:
1451:
1438:
1397:hydrogen bonds
1380:, liquid, and
1346:
1335:
1332:
1331:
1326:
1304:
1303:
1299:standard state
1296:
1293:
1292:
1286:
1285:
1281:
1280:
1278:
1277:
1272:
1267:
1262:
1257:
1251:
1249:
1243:
1240:
1239:
1237:
1236:
1231:
1226:
1221:
1216:
1210:
1208:
1202:
1199:
1198:
1194:
1193:
1188:
1181:
1180:
1179:Non-flammable
1177:
1171:
1170:
1160:
1153:
1146:
1131:
1130:
1129:
1128:
1126:
1117:
1116:
1102:
1099:
1096:
1095:
1086:
1085:
1081:
1080:
1077:
1068:
1062:
1059:
1058:
1055:
1049:
1043:
1035:
1032:
1031:
1028:
1022:
1011:
1008:
1007:
1004:
992:
989:
988:
984:
983:
977:
972:
969:
968:
963:
958:
955:
954:
951:
948:
943:
940:
939:
934:
929:
926:
925:
921:
920:
913:
907:
906:
903:
898:
890:
887:
886:
883:
877:
876:
872:
866:
864:Conjugate base
860:
859:
855:
851:
845:
843:Conjugate acid
839:
838:
835:
831:
821:
820:
817:
813:
803:
802:
799:
797:Vapor pressure
793:
792:
713:Miscible with
689:hydrocarbons,
675:
669:
668:
665:
659:
658:
655:
649:
648:
646:
645:
642:
639:
633:(96) g/mL
623:
617:(83) g/mL
607:
596:(84) g/mL
586:
580:(84) g/mL
570:
562:
560:
554:
553:
550:
544:
543:
536:
532:
531:
528:
522:
521:
513:
506:
501:
498:
497:
493:
492:
490:
489:
486:
478:
477:
476:
473:
472:
470:
469:
465:
462:
461:
453:
452:
451:
448:
447:
445:
444:
436:
434:
428:
427:
425:
424:
420:
418:
412:
411:
409:
408:
400:
398:
390:
387:
386:
384:
383:
375:
373:
367:
366:
363:
358:
355:
354:
352:
351:
347:
345:
337:
336:
334:
333:
325:
323:
317:
316:
314:
313:
305:
303:
297:
296:
294:
293:
285:
283:
277:
276:
274:
273:
265:
263:
257:
256:
253:
248:
245:
244:
242:
241:
233:
231:
224:
221:
220:
218:
217:
209:
207:
202:
199:
198:
194:
193:
190:
189:
188:Neutral liquid
186:
183:
180:
177:
174:
173:Hydroxoic acid
171:
168:
165:
162:
159:
153:
150:
147:
146:Hydrogen oxide
143:
142:
138:
137:
133:
132:
126:
125:
121:
120:
114:
113:
109:
108:
100:
99:
96:
95:
88:
82:
81:
71:
56:
55:
26:
18:Water molecule
9:
6:
4:
3:
2:
17939:
17928:
17925:
17923:
17922:Heat transfer
17920:
17918:
17915:
17913:
17910:
17908:
17905:
17903:
17900:
17898:
17895:
17893:
17890:
17888:
17885:
17883:
17880:
17878:
17875:
17873:
17870:
17868:
17865:
17863:
17860:
17858:
17855:
17853:
17850:
17849:
17847:
17826:
17823:
17822:
17820:
17818:
17814:
17808:
17802:
17800:
17794:
17792:
17786:
17784:
17778:
17776:
17770:
17768:
17762:
17760:
17754:
17752:
17746:
17744:
17738:
17736:
17730:
17728:
17722:
17720:
17714:
17712:
17706:
17704:
17698:
17688:
17686:
17680:
17678:
17672:
17670:
17664:
17663:
17661:
17659:
17655:
17649:
17643:
17641:
17635:
17633:
17627:
17625:
17619:
17617:
17611:
17609:
17603:
17601:
17595:
17593:
17587:
17585:
17579:
17577:
17571:
17569:
17563:
17561:
17555:
17553:
17547:
17545:
17539:
17537:
17531:
17529:
17523:
17521:
17515:
17513:
17507:
17505:
17499:
17497:
17491:
17489:
17483:
17481:
17475:
17473:
17467:
17465:
17459:
17457:
17451:
17449:
17443:
17441:
17435:
17434:
17432:
17430:
17426:
17419:
17415:
17414:
17407:
17405:
17399:
17397:
17387:
17385:
17382:
17380:
17374:
17372:
17366:
17363:
17359:
17358:
17354:
17352:
17349:
17346:
17342:
17341:
17334:
17332:
17330:
17325:
17319:
17317:
17315:
17310:
17304:
17302:
17299:
17297:
17291:
17289:
17283:
17281:
17275:
17273:
17267:
17265:
17259:
17257:
17254:
17252:
17246:
17244:
17238:
17236:
17233:
17231:
17225:
17223:
17220:
17218:
17212:
17210:
17207:
17205:
17199:
17197:
17194:
17192:
17186:
17184:
17178:
17176:
17170:
17168:
17162:
17160:
17154:
17152:
17146:
17144:
17138:
17136:
17130:
17128:
17122:
17120:
17114:
17112:
17106:
17104:
17098:
17096:
17090:
17089:
17087:
17085:
17081:
17075:
17071:
17070:
17066:
17064:
17061:
17059:
17056:
17054:
17051:
17049:
17046:
17044:
17041:
17039:
17037:
17031:
17025:
17018:
17016:
17009:
17007:
17004:
17002:
16999:
16997:
16993:
16985:
16983:
16977:
16975:
16969:
16967:
16964:
16963:
16953:
16949:
16948:
16941:
16940:
16938:
16934:
16924:
16918:
16917:
16915:
16911:
16901:
16891:
16889:
16882:
16881:
16879:
16875:
16865:
16855:
16853:
16846:
16845:
16843:
16841:
16837:
16827:
16824:
16822:
16812:
16810:
16800:
16798:
16788:
16786:
16776:
16774:
16764:
16762:
16752:
16750:
16740:
16738:
16728:
16726:
16716:
16714:
16707:
16706:
16704:
16702:
16698:
16689:
16686:
16684:
16674:
16672:
16662:
16660:
16650:
16648:
16638:
16636:
16629:
16627:
16625:
16621:
16618:
16616:
16608:
16602:
16598:
16591:
16589:
16583:
16581:
16578:
16576:
16570:
16569:
16559:
16555:
16554:
16547:
16546:
16544:
16540:
16530:
16524:
16523:
16521:
16517:
16507:
16501:
16500:
16498:
16494:
16484:
16474:
16472:
16466:
16465:
16463:
16461:
16457:
16447:
16437:
16435:
16425:
16423:
16413:
16412:
16410:
16408:
16404:
16394:
16391:
16389:
16379:
16377:
16367:
16365:
16355:
16353:
16343:
16341:
16331:
16329:
16319:
16317:
16307:
16305:
16295:
16293:
16283:
16281:
16275:
16274:
16272:
16270:
16266:
16256:
16246:
16244:
16234:
16232:
16222:
16221:
16219:
16217:
16213:
16203:
16200:
16198:
16188:
16186:
16176:
16174:
16164:
16162:
16152:
16150:
16140:
16138:
16128:
16126:
16116:
16114:
16104:
16102:
16092:
16090:
16084:
16083:
16081:
16079:
16075:
16072:
16070:
16064:
16054:
16048:
16046:
16040:
16038:
16035:
16034:
16032:
16029:
16025:
16021:
16011:
16005:
16004:
16002:
15998:
15988:
15978:
15976:
15970:
15969:
15967:
15965:
15961:
15951:
15941:
15939:
15929:
15927:
15917:
15915:
15905:
15903:
15897:
15896:
15894:
15892:
15888:
15878:
15875:
15873:
15863:
15862:
15860:
15858:
15854:
15844:
15834:
15833:
15831:
15829:
15825:
15815:
15812:
15810:
15800:
15798:
15788:
15786:
15776:
15774:
15764:
15762:
15752:
15750:
15740:
15738:
15728:
15726:
15716:
15714:
15704:
15702:
15696:
15695:
15693:
15691:
15687:
15677:
15670:
15668:
15662:
15660:
15654:
15652:
15649:
15648:
15645:
15635:
15632:
15630:
15627:
15625:
15622:
15620:
15617:
15615:
15612:
15610:
15607:
15605:
15602:
15601:
15599:
15597:
15593:
15590:
15588:
15584:
15574:
15568:
15566:
15556:
15554:
15548:
15546:
15543:
15542:
15540:
15537:
15533:
15529:
15519:
15509:
15507:
15501:
15500:
15498:
15494:
15484:
15474:
15472:
15466:
15465:
15463:
15459:
15449:
15439:
15437:
15431:
15430:
15428:
15424:
15414:
15404:
15402:
15396:
15395:
15393:
15389:
15379:
15369:
15367:
15357:
15355:
15345:
15343:
15333:
15331:
15321:
15319:
15309:
15307:
15297:
15295:
15285:
15283:
15273:
15271:
15261:
15259:
15256:
15254:
15248:
15247:
15245:
15243:
15239:
15236:
15234:
15228:
15218:
15212:
15210:
15204:
15202:
15196:
15194:
15188:
15186:
15180:
15179:
15177:
15173:
15163:
15160:
15158:
15155:
15153:
15150:
15148:
15145:
15143:
15140:
15139:
15137:
15133:
15130:
15128:
15120:
15114:
15111:
15109:
15106:
15104:
15101:
15099:
15096:
15094:
15091:
15090:
15088:
15086:
15083:Alkali metal
15080:
15076:
15069:
15064:
15062:
15057:
15055:
15050:
15049:
15046:
15030:
15027:
15024:
15021:
15018:
15015:
15013:
15005:
15003:
14995:
14992:
14990:
14977:
14968:
14960:
14958:
14945:
14936:
14928:
14925:
14923:
14915:
14913:
14905:
14902:
14901:
14898:
14890:
14888:
14880:
14878:
14870:
14868:
14860:
14858:
14850:
14848:
14840:
14838:
14830:
14828:
14819:
14811:
14809:
14801:
14799:
14791:
14789:
14781:
14779:
14771:
14769:
14760:
14752:
14750:
14742:
14739:
14738:
14731:
14727:
14724:
14721:
14718:
14715:
14712:
14709:
14706:
14703:
14700:
14697:
14694:
14691:
14688:
14685:
14682:
14679:
14677:
14669:
14667:
14664:
14663:
14659:
14656:
14653:
14651:
14643:
14641:
14632:
14624:
14622:
14613:
14610:
14608:
14600:
14598:
14590:
14587:
14584:
14581:
14578:
14575:
14572:
14569:
14567:
14559:
14556:
14554:
14546:
14544:
14541:
14540:
14536:
14534:
14531:
14529:
14521:
14519:
14511:
14509:
14500:
14492:
14490:
14482:
14480:
14472:
14470:
14467:
14464:
14462:
14454:
14451:
14449:
14441:
14438:
14435:
14433:
14425:
14423:
14415:
14413:
14411:
14403:
14401:
14398:
14397:
14393:
14391:
14388:
14385:
14383:
14375:
14373:
14365:
14363:
14355:
14353:
14345:
14343:
14334:
14331:
14329:
14321:
14319:
14311:
14309:
14300:
14292:
14290:
14282:
14280:
14271:
14263:
14261:
14252:
14244:
14242:
14233:
14224:
14216:
14214:
14206:
14204:
14202:
14194:
14192:
14189:
14188:
14184:
14182:
14179:
14177:
14169:
14167:
14159:
14157:
14149:
14147:
14139:
14137:
14129:
14127:
14124:
14123:
14119:
14117:
14114:
14112:
14104:
14102:
14092:
14084:
14082:
14074:
14072:
14064:
14062:
14054:
14052:
14049:
14048:
14044:
14038:
14036:
14033:
14032:
14026:
14022:
14015:
14010:
14008:
14003:
14001:
13996:
13995:
13992:
13982:
13978:
13972:
13966:
13963:
13961:
13958:
13956:
13953:
13951:
13948:
13946:
13943:
13941:
13938:
13936:
13933:
13932:
13930:
13926:
13915:
13911:
13908:
13901:
13897:
13894:
13887:
13883:
13880:
13873:
13869:
13866:
13859:
13855:
13852:
13851:
13849:
13845:
13831:
13827:
13824:
13814:
13810:
13807:
13797:
13793:
13790:
13780:
13776:
13773:
13772:
13770:
13766:
13755:
13751:
13748:
13741:
13737:
13734:
13727:
13723:
13720:
13713:
13709:
13706:
13699:
13695:
13692:
13685:
13681:
13678:
13671:
13667:
13664:
13657:
13653:
13650:
13643:
13639:
13636:
13629:
13625:
13622:
13621:
13619:
13615:
13601:
13597:
13594:
13584:
13580:
13577:
13567:
13563:
13560:
13550:
13546:
13543:
13533:
13529:
13526:
13516:
13512:
13509:
13499:
13495:
13492:
13482:
13478:
13475:
13465:
13461:
13458:
13457:
13455:
13451:
13440:
13436:
13433:
13426:
13422:
13419:
13412:
13408:
13405:
13398:
13394:
13391:
13384:
13380:
13377:
13370:
13366:
13363:
13356:
13352:
13349:
13342:
13338:
13335:
13328:
13324:
13321:
13314:
13310:
13307:
13300:
13296:
13293:
13286:
13282:
13279:
13272:
13268:
13265:
13258:
13254:
13251:
13244:
13240:
13237:
13230:
13226:
13223:
13216:
13212:
13209:
13202:
13198:
13195:
13188:
13184:
13181:
13174:
13170:
13167:
13160:
13156:
13153:
13146:
13142:
13139:
13132:
13128:
13125:
13118:
13114:
13111:
13104:
13100:
13097:
13090:
13086:
13083:
13076:
13072:
13069:
13062:
13058:
13055:
13048:
13044:
13041:
13031:
13027:
13024:
13017:
13013:
13010:
13003:
12999:
12996:
12989:
12985:
12982:
12975:
12971:
12968:
12961:
12957:
12954:
12947:
12943:
12940:
12933:
12929:
12926:
12919:
12915:
12912:
12905:
12901:
12898:
12891:
12887:
12884:
12883:
12881:
12877:
12863:
12859:
12856:
12846:
12842:
12839:
12829:
12825:
12822:
12812:
12808:
12805:
12795:
12791:
12788:
12778:
12774:
12771:
12761:
12757:
12754:
12744:
12740:
12737:
12727:
12723:
12720:
12710:
12706:
12703:
12693:
12689:
12686:
12676:
12672:
12669:
12659:
12655:
12652:
12642:
12638:
12635:
12625:
12621:
12618:
12608:
12604:
12601:
12591:
12587:
12584:
12574:
12570:
12567:
12557:
12553:
12550:
12540:
12536:
12533:
12523:
12519:
12516:
12506:
12502:
12499:
12489:
12485:
12482:
12472:
12468:
12465:
12455:
12451:
12448:
12438:
12434:
12431:
12421:
12417:
12414:
12404:
12400:
12397:
12387:
12383:
12380:
12370:
12366:
12363:
12353:
12349:
12346:
12336:
12332:
12329:
12319:
12315:
12312:
12302:
12298:
12295:
12285:
12281:
12278:
12268:
12264:
12261:
12251:
12247:
12244:
12234:
12230:
12227:
12217:
12213:
12210:
12200:
12196:
12193:
12183:
12179:
12176:
12166:
12162:
12159:
12149:
12145:
12142:
12141:
12139:
12135:
12128:
12124:
12121:
12118:
12114:
12111:
12108:
12104:
12101:
12098:
12094:
12091:
12088:
12084:
12083:Tin(II) oxide
12081:
12078:
12074:
12071:
12061:
12057:
12054:
12051:
12047:
12044:
12041:
12037:
12034:
12031:
12027:
12024:
12021:
12017:
12014:
12011:
12007:
12004:
12001:
11997:
11994:
11991:
11987:
11984:
11981:
11977:
11974:
11971:
11967:
11964:
11961:
11957:
11954:
11951:
11947:
11944:
11941:
11937:
11934:
11931:
11927:
11924:
11921:
11917:
11914:
11911:
11907:
11904:
11901:
11897:
11894:
11891:
11887:
11884:
11881:
11877:
11874:
11864:
11860:
11857:
11854:
11850:
11847:
11844:
11840:
11837:
11834:
11830:
11827:
11824:
11820:
11817:
11814:
11810:
11807:
11804:
11800:
11799:Calcium oxide
11797:
11794:
11790:
11789:Cadmium oxide
11787:
11784:
11780:
11777:
11774:
11770:
11767:
11764:
11760:
11757:
11754:
11750:
11747:
11744:
11740:
11737:
11736:
11734:
11730:
11720:
11716:
11713:
11707:
11703:
11700:
11694:
11690:
11687:
11681:
11677:
11674:
11668:
11664:
11661:
11655:
11651:
11648:
11642:
11638:
11637:Nitrous oxide
11635:
11629:
11625:
11622:
11616:
11612:
11611:Lithium oxide
11609:
11603:
11599:
11596:
11590:
11586:
11583:
11577:
11573:
11570:
11564:
11560:
11557:
11551:
11547:
11544:
11538:
11534:
11531:
11525:
11521:
11518:
11517:
11515:
11511:
11497:
11493:
11490:
11480:
11476:
11473:
11463:
11459:
11456:
11446:
11442:
11439:
11429:
11425:
11422:
11412:
11408:
11405:
11395:
11391:
11388:
11378:
11374:
11371:
11361:
11357:
11354:
11344:
11340:
11337:
11327:
11323:
11320:
11310:
11306:
11303:
11293:
11289:
11286:
11276:
11272:
11269:
11259:
11255:
11252:
11242:
11238:
11235:
11234:
11232:
11228:
11224:
11217:
11212:
11210:
11205:
11203:
11198:
11197:
11194:
11182:
11179:
11176:
11165:
11162:
11151:
11148:
11137:
11134:
11123:
11120:
11109:
11106:
11095:
11092:
11085:
11082:
11078:
11075:
11067:
11064:
11053:
11051:
11045:
11043:
11040:
11038:
11028:
11026:
11019:
11017:
11011:
11009:
10999:
10997:
10987:
10985:
10975:
10973:
10966:
10964:
10958:
10956:
10953:
10951:
10941:
10939:
10933:
10931:
10925:
10923:
10913:
10911:
10901:
10899:
10893:
10891:
10888:
10886:
10876:
10874:
10871:
10869:
10859:
10857:
10847:
10845:
10838:
10836:
10826:
10824:
10814:
10812:
10806:
10804:
10798:
10796:
10786:
10784:
10774:
10772:
10762:
10760:
10750:
10748:
10738:
10736:
10733:
10731:
10721:
10719:
10712:
10710:
10707:
10705:
10699:
10697:
10687:
10685:
10678:
10676:
10669:
10667:
10660:
10658:
10651:
10649:
10643:
10641:
10638:
10636:
10626:
10624:
10617:
10615:
10605:
10603:
10593:
10591:
10588:
10586:
10576:
10574:
10564:
10562:
10552:
10550:
10547:
10545:
10535:
10533:
10527:
10525:
10515:
10513:
10503:
10501:
10491:
10489:
10483:
10481:
10471:
10469:
10459:
10457:
10454:
10452:
10442:
10440:
10430:
10428:
10418:
10416:
10406:
10404:
10394:
10392:
10385:
10383:
10377:
10375:
10365:
10363:
10353:
10351:
10345:
10343:
10340:
10338:
10332:
10330:
10327:
10325:
10315:
10313:
10307:
10305:
10302:
10300:
10278:
10276:
10266:
10264:
10254:
10252:
10246:
10244:
10234:
10232:
10226:
10224:
10214:
10212:
10209:
10207:
10204:
10202:
10192:
10190:
10180:
10178:
10168:
10166:
10156:
10154:
10144:
10142:
10136:
10134:
10124:
10122:
10112:
10111:
10108:
10104:
10097:
10092:
10090:
10085:
10083:
10078:
10077:
10074:
10062:
10053:
10051:
10042:
10040:
10028:
10026:
10014:
10012:
10000:
9998:
9989:
9987:
9974:
9972:
9956:
9954:
9938:
9936:
9920:
9918:
9909:
9907:
9891:
9889:
9877:
9875:
9863:
9861:
9849:
9847:
9844:
9842:
9839:
9837:
9824:
9822:
9810:
9808:
9796:
9794:
9782:
9780:
9771:
9769:
9757:
9755:
9746:
9744:
9731:
9729:
9713:
9711:
9695:
9693:
9681:
9679:
9667:
9665:
9653:
9651:
9639:
9637:
9625:
9623:
9611:
9609:
9597:
9595:
9586:
9584:
9572:
9570:
9561:
9559:
9543:
9541:
9533:
9531:
9523:
9521:
9518:
9516:
9513:
9511:
9503:
9501:
9498:
9496:
9484:
9482:
9470:
9468:
9460:
9458:
9450:
9448:
9440:
9438:
9430:
9428:
9425:
9423:
9420:
9418:
9415:
9413:
9410:
9408:
9396:
9394:
9382:
9380:
9353:
9351:
9348:
9346:
9343:
9341:
9333:
9331:
9323:
9321:
9313:
9311:
9308:
9306:
9303:
9301:
9293:
9291:
9283:
9281:
9273:
9271:
9268:
9266:
9263:
9261:
9252:
9250:
9241:
9239:
9236:
9234:
9231:
9229:
9217:
9215:
9203:
9202:
9199:
9195:
9188:
9183:
9181:
9176:
9174:
9169:
9168:
9165:
9159:
9152:
9140:
9136:
9131:
9129:
9120:
9118:
9109:
9107:
9103:
9098:
9097:
9094:
9088:
9085:
9081:
9078:
9076:
9073:
9072:
9071:
9068:
9067:
9065:
9061:
9053:
9050:
9048:
9045:
9043:
9040:
9038:
9035:
9034:
9032:
9028:
9025:
9023:
9022:Hycean planet
9020:
9018:
9015:
9013:
9010:
9008:
9005:
9004:
9003:
9000:
8999:
8997:
8993:
8987:
8984:
8980:
8977:
8975:
8972:
8971:
8970:
8967:
8965:
8962:
8960:
8957:
8953:
8950:
8948:
8945:
8944:
8943:
8940:
8938:
8935:
8933:
8930:
8929:
8927:
8925:
8920:
8914:
8911:
8909:
8906:
8904:
8901:
8899:
8896:
8894:
8891:
8889:
8886:
8885:
8883:
8879:
8871:
8868:
8867:
8866:
8863:
8861:
8858:
8856:
8853:
8851:
8848:
8847:
8845:
8843:
8839:
8833:Water droplet
8831:
8821:
8818:
8816:
8813:
8811:
8808:
8806:
8803:
8802:
8800:
8796:
8792:
8785:
8780:
8778:
8773:
8771:
8766:
8765:
8762:
8756:
8752:
8749:
8747:
8744:
8741:
8737:
8733:
8729:
8725:
8721:
8717:
8713:
8711:9781119300755
8707:
8703:
8699:
8695:
8691:
8686:
8684:
8681:
8678:
8675:
8664:. May 2, 2016
8663:
8659:
8655:
8654:
8645:
8641:
8637:
8633:
8632:
8621:
8615:
8611:
8607:
8602:
8598:
8592:
8588:
8584:
8580:
8579:
8572:
8568:
8562:
8558:
8553:
8552:
8546:
8542:
8538:
8532:
8527:
8526:
8519:
8515:
8509:
8505:
8500:
8489:
8485:
8479:
8475:
8471:
8470:
8464:
8460:
8455:
8448:
8444:
8440:
8436:
8430:
8423:
8422:
8416:
8402:
8398:
8392:
8385:
8384:
8379:
8375:
8371:
8365:
8361:
8357:
8353:
8349:
8345:
8339:
8335:
8334:
8328:
8317:
8313:
8307:
8303:
8302:
8296:
8292:
8286:
8281:
8280:
8273:
8269:
8263:
8259:
8258:
8252:
8248:
8242:
8238:
8234:
8230:
8229:Water Quality
8225:
8224:
8204:
8200:
8199:
8194:
8188:
8181:
8176:
8161:
8157:
8153:
8149:
8143:
8137:, p. 99.
8136:
8131:
8125:, p. 85.
8124:
8119:
8113:, p. 34.
8112:
8107:
8105:
8096:
8092:
8088:
8084:
8080:
8076:
8069:
8054:
8050:
8046:
8040:
8025:
8021:
8018:
8012:
8005:
8000:
7998:
7990:
7985:
7983:
7975:
7970:
7963:
7958:
7951:
7946:
7939:
7934:
7927:
7922:
7907:
7903:
7899:
7895:
7889:
7882:
7877:
7870:
7865:
7858:
7853:
7851:
7843:
7838:
7823:
7819:
7815:
7809:
7791:
7787:
7783:
7776:
7769:
7763:, p. 29.
7762:
7757:
7749:
7745:
7741:
7737:
7733:
7729:
7725:
7721:
7714:
7698:
7694:
7687:
7671:
7667:
7661:
7653:
7649:
7645:
7641:
7637:
7630:
7622:
7618:
7614:
7610:
7606:
7602:
7601:J. Chem. Phys
7595:
7577:
7573:
7572:
7564:
7558:
7551:
7546:
7539:
7534:
7527:
7526:
7511:
7507:
7501:
7497:
7493:
7486:
7479:
7477:
7474:O are called
7459:
7457:9780060936778
7453:
7449:
7444:
7443:
7434:
7426:
7422:
7418:
7414:
7409:
7404:
7400:
7396:
7392:
7388:
7384:
7377:
7369:
7365:
7361:
7357:
7353:
7349:
7345:
7341:
7337:
7333:
7326:
7311:
7307:
7303:
7298:
7293:
7289:
7285:
7281:
7277:
7273:
7266:
7258:
7254:
7250:
7246:
7241:
7236:
7232:
7228:
7224:
7220:
7216:
7209:
7194:
7190:
7186:
7182:
7178:
7174:
7170:
7163:
7156:
7151:
7144:
7139:
7132:
7127:
7121:, p. 46.
7120:
7115:
7113:
7111:
7103:
7098:
7090:
7084:
7076:
7069:
7063:
7055:
7051:
7046:
7041:
7037:
7033:
7029:
7025:
7021:
7017:
7013:
7006:
7000:, p. 47.
6999:
6994:
6987:
6982:
6975:
6970:
6962:
6958:
6954:
6950:
6946:
6942:
6935:
6921:on 2009-05-10
6920:
6916:
6912:
6905:
6897:
6893:
6889:
6885:
6881:
6877:
6870:
6868:
6858:
6840:
6836:
6829:
6823:
6817:, p. 27.
6816:
6811:
6793:
6789:
6785:
6781:
6777:
6773:
6769:
6768:Physics Today
6762:
6755:
6740:
6736:
6732:
6727:
6722:
6718:
6714:
6710:
6706:
6702:
6695:
6688:
6683:
6675:
6671:
6664:
6653:
6649:
6642:
6641:
6633:
6614:
6610:
6603:
6602:
6595:
6588:
6583:
6581:
6564:
6560:
6556:
6550:
6535:
6531:
6527:
6523:
6519:
6515:
6511:
6508:(3): 033101.
6507:
6503:
6499:
6497:
6484:
6469:
6465:
6461:
6457:
6450:
6448:
6439:
6435:
6431:
6427:
6423:
6419:
6412:
6397:
6393:
6389:
6383:
6381:
6379:
6371:
6366:
6359:
6354:
6346:
6342:
6338:
6334:
6330:
6326:
6322:
6318:
6314:
6308:
6300:
6294:
6290:
6283:
6281:
6265:
6261:
6257:
6250:
6248:
6246:
6244:
6242:
6226:
6222:
6220:0-8155-1237-6
6216:
6212:
6208:
6201:
6187:on 2016-06-04
6183:
6179:
6175:
6171:
6167:
6163:
6159:
6155:
6151:
6146:
6141:
6138:(1): 011202.
6137:
6133:
6126:
6119:
6112:
6107:
6105:
6103:
6095:
6093:
6080:
6076:
6070:
6063:
6058:
6051:
6046:
6039:
6024:
6017:
6013:
6007:
5999:
5995:
5991:
5987:
5983:
5979:
5975:
5971:
5967:
5963:
5956:
5948:
5944:
5939:
5934:
5929:
5924:
5920:
5916:
5912:
5908:
5904:
5897:
5890:
5885:
5870:
5866:
5860:
5856:
5852:
5848:
5844:
5838:
5832:, p. 44.
5831:
5826:
5819:
5814:
5812:
5810:
5803:, p. 48.
5802:
5797:
5781:
5777:
5773:
5767:
5760:
5755:
5749:
5745:
5742:
5736:
5729:
5724:
5717:
5715:
5710:
5705:
5703:
5701:
5699:
5691:
5686:
5679:
5674:
5666:
5662:
5658:
5654:
5650:
5646:
5642:
5638:
5631:
5623:
5619:
5615:
5611:
5607:
5603:
5596:
5581:
5577:
5573:
5567:
5561:, p. 13.
5560:
5555:
5548:
5543:
5536:
5531:
5516:
5512:
5505:
5503:
5501:
5493:
5488:
5472:
5468:
5464:
5460:
5459:
5451:
5443:
5439:
5435:
5431:
5424:
5422:
5420:
5401:
5397:
5393:
5389:
5385:
5381:
5377:
5373:
5369:
5362:
5355:
5353:
5351:
5334:
5330:
5326:
5320:
5313:
5301:
5297:
5293:
5287:
5283:
5266:
5260:
5251:
5244:
5240:
5234:
5227:
5226:ideal gas law
5223:
5222:
5216:
5207:
5200:
5196:
5192:
5188:
5187:melting point
5184:
5178:
5171:
5167:
5166:molten silica
5161:
5150:
5102:
5095:
5090:
5088:
5074:
5070:
5060:
5057:
5055:
5052:
5050:
5047:
5045:
5042:
5040:
5039:Water cluster
5037:
5035:
5032:
5030:
5027:
5025:
5022:
5020:
5017:
5015:
5012:
5010:
5007:
5005:
5002:
5000:
4997:
4995:
4992:
4990:
4987:
4985:
4982:
4980:
4977:
4975:
4972:
4971:
4966:
4960:
4955:
4948:
4946:
4942:
4938:
4930:
4926:
4924:
4901:
4897:
4893:
4892:hydroxic acid
4888:
4886:
4882:
4878:
4874:
4870:
4866:
4862:
4858:
4854:
4849:
4847:
4843:
4839:
4835:
4831:
4827:
4823:
4819:
4816:The accepted
4809:
4807:
4803:
4799:
4795:
4791:
4787:
4783:
4779:
4775:
4771:
4766:
4764:
4760:
4756:
4754:
4750:
4746:
4742:
4738:
4734:
4725:
4665:
4664:
4663:
4617:
4616:
4615:
4611:
4562:
4561:
4560:
4558:
4532:
4528:
4526:
4522:
4518:
4514:
4510:
4456:
4455:
4454:
4452:
4448:
4444:
4440:
4430:
4428:
4424:
4420:
4412:
4411:leaving group
4408:
4404:
4400:
4396:
4381:
4377:
4364:
4351:
4347:
4345:
4340:
4317:
4313:
4309:
4284:
4280:
4276:
4219:
4218:
4217:
4215:
4211:
4207:
4128:
4054:
3985:
3984:
3983:
3981:
3977:
3973:
3969:
3891:
3890:
3889:
3864:
3788:
3787:
3786:
3784:
3768:
3756:
3752:
3748:
3744:
3729:
3727:
3723:
3719:
3715:
3710:
3682:
3676:
3675:
3670:
3653:
3612:
3581:
3558:
3554:
3551:. Tritium is
3550:
3546:
3542:
3524:
3478:
3474:
3459:
3455:
3451:
3447:
3443:
3442:isotopologues
3439:
3432:Isotopologues
3429:
3427:
3423:
3419:
3409:
3385:
3381:
3377:
3372:
3370:
3366:
3365:clay minerals
3362:
3358:
3354:
3353:water erosion
3350:
3340:
3320:
3297:
3287:
3274:
3263:
3259:
3245:
3209:
3198:
3188:
3178:
3165:
3161:
3153:
3143:
3133:
3126:
3112:
3104:
3103:
3102:
3100:
3095:
3093:
3067:
3063:
3040:
3017:
3007:
2994:
2983:
2974:
2973:ionic product
2970:
2919:
2918:
2917:
2915:
2911:
2907:
2901:
2886:
2884:
2881:explanation (
2880:
2876:
2872:
2868:
2864:
2858:
2848:
2846:
2845:string theory
2841:
2836:
2833:
2829:
2825:
2821:
2820:thermodynamic
2817:
2813:
2809:
2801:
2796:
2787:
2785:
2781:
2777:
2773:
2769:
2765:
2761:
2757:
2753:
2749:
2745:
2744:visible light
2739:
2729:
2727:
2723:
2719:
2718:hydrogen bond
2715:
2709:
2699:
2697:
2693:
2689:
2675:
2661:
2656:
2654:
2649:
2645:
2641:
2636:
2634:
2630:
2625:
2623:
2619:
2615:
2611:
2607:
2599:
2595:
2591:
2588:
2583:
2578:
2568:
2560:
2558:
2549:
2542:
2537:
2525:
2521:
2517:
2513:
2508:
2504:
2501:
2497:
2493:
2489:
2485:
2480:
2478:
2471:
2467:
2463:
2459:
2452:
2450:
2446:
2416:
2410:
2408:
2404:
2400:
2396:
2392:
2388:
2383:
2381:
2377:
2373:
2369:
2360:
2355:
2345:
2339:
2335:
2331:
2327:
2323:
2319:
2314:
2311:
2307:
2303:
2298:
2296:
2292:
2287:
2254:
2250:
2246:
2231:
2229:
2225:
2224:with pressure
2221:
2216:
2212:
2205:Melting point
2197:
2194:
2191:
2190:
2186:
2183:
2180:
2179:
2175:
2172:
2169:
2161:
2160:
2157:0.16 °C
2156:
2153:
2151:
2147:
2146:
2142:
2139:
2137:
2133:
2132:
2128:
2125:
2123:
2115:
2114:
2110:
2107:
2104:
2097:
2096:
2092:
2089:
2086:
2085:
2079:
2077:
2073:
2068:
2066:
2062:
2058:
2054:
2050:
2046:
2038:
2030:
2025:
2015:
2011:
2009:
2004:
1999:
1973:
1959:
1954:
1944:
1940:
1938:
1934:
1930:
1926:
1922:
1917:
1915:
1912:such as with
1911:
1907:
1903:
1899:
1895:
1889:
1883:
1874:
1865:
1863:
1859:
1858:
1853:
1849:
1845:
1840:
1838:
1834:
1829:
1825:
1816:
1812:
1803:
1801:
1797:
1787:
1783:
1781:
1777:
1773:
1769:
1765:
1760:
1756:
1754:
1753:hexagonal ice
1750:
1746:
1741:
1732:
1723:
1720:
1718:
1717:refrigeration
1714:
1710:
1706:
1703:The specific
1701:
1699:
1695:
1691:
1687:
1683:
1679:
1670:
1661:
1658:
1655:
1651:
1647:
1643:
1639:
1635:
1631:
1626:
1624:
1620:
1616:
1615:gaseous phase
1612:
1611:phases of ice
1608:
1605:. Aside from
1604:
1600:
1596:
1592:
1588:
1584:
1580:
1570:
1568:
1564:
1563:oxygen family
1560:
1555:
1553:
1549:
1545:
1541:
1537:
1534:
1531:
1528:
1524:
1508:
1504:
1501:Water is the
1498:
1488:
1464:
1460:
1456:
1443:
1430:
1426:
1422:
1417:
1415:
1414:heat capacity
1412:, and a high
1411:
1407:
1406:boiling point
1402:
1398:
1393:
1391:
1387:
1383:
1379:
1375:
1371:
1367:
1363:
1359:
1355:
1351:
1341:
1329:
1322:
1317:
1300:
1294:
1291:
1287:
1282:
1276:
1273:
1271:
1268:
1266:
1263:
1261:
1258:
1256:
1253:
1252:
1250:
1247:
1242:
1241:
1235:
1232:
1230:
1227:
1225:
1222:
1220:
1217:
1215:
1212:
1211:
1209:
1206:
1201:
1200:
1195:
1192:
1189:
1186:
1183:
1182:
1178:
1176:
1173:
1172:
1165:
1158:
1151:
1127:
1124:
1123:
1119:
1118:
1115:
1110:
1106:
1103:
1098:
1097:
1093:
1092:
1087:
1082:
1078:
1073:
1065:
1061:
1060:
1056:
1048:
1040:
1034:
1033:
1029:
1021:
1016:
1010:
1009:
1005:
1000:
995:
994:Heat capacity
991:
990:
985:
982:
978:
975:
974:Dipole moment
971:
970:
967:
964:
961:
957:
956:
949:
946:
942:
941:
938:
935:
932:
928:
927:
922:
918:
914:
912:
909:
908:
904:
897:
893:
889:
888:
884:
882:
879:
878:
870:
867:
865:
862:
861:
849:
846:
844:
841:
840:
836:
830:
826:
823:
822:
818:
812:
808:
805:
804:
800:
798:
795:
794:
790:
786:
785:ethyl acetate
782:
778:
774:
773:diethyl ether
768:
764:
760:
756:
752:
748:
744:
740:
736:
732:
728:
724:
720:
716:
710:
706:
702:
698:
692:
688:
684:
680:
676:
674:
671:
670:
666:
664:
663:Boiling point
661:
660:
656:
654:
653:Melting point
651:
650:
643:
640:
624:
608:
604:(670) °C
587:
571:
568:
564:
563:
561:
559:
556:
555:
551:
549:
546:
545:
541:
537:
534:
533:
529:
527:
524:
523:
507:
504:
500:
499:
494:
485:
484:
481:
474:
460:
459:
456:
449:
442:
438:
437:
435:
433:
430:
429:
422:
421:
419:
417:
414:
413:
406:
402:
401:
399:
393:
389:
388:
381:
377:
376:
374:
372:
369:
368:
364:
361:
357:
356:
349:
348:
346:
344:
339:
338:
331:
327:
326:
324:
322:
319:
318:
311:
307:
306:
304:
302:
299:
298:
291:
290:ChEMBL1098659
287:
286:
284:
282:
279:
278:
271:
267:
266:
264:
262:
259:
258:
254:
251:
247:
246:
239:
235:
234:
232:
228:
223:
222:
215:
211:
210:
208:
205:
201:
200:
195:
187:
184:
181:
178:
175:
172:
170:Hydroxic acid
169:
166:
163:
160:
157:
154:
151:
148:
145:
144:
139:
131:
127:
119:
115:
110:
106:
101:
76:
72:
66:
62:
61:
57:
53:
48:
40:
33:
19:
17907:Oceanography
17417:
17408:
17361:
17355:
17344:
17335:
17328:
17323:
17313:
17308:
17073:
17067:
16986:
16951:
16942:
16936:Livermoranes
16701:Polysulfanes
16630:
16624:Polyoxidanes
16592:
16557:
16548:
16049:
16041:
16036:
16027:
15629:Cycloalkynes
15624:Cycloalkenes
15619:Cycloalkanes
15596:Hydrocarbons
15569:
15557:
15549:
15544:
15535:
15135:Monohydrides
14034:
13099:Lead dioxide
12016:Radium oxide
11966:Nitric oxide
11749:Barium oxide
11714:
11702:Sodium oxide
11676:Silver oxide
10652:
10056:H[Co(CO)
9587:
8952:Hydrobiology
8937:Distribution
8819:
8693:
8666:. Retrieved
8639:
8636:Ben-Naim, A.
8605:
8577:
8550:
8524:
8503:
8492:. Retrieved
8474:CRC Handbook
8468:
8458:
8447:the original
8420:
8408:. Retrieved
8382:
8355:
8332:
8320:. Retrieved
8300:
8278:
8257:Biochemistry
8256:
8228:
8220:Bibliography
8207:. Retrieved
8196:
8187:
8175:
8164:. Retrieved
8151:
8142:
8130:
8118:
8078:
8074:
8068:
8057:. Retrieved
8048:
8039:
8028:. Retrieved
8019:
8011:
7974:Charlot 2007
7969:
7957:
7945:
7933:
7921:
7910:. Retrieved
7897:
7888:
7876:
7864:
7837:
7826:. Retrieved
7817:
7808:
7797:. Retrieved
7785:
7781:
7768:
7756:
7726:(1): 69–74.
7723:
7719:
7713:
7701:. Retrieved
7696:
7686:
7674:. Retrieved
7669:
7660:
7635:
7629:
7604:
7600:
7594:
7583:. Retrieved
7569:
7557:
7545:
7533:
7524:
7521:
7514:. Retrieved
7495:
7485:
7475:
7461:
7441:
7433:
7390:
7386:
7376:
7335:
7331:
7325:
7314:. Retrieved
7279:
7275:
7265:
7222:
7218:
7208:
7197:. Retrieved
7172:
7162:
7150:
7138:
7126:
7097:
7074:
7062:
7019:
7015:
7005:
6993:
6981:
6969:
6944:
6940:
6934:
6923:. Retrieved
6919:the original
6904:
6879:
6875:
6857:
6846:. Retrieved
6822:
6810:
6799:. Retrieved
6774:(6): 40–46.
6771:
6767:
6754:
6743:. Retrieved
6708:
6704:
6694:
6682:
6669:
6663:
6652:the original
6639:
6632:
6620:. Retrieved
6600:
6594:
6589:, p. 5.
6567:. Retrieved
6549:
6538:. Retrieved
6505:
6501:
6495:
6483:
6472:. Retrieved
6460:HyperPhysics
6459:
6424:(10): 5529.
6421:
6417:
6411:
6400:. Retrieved
6391:
6365:
6353:
6320:
6316:
6307:
6298:0130-39913-2
6288:
6268:. Retrieved
6259:
6229:. Retrieved
6210:
6200:
6189:. Retrieved
6182:the original
6135:
6132:Phys. Rev. E
6131:
6118:
6091:
6090:
6083:. Retrieved
6079:the original
6069:
6057:
6045:
6036:
6030:. Retrieved
6006:
5965:
5961:
5955:
5910:
5906:
5896:
5884:
5873:. Retrieved
5846:
5837:
5825:
5820:, p. 2.
5796:
5786:December 15,
5784:. Retrieved
5775:
5766:
5754:
5735:
5723:
5712:
5685:
5673:
5640:
5636:
5630:
5605:
5601:
5595:
5584:. Retrieved
5566:
5554:
5542:
5530:
5519:. Retrieved
5487:
5475:. Retrieved
5457:
5450:
5433:
5429:
5407:. Retrieved
5371:
5367:
5337:. Retrieved
5328:
5319:
5311:
5304:. Retrieved
5300:the original
5295:
5286:
5259:
5250:
5233:
5219:
5215:
5206:
5177:
5160:
5148:
5101:
5073:
4965:Water portal
4928:
4927:
4899:
4895:
4891:
4889:
4880:
4872:
4852:
4850:
4841:
4825:
4821:
4815:
4812:Nomenclature
4806:triple point
4767:
4757:
4737:electrolysis
4731:
4722:
4661:
4613:
4604:Electrolysis
4529:
4506:
4436:
4376:carbocations
4373:
4272:
4203:
4171:(Lewis acid)
4146:(Lewis base)
4097:(Lewis base)
4072:(Lewis acid)
4028:(Lewis base)
4003:(Lewis acid)
3965:
3860:
3740:
3711:
3688:
3673:
3671:
3654:
3582:
3525:
3435:
3415:
3373:
3357:metasomatism
3346:
3343:Geochemistry
3228:
3096:
2966:
2903:
2860:
2837:
2805:
2741:
2711:
2657:
2637:
2626:
2603:
2598:Havasu Falls
2585:Presence of
2566:
2554:
2481:
2474:
2458:
2443:is a gas at
2411:
2384:
2380:sigma bonded
2365:
2330:electrolyzed
2315:
2299:
2288:
2242:
2208:
2198:−70 °C
2187:−24 °C
2176:−35 °C
2093:Temperature
2069:
2053:triple point
2042:
2018:Triple point
2012:
2003:bulk modulus
2000:
1969:
1941:
1918:
1914:hydrocarbons
1891:
1855:
1841:
1833:Arctic Ocean
1827:
1820:
1792:
1761:
1757:
1737:
1721:
1702:
1675:
1627:
1579:liquid phase
1576:
1556:
1538:to a single
1500:
1418:
1394:
1343:
1339:
1338:
1121:
1100:Main hazards
1089:
1071:
1046:
1019:
998:
895:
828:
810:
767:acetonitrile
751:acetaldehyde
697:carboxylates
540:hint of blue
416:RTECS number
197:Identifiers
141:Other names
16519:Bismuthanes
13965:Oxypnictide
13365:Tin dioxide
9017:Ocean world
8942:Hydrosphere
8870:superheated
6622:19 November
6207:"Chapter 6"
5962:Soft Matter
5741:PubChem 962
5296:www.iun.edu
5115:represents
5054:Water model
5049:Water dimer
5004:Heavy water
4763:heavy water
4747:. In 1805,
4344:monodentate
4214:baking soda
3980:HSAB theory
3976:Lewis acids
3785:is formed:
3674:Light water
3553:radioactive
3541:heavy water
3473:quintillion
3452:. Only 155
3378:, known as
2883:Bent's rule
2832:tetrahedral
2750:light, and
2696:table sugar
2614:hydrophobic
2610:hydrophilic
2496:hydrophilic
2376:tetrahedral
2211:supercooled
2108:611.657 Pa
2045:temperature
1837:fresh water
1798:in central
1796:Lake Baikal
1778:(HDA), and
1751:. Regular,
1619:water vapor
1583:solid phase
1175:Flash point
1094:(OHS/OSH):
945:Point group
739:1,4-dioxane
727:isopropanol
679:haloalkanes
535:Appearance
496:Properties
270:CHEBI:15377
164:Hydric acid
17877:Hydroxides
17846:Categories
16542:Moscovanes
16407:Phosphenes
16269:Phosphanes
16067:Pnictogen
16024:Flerovanes
15175:Dihydrides
15125:(Group 2)
14021:Hydroxides
13960:Superoxide
12123:Zinc oxide
9139:Wiktionary
8974:management
8820:Properties
8668:August 31,
8494:2016-05-29
8410:2016-07-31
8322:2008-11-19
8209:2017-09-08
8166:2016-07-31
8123:IUPAC 2005
8081:(6): 341.
8059:2016-05-26
8030:2016-06-24
7912:2016-06-25
7894:"Hydrides"
7828:2023-04-11
7799:2023-04-11
7585:2008-03-21
7316:2019-09-08
7199:2019-07-05
6947:(1): 1–8.
6925:2009-12-06
6848:2013-02-19
6815:Sharp 1988
6801:2011-11-22
6745:2020-08-31
6646:(Thesis).
6540:2021-08-03
6474:2007-10-26
6402:2016-06-09
6270:2016-06-03
6231:2010-07-11
6191:2009-07-07
6032:2017-12-22
5875:2018-08-09
5586:2016-04-09
5521:2014-06-01
5477:17 October
5430:Metrologia
5409:2018-08-09
5374:(8): 612.
5274:References
5265:amphoteric
5239:atmosphere
4999:Hard water
4824:or simply
4786:Fahrenheit
4395:hydrolysis
3972:Lewis base
3968:lone pairs
3743:amphoteric
3714:frost line
3685:Occurrence
3444:of water.
3349:weathering
2975:of water,
2855:See also:
2780:absorption
2764:microwaves
2660:table salt
2600:turquoise.
2541:paper clip
2492:organelles
2470:spider web
2372:lone pairs
2352:See also:
2215:nucleation
2154:632.4 MPa
2140:350.1 MPa
2126:209.9 MPa
2072:polymorphs
1996:10 Pa
1988:10 Pa
1980:10 Pa
1886:See also:
1764:polymorphs
1644:is 22.064
1593:, such as
1533:covalently
1495:See also:
1463:activities
1421:amphoteric
1410:molar mass
924:Structure
673:Solubility
637:95 °C
621:25 °C
526:Molar mass
441:059QF0KO0R
301:ChemSpider
225:3D model (
204:CAS Number
17902:Limnology
17418:predicted
17362:predicted
17345:predicted
17074:predicted
16952:predicted
16611:Hydrogen
16558:predicted
16028:predicted
16000:Plumbanes
15964:Stannanes
15634:Annulenes
15536:predicted
15532:Nihonanes
15496:Thallanes
15461:Indiganes
15231:Group 13
15123:Alkaline
13935:Oxocarbon
9738:[PtCl
9052:Enceladus
9033:Specific
8969:Resources
8964:Pollution
8947:Hydrology
8913:Hydronium
8903:Tritiated
8893:Semiheavy
8798:Overviews
8720:213738895
8606:Chemistry
7550:Boyd 2000
7538:Boyd 2000
7249:0036-8075
7102:Lide 2003
6896:1099-0062
6735:122365938
6530:105357042
6454:Nave, R.
6345:1463-9084
6085:August 3,
6062:Lide 2003
6050:Lide 2003
6012:Rhein, M.
5990:1744-6848
5728:Lide 2003
5690:Lide 2003
5678:Lide 2003
5665:0047-2689
5547:Lide 2003
5535:Lide 2003
5492:Lide 2003
5396:0021-9584
5306:1 October
5195:germanium
5066:Footnotes
4765:in 1933.
4513:beryllium
4509:aluminium
4457:2 Na + 2
4403:digestion
4253:⇌ NaOH +
4206:hydrolyze
3741:Water is
3732:Reactions
3557:half-life
3458:deuterium
3426:acid rain
3371:hardens.
3321:−
3260:≈
3179:−
3162:⋅
3092:data page
3066:hydroxide
3041:−
2914:hydroxide
2912:ions and
2910:hydronium
2798:Model of
2790:Structure
2629:hydration
2587:colloidal
2387:structure
2249:insulator
2090:Pressure
1929:dew point
1892:Water is
1713:drift ice
1595:ice cubes
1419:Water is
1111:(as snow)
1109:Avalanche
1039:formation
1013:Std molar
937:Hexagonal
911:Viscosity
869:Hydroxide
848:Hydronium
747:sulfolane
683:aliphatic
584:0 °C
552:Odorless
423:ZC0110000
350:231-791-2
342:EC Number
214:7732-18-5
17331:< 1)
16877:Tellanes
16496:Stibanes
15891:Germanes
15426:Gallanes
15233:hydrides
13955:Peroxide
13945:Oxyanion
13940:Suboxide
9831:[SiF
9255:H[BF
9117:Category
8742:of water
8638:(2011),
8547:(1988).
8488:Archived
8443:37341352
8401:Archived
8380:(2005).
8316:Archived
8203:Archived
8160:Archived
8053:Archived
8024:Archived
7906:Archived
7902:UC Davis
7898:Chemwiki
7822:Archived
7790:Archived
7748:39474797
7576:Archived
7574:. 2001.
7516:21 April
7510:Archived
7462:Water, H
7417:18354466
7368:11061625
7360:18264420
7310:Archived
7306:27152824
7257:26989250
7193:Archived
7083:cite web
7054:24699509
7022:: 4565.
6839:Archived
6792:Archived
6739:Archived
6613:Archived
6569:9 August
6563:Archived
6534:Archived
6468:Archived
6396:Archived
6264:Archived
6225:Archived
6170:12241346
6023:Archived
5998:32900070
5947:16179387
5869:Archived
5780:Archived
5776:usgs.gov
5744:Archived
5692:, 6.186.
5580:Archived
5515:Archived
5471:Archived
5400:Archived
5339:21 April
5333:Archived
4951:See also
4842:oxidanyl
4838:hydroxyl
4557:catalyst
4443:hydrides
4361:O. This
4316:hydrates
4285:such as
3475:include
3456:include
3438:isotopes
3436:Several
3418:nitrogen
2816:methanol
2722:monomers
2644:alcohols
2484:adhesion
2477:cohesion
2195:626 MPa
2184:344 MPa
2173:213 MPa
2049:pressure
1906:polarity
1894:miscible
1882:Humidity
1852:salinity
1848:seawater
1782:(VHDA).
1709:glaciers
1654:volcanic
1640:and the
1599:granular
1591:crystals
1552:glaciers
1527:hydrogen
1523:molecule
1265:Methanol
1246:solvents
1244:Related
1122:NFPA 704
1105:Drowning
1084:Hazards
825:Basicity
735:glycerol
723:propanol
715:methanol
701:alcohols
687:aromatic
321:DrugBank
255:3587155
17316:< 1)
16913:Polanes
16840:Selanes
16826:more...
16688:more...
16460:Arsanes
16393:more...
16202:more...
15857:Silynes
15828:Silenes
15814:more...
15690:Silanes
15614:alkynes
15609:alkenes
15604:alkanes
15242:Boranes
14735:
14096:[NH
13950:Ozonide
13928:Related
11181:more...
9128:Commons
8805:Outline
8279:Biology
8152:Pubchem
8083:Bibcode
7640:Bibcode
7636:Science
7609:Bibcode
7425:4365814
7395:Bibcode
7340:Bibcode
7284:Bibcode
7227:Bibcode
7219:Science
7189:6642535
7045:3988813
7024:Bibcode
6949:Bibcode
6776:Bibcode
6713:Bibcode
6510:Bibcode
6494:O Ice I
6426:Bibcode
6325:Bibcode
6178:6109212
6150:Bibcode
5970:Bibcode
5938:1242322
5915:Bibcode
5645:Bibcode
5610:Bibcode
5376:Bibcode
5199:bismuth
5191:gallium
5183:silicon
5170:kelvins
4822:oxidane
4798:Réaumur
4790:Delisle
4782:Rankine
4778:Celsius
4728:History
4521:rusting
4517:passive
4312:rhenium
3863:ammonia
3722:ammonia
3718:methane
3681:level.
3679:155 ppm
3477:tritium
3450:protium
2908:giving
2824:kinetic
2766:. Most
2752:far-red
2726:hexamer
2674:cations
2606:solvent
2415:melting
2334:protons
2228:Ice VII
2122:ice III
2059:, but,
1910:soluble
1898:ethanol
1844:surface
1842:As the
1800:Siberia
1772:ice III
1740:density
1694:climate
1682:ammonia
1636:is 647
1370:solvent
1352:) is a
1321:what is
1319: (
1275:Ammonia
1260:Ethanol
1255:Acetone
1067:(Δ
1015:entropy
979:1.8546
837:13.995
819:13.995
807:Acidity
789:bromine
731:acetone
719:ethanol
705:ketones
558:Density
520:
392:PubChem
330:DB09145
134:Oxidane
17892:Oxides
16216:Azenes
16078:Azanes
15391:Alanes
15008:Cm(OH)
14998:Am(OH)
14972:Np(OH)
14963:Np(OH)
14918:Th(OH)
14908:Ac(OH)
14893:Yb(OH)
14883:Tm(OH)
14873:Er(OH)
14863:Ho(OH)
14853:Dy(OH)
14843:Tb(OH)
14833:Gd(OH)
14823:Eu(OH)
14814:Eu(OH)
14804:Sm(OH)
14794:Pm(OH)
14784:Nd(OH)
14774:Pr(OH)
14764:Ce(OH)
14755:Ce(OH)
14745:La(OH)
14672:Ra(OH)
14646:Bi(OH)
14636:Pb(OH)
14627:Pb(OH)
14617:Tl(OH)
14603:Hg(OH)
14593:Au(OH)
14562:Lu(OH)
14549:Ba(OH)
14524:Te(OH)
14514:Sb(OH)
14504:Sn(OH)
14495:Sn(OH)
14485:In(OH)
14475:Cd(OH)
14457:Rh(OH)
14444:Tc(OH)
14428:Zr(OH)
14406:Sr(OH)
14378:As(OH)
14368:Ge(OH)
14358:Ga(OH)
14348:Zn(OH)
14338:Cu(OH)
14324:Ni(OH)
14314:Co(OH)
14304:Fe(OH)
14295:Fe(OH)
14285:Mn(OH)
14275:Cr(OH)
14266:Cr(OH)
14237:Ti(OH)
14228:Ti(OH)
14219:Ti(OH)
14209:Sc(OH)
14197:Ca(OH)
14152:Si(OH)
14142:Al(OH)
14132:Mg(OH)
14057:Be(OH)
11223:Oxides
9132:
9121:
9110:
9106:Portal
9099:
9037:Europa
8986:Supply
8979:policy
8959:Origin
8850:Liquid
8842:States
8738:, and
8718:
8708:
8616:
8593:
8563:
8533:
8510:
8480:
8441:
8431:
8393:
8366:
8340:
8308:
8287:
8264:
8243:
7746:
7740:515218
7738:
7502:
7454:
7423:
7415:
7387:Nature
7366:
7358:
7304:
7255:
7247:
7187:
7052:
7042:
6894:
6733:
6528:
6343:
6295:
6217:
6176:
6168:
6092:Gramme
5996:
5988:
5945:
5935:
5861:
5663:
5394:
4923:hydron
4898:, and
4863:, and
4800:, and
4794:Newton
4784:, and
4774:Kelvin
4447:alkali
4275:ligand
3935:(acid)
3910:(base)
3822:(base)
3797:(acid)
3726:Tethys
3422:sulfur
2916:ions.
2873:. The
2867:linear
2865:, not
2828:steric
2762:, and
2688:anions
2646:, and
2633:solute
2403:dipole
2291:solute
2168:Ice II
2150:ice VI
2120:, and
2057:kelvin
2008:oceans
1768:ice II
1657:plumes
1632:. The
1557:Under
1540:oxygen
1536:bonded
1521:; one
1362:liquid
1316:verify
1313:
1205:anions
1203:Other
1187:(SDS)
871:OH (pK
709:amines
691:ethers
641:Solid:
480:SMILES
380:C00001
281:ChEMBL
176:Hydrol
112:Names
90:
84:
45:Water
14940:U(OH)
14931:U(OH)
14418:Y(OH)
14256:V(OH)
14247:V(OH)
14172:S(OH)
14162:P(OH)
14107:O(OH)
14087:N(OH)
14077:C(OH)
14067:B(OH)
9506:NaHCO
8932:Cycle
8924:Earth
8898:Heavy
8881:Forms
8865:Steam
8860:Vapor
8815:Model
8791:Water
8716:S2CID
8450:(PDF)
8425:(PDF)
8404:(PDF)
8387:(PDF)
7793:(PDF)
7778:(PDF)
7744:S2CID
7703:7 Jan
7676:7 Jan
7579:(PDF)
7571:IAPWS
7566:(PDF)
7470:and H
7421:S2CID
7364:S2CID
7071:(PDF)
6842:(PDF)
6835:IAPWS
6831:(PDF)
6795:(PDF)
6764:(PDF)
6731:S2CID
6655:(PDF)
6644:(PDF)
6616:(PDF)
6605:(PDF)
6526:S2CID
6185:(PDF)
6174:S2CID
6140:arXiv
6128:(PDF)
6026:(PDF)
6019:(PDF)
5739:GHS:
5709:Water
5403:(PDF)
5364:(PDF)
5279:Notes
5024:Steam
4941:steam
4937:water
4826:water
4818:IUPAC
4802:Rømer
4255:NaHCO
3749:or a
3547:as a
3068:ion (
2648:salts
2640:acids
2539:This
2488:glass
2336:(see
2162:ice I
2136:ice V
2100:ice I
2034:ice I
1623:steam
1530:atoms
1505:with
1427:or a
1378:solid
1374:Earth
1354:polar
1340:Water
875:= 0)
858:= 0)
854:O (pK
626:0.961
610:0.997
600:3.983
589:0.999
573:0.999
567:VSMOW
455:InChI
261:ChEBI
227:JSmol
122:Water
39:Water
14985:(OH)
14953:(OH)
14666:FrOH
14612:TlOH
14543:CsOH
14469:AgOH
14400:RbOH
14390:BrOH
14333:CuOH
14181:ClOH
14126:NaOH
14051:LiOH
9846:HNCS
9841:HSCN
9515:HNCO
9463:HMnO
9350:HCNO
9336:HClO
9326:HClO
9316:HClO
9310:HClO
9296:HBrO
9286:HBrO
9276:HBrO
9270:HBrO
9233:HArF
9047:Moon
9042:Mars
8810:Data
8755:NASA
8706:ISBN
8670:2016
8614:ISBN
8591:ISBN
8561:ISBN
8531:ISBN
8508:ISBN
8478:ISBN
8439:OCLC
8429:ISBN
8391:ISBN
8364:ISBN
8338:ISBN
8306:ISBN
8285:ISBN
8262:ISBN
8241:ISBN
7736:PMID
7705:2011
7678:2011
7518:2019
7500:ISBN
7476:bent
7452:ISBN
7413:PMID
7356:PMID
7302:PMID
7253:PMID
7245:ISSN
7185:OSTI
7089:link
7050:PMID
6892:ISSN
6624:2018
6571:2018
6341:ISSN
6293:ISBN
6215:ISBN
6166:PMID
6087:2016
5994:PMID
5986:ISSN
5943:PMID
5859:ISBN
5788:2020
5661:ISSN
5479:2023
5392:ISSN
5341:2019
5308:2018
4751:and
4743:and
4702:+ 4
4691:+ 4
4629:+ 2
4575:+ 2
4525:iron
4511:and
4493:+ 2
4482:+ 2
4421:and
4321:FeSO
4287:Fe(H
4212:and
4210:soap
3751:base
3747:acid
3720:and
3701:and
3596:and
3420:and
3397:·23H
3386:, 4
3351:and
2967:The
2863:bent
2770:and
2712:The
2676:and
2594:lime
2397:, a
2295:salt
2245:ions
2047:and
2043:The
2001:The
1970:The
1902:oils
1738:The
1711:and
1621:(or
1603:snow
1476:and
1444:and
1429:base
1425:acid
1401:ions
1388:and
966:Bent
685:and
548:Odor
432:UNII
371:KEGG
365:117
185:Aqua
17825:PsH
17804:CfH
17796:CfH
17788:BkH
17780:BkH
17772:CmH
17764:AmH
17756:AmH
17748:PuH
17740:PuH
17732:NpH
17724:NpH
17700:PaH
17682:ThH
17674:ThH
17666:AcH
17645:LuH
17637:LuH
17629:YbH
17621:TmH
17613:TmH
17605:ErH
17597:ErH
17589:HoH
17581:HoH
17573:DyH
17565:DyH
17557:TbH
17549:TbH
17541:GdH
17533:GdH
17525:EuH
17517:SmH
17509:SmH
17501:NdH
17493:NdH
17485:PrH
17477:PrH
17469:CeH
17461:CeH
17453:LaH
17445:LaH
17437:LaH
17410:CnH
17401:HgH
17384:HgH
17376:CdH
17368:ZnH
17357:RgH
17351:CuH
17337:DsH
17321:PtH
17306:PdH
17301:NiH
17293:IrH
17285:RhH
17277:CoH
17269:FeH
17261:FeH
17256:FeH
17248:CrH
17240:CrH
17235:CrH
17227:TaH
17222:TaH
17214:NbH
17209:NbH
17188:HfH
17180:HfH
17172:ZrH
17164:ZrH
17156:TiH
17148:TiH
17140:LuH
17132:LuH
17092:ScH
17069:HTs
17063:HAt
17053:HBr
17048:HCl
17006:HDO
16995:(?)
16992:O–O
16944:LvH
16920:PoH
16600:(?)
16550:McH
16526:BiH
16503:SbH
16468:AsH
16050:FlH
16042:FlH
16037:FlH
16007:PbH
15972:SnH
15899:GeH
15877:SiH
15698:SiH
15570:NhH
15550:NhH
15545:NhH
15503:TlH
15468:InH
15433:GaH
15398:AlH
15214:BaH
15206:SrH
15198:CaH
15190:MgH
15182:BeH
15162:BaH
15157:SrH
15152:CaH
15147:MgH
15142:BeH
15113:CsH
15108:RbH
15098:NaH
15093:LiH
15031:No
15028:Md
15025:Fm
15022:Es
15019:Cf
15016:Bk
14993:Pu
14981:NpO
14926:Pa
14903:**
14728:Og
14725:Ts
14722:Lv
14719:Mc
14716:Fl
14713:Nh
14710:Cn
14707:Rg
14704:Ds
14701:Mt
14698:Hs
14695:Bh
14692:Sg
14689:Db
14686:Rf
14683:Lr
14680:**
14660:Rn
14657:At
14654:Po
14588:Pt
14585:Ir
14582:Os
14579:Re
14573:Ta
14570:Hf
14537:Xe
14533:IOH
14465:Pd
14452:Ru
14439:Mo
14436:Nb
14394:Kr
14386:Se
14191:KOH
14185:Ar
14120:Ne
14116:FOH
14100:]OH
14045:He
14035:HOH
11172:PtF
11047:NbO
11042:NbO
11013:NaO
10935:MoO
10927:MoO
10895:MnO
10890:MnO
10873:MgO
10800:IrO
10709:HgO
10701:HfO
10645:GeO
10640:GeO
10590:FeO
10549:CuO
10529:CsO
10485:CrO
10456:CoO
10379:ClO
10347:CeO
10342:CdO
10334:CaO
10329:CaO
10248:BrO
10228:BiO
10211:BeO
10206:BaO
10138:AmO
10035:TiO
10021:TeO
10007:TeO
9817:SiO
9803:SeO
9789:SeO
9579:NSO
9564:HNO
9536:HNO
9526:HNO
9520:HNO
9500:HNC
9491:MoO
9477:MnO
9453:HIO
9443:HIO
9433:HIO
9427:HIO
9417:HFO
9360:CrO
9345:HCN
9305:HCl
9265:HBr
9244:HSO
9238:HAt
9224:AsO
9210:AsO
8922:On
8855:Ice
8698:doi
8583:doi
8233:doi
8091:doi
7728:doi
7648:doi
7617:doi
7605:114
7403:doi
7391:452
7348:doi
7292:doi
7280:116
7235:doi
7223:351
7177:doi
7040:PMC
7032:doi
6957:doi
6884:doi
6784:doi
6721:doi
6709:131
6518:doi
6434:doi
6333:doi
6158:doi
5978:doi
5933:PMC
5923:doi
5911:102
5851:doi
5653:doi
5618:doi
5463:doi
5438:doi
5384:doi
5243:bar
5014:Ice
4945:ice
4565:AgF
4523:of
4413:in
4330:·7H
4306:to
4277:in
4178:Cl(
4104:Fe(
3792:HCl
3565:HDO
3561:THO
3454:ppm
3359:or
2466:Dew
2230:).
1992:3.9
1984:4.4
1976:5.1
1937:dew
1935:or
1933:fog
1828:not
1815:WOA
1646:MPa
1587:ice
1392:).
1382:gas
1191:SDS
1050:298
1023:298
635:at
628:887
619:at
612:047
602:035
598:at
591:974
582:at
575:842
405:962
395:CID
310:937
17848::
17716:UH
17708:UH
17695:15
17689:Th
17455:10
17389:Hg
17201:VH
17196:VH
17124:YH
17116:YH
17108:YH
17100:YH
17058:HI
17043:HF
17001:HS
16979:HO
16971:HO
16966:HO
16897:Te
16888:Te
16861:Se
16852:Se
16820:10
16594:NH
16585:HN
16580:NH
16572:HN
16476:As
16387:12
16383:10
16375:11
16363:10
16277:PH
16196:12
16192:10
16184:11
16172:10
16086:NH
15980:Sn
15949:12
15943:Ge
15937:10
15931:Ge
15919:Ge
15907:Ge
15865:Si
15836:Si
15808:22
15804:10
15802:Si
15796:20
15790:Si
15784:18
15778:Si
15772:16
15766:Si
15760:14
15754:Si
15748:12
15742:Si
15736:10
15730:Si
15718:Si
15706:Si
15664:CH
15656:CH
15651:CH
15558:Nh
15511:Tl
15476:In
15441:Ga
15406:Al
15377:22
15373:18
15365:14
15361:10
15353:12
15341:10
15329:11
15305:10
15258:BH
15250:BH
15103:KH
14949:UO
14740:*
14576:W
14557:*
13979:.
13914:Hs
13900:Xe
13886:Ru
13872:Os
13858:Ir
13830:Tc
13813:Re
13796:Mn
13779:Cl
13754:Xe
13712:Te
13684:Se
13670:Re
13656:Po
13642:Mo
13628:Cr
13583:Ta
13566:Pa
13532:Nb
13498:Bi
13481:As
13464:Sb
13439:Zr
13383:Ti
13369:Sn
13355:Th
13341:Tb
13327:Te
13313:Tc
13285:Si
13271:Se
13257:Ru
13243:Rh
13229:Pa
13215:Pr
13201:Po
13187:Pu
13173:Pt
13159:Os
13131:Np
13117:Mn
13103:Pb
13089:Hf
13075:Ir
13047:Ge
13016:Cm
13002:Cr
12988:Cl
12974:Ce
12932:Cf
12918:Br
12904:Bk
12890:Am
12845:Yb
12794:Ti
12777:Tm
12760:Tl
12743:Tb
12726:Sc
12709:Sm
12692:Rh
12675:Pm
12658:Pr
12624:Ni
12607:Nd
12590:Mn
12573:Lu
12556:La
12539:Fe
12522:In
12505:Ho
12488:Au
12471:Ga
12454:Gd
12437:Eu
12420:Er
12403:Es
12386:Dy
12352:Co
12335:Cr
12318:Ce
12301:Cf
12284:Cs
12250:Bi
12233:Bk
12216:As
12199:Sb
12182:Am
12165:Al
12148:Ac
12129:O)
12127:Zn
12119:O)
12109:O)
12099:O)
12097:Ti
12089:O)
12087:Sn
12079:O)
12077:Th
12052:O)
12042:O)
12040:Sr
12032:O)
12030:Si
12022:O)
12020:Ra
12012:O)
12010:Pa
12002:O)
12000:Po
11992:O)
11982:O)
11980:Pd
11972:O)
11962:O)
11960:Ni
11952:O)
11950:Hg
11942:O)
11940:Mn
11932:O)
11930:Mg
11922:O)
11920:Pb
11912:O)
11902:O)
11900:Fe
11892:O)
11890:Ge
11882:O)
11880:Eu
11855:O)
11853:Cu
11845:O)
11843:Co
11835:O)
11833:Cr
11825:O)
11823:Cl
11815:O)
11805:O)
11803:Ca
11795:O)
11793:Cd
11785:O)
11783:Br
11775:O)
11773:Be
11765:O)
11763:Bk
11755:O)
11753:Ba
11745:O)
11743:Al
11724:O)
11711:O)
11706:Na
11698:O)
11693:Tl
11685:O)
11680:Ag
11672:O)
11667:Rb
11659:O)
11646:O)
11633:O)
11628:Hg
11620:O)
11615:Li
11607:O)
11594:O)
11589:Ga
11581:O)
11576:Cl
11568:O)
11555:O)
11550:Cs
11542:O)
11537:Cu
11529:O)
11524:Al
11479:Br
11462:Tb
11445:Ag
11435:11
11428:Pr
11414:12
11394:Mn
11377:Pb
11360:Fe
11343:Cl
11326:Co
11309:Cl
11292:Cl
11261:12
11241:Sb
11088:OF
11081:OF
11056:Nd
11030:Na
11021:Na
10960:NO
10955:NO
10943:Mo
10915:Mn
10903:Mn
10878:Mg
10861:Lu
10849:Li
10840:Li
10828:La
10808:KO
10788:In
10735:IO
10723:Ho
10714:Hg
10628:Ga
10619:Ga
10607:Fe
10595:Fe
10578:Eu
10566:Er
10554:Dy
10537:Cs
10523:12
10517:Cr
10505:Cr
10493:Cr
10473:Co
10461:Co
10444:Cl
10432:Cl
10420:Cl
10408:Cl
10396:Cl
10387:Cl
10367:Ce
10355:Ce
10309:CO
10304:CO
10281:Br
10268:Br
10256:Br
10236:Bi
10216:Bi
10182:Au
10170:As
10158:As
10146:Am
10126:Al
10114:Ag
10049:Po
9996:Te
9981:SO
9977:CF
9884:SO
9870:SO
9856:SO
9778:Se
9726:10
9688:PO
9674:PO
9660:PO
9422:HI
9412:HF
9403:CS
9389:CO
9371:Cr
8753:,
8734:,
8730:,
8714:.
8704:.
8692:.
8660:.
8642:,
8612:.
8589:.
8559:.
8557:27
8486:.
8472:.
8437:.
8399:.
8362:.
8314:.
8239:.
8195:.
8158:.
8154:.
8150:.
8103:^
8089:.
8077:.
8047:.
7996:^
7981:^
7900:.
7896:.
7849:^
7816:.
7786:26
7784:.
7780:.
7742:.
7734:.
7724:23
7722:.
7695:.
7668:.
7646:.
7615:.
7603:.
7568:.
7520:.
7508:.
7494:.
7460:.
7450:.
7448:59
7419:.
7411:.
7401:.
7389:.
7385:.
7362:.
7354:.
7346:.
7336:36
7334:.
7308:.
7300:.
7290:.
7278:.
7274:.
7251:.
7243:.
7233:.
7221:.
7217:.
7191:.
7183:.
7171:.
7109:^
7085:}}
7081:{{
7073:.
7048:.
7038:.
7030:.
7018:.
7014:.
6955:.
6945:74
6943:.
6913:.
6890:.
6878:.
6866:^
6833:.
6790:.
6782:.
6772:56
6770:.
6766:.
6737:.
6729:.
6719:.
6707:.
6703:.
6672:.
6607:.
6579:^
6561:.
6557:.
6532:.
6524:.
6516:.
6506:47
6504:.
6500:.
6466:.
6462:.
6458:.
6446:^
6432:.
6422:59
6420:.
6390:.
6377:^
6339:.
6331:.
6319:.
6279:^
6262:.
6258:.
6240:^
6223:.
6172:.
6164:.
6156:.
6148:.
6136:66
6134:.
6130:.
6101:^
6089:.
6035:.
5992:.
5984:.
5976:.
5964:.
5941:.
5931:.
5921:.
5909:.
5905:.
5867:.
5857:.
5845:.
5808:^
5697:^
5659:.
5651:.
5641:24
5639:.
5616:.
5606:94
5604:.
5574:.
5513:.
5499:^
5469:.
5434:38
5432:.
5418:^
5398:.
5390:.
5382:.
5372:70
5370:.
5366:.
5349:^
5331:.
5327:.
5310:.
5294:.
5143:O)
5134:(H
5086:^
4939:,
4912:OH
4894:,
4887:.
4859:,
4848:.
4796:,
4792:,
4780:,
4776:,
4679:→
4666:2
4647:→
4618:2
4563:4
4544:/H
4495:OH
4484:Na
4470:→
4445:,
4429:.
4384:OH
4357:7H
4296:O)
4240:+
4230:CO
4221:Na
4176:→
4151:+
4133:Cl
4102:→
4077:+
4059:Fe
4033:→
4008:+
3954:OH
3952:+
3942:NH
3940:⇌
3915:+
3896:NH
3867:NH
3865:,
3849:Cl
3847:+
3827:⇌
3802:+
3703:CO
3652:.
3428:.
3388:CH
3070:OH
3062:pH
2955:OH
2953:+
2933:⇌
2920:2
2822:,
2786:.
2758:,
2746:,
2678:Cl
2664:Na
2642:,
2522:,
2518:,
2514:,
2326:cm
2322:μS
2308:,
2302:kΩ
2257:OH
2166:,
1916:.
1864:.
1774:,
1770:,
1700:.
1478:OH
1447:OH
1416:.
1042:(Δ
952:2v
919:)
917:cP
834:)
827:(p
816:)
809:(p
791:.
787:,
783:,
779:,
775:,
769:.
765:,
761:,
757:,
753:,
749:,
745:,
741:,
737:,
733:,
729:,
725:,
721:,
717:,
711:.
707:,
703:,
699:,
693:.
681:,
631:91
615:02
594:95
578:83
569:):
17806:3
17798:2
17790:3
17782:2
17774:2
17766:3
17758:2
17750:3
17742:2
17734:3
17726:2
17718:4
17710:3
17702:3
17693:H
17691:4
17684:4
17676:2
17668:2
17647:3
17639:2
17631:2
17623:3
17615:2
17607:3
17599:2
17591:3
17583:2
17575:3
17567:2
17559:3
17551:2
17543:3
17535:2
17527:2
17519:3
17511:2
17503:3
17495:2
17487:3
17479:2
17471:3
17463:2
17447:3
17439:2
17420:)
17416:(
17412:2
17403:2
17395:2
17393:H
17391:2
17378:2
17370:2
17364:)
17360:(
17347:)
17343:(
17339:2
17329:x
17327:(
17324:x
17314:x
17312:(
17309:x
17295:3
17287:2
17279:2
17271:5
17263:2
17250:x
17242:2
17229:2
17216:2
17203:2
17190:4
17182:2
17174:4
17166:2
17158:4
17150:2
17142:3
17134:2
17126:9
17118:6
17110:3
17102:2
17094:2
17076:)
17072:(
17024:O
17022:2
17020:T
17015:O
17013:2
17011:D
16990:2
16988:H
16981:3
16973:2
16954:)
16950:(
16946:2
16922:2
16899:2
16895:2
16893:H
16886:2
16884:H
16863:2
16859:2
16857:H
16850:2
16848:H
16818:S
16816:2
16814:H
16808:9
16806:S
16804:2
16802:H
16796:8
16794:S
16792:2
16790:H
16784:7
16782:S
16780:2
16778:H
16772:6
16770:S
16768:2
16766:H
16760:5
16758:S
16756:2
16754:H
16748:4
16746:S
16744:2
16742:H
16736:3
16734:S
16732:2
16730:H
16724:2
16722:S
16720:2
16718:H
16713:S
16711:2
16709:H
16682:5
16680:O
16678:2
16676:H
16670:4
16668:O
16666:2
16664:H
16658:3
16656:O
16654:2
16652:H
16646:2
16644:O
16642:2
16640:H
16635:O
16633:2
16631:H
16596:5
16587:5
16574:3
16560:)
16556:(
16552:3
16528:3
16505:3
16482:4
16480:H
16478:2
16470:3
16445:4
16443:H
16441:4
16439:P
16433:3
16431:H
16429:3
16427:P
16421:2
16419:H
16417:2
16415:P
16385:H
16381:P
16373:H
16371:9
16369:P
16361:H
16359:8
16357:P
16351:9
16349:H
16347:7
16345:P
16339:8
16337:H
16335:6
16333:P
16327:7
16325:H
16323:5
16321:P
16315:6
16313:H
16311:4
16309:P
16303:5
16301:H
16299:3
16297:P
16291:4
16289:H
16287:2
16285:P
16279:3
16254:4
16252:H
16250:4
16248:N
16242:3
16240:H
16238:3
16236:N
16230:2
16228:H
16226:2
16224:N
16194:H
16190:N
16182:H
16180:9
16178:N
16170:H
16168:8
16166:N
16160:9
16158:H
16156:7
16154:N
16148:8
16146:H
16144:6
16142:N
16136:7
16134:H
16132:5
16130:N
16124:6
16122:H
16120:4
16118:N
16112:5
16110:H
16108:3
16106:N
16100:4
16098:H
16096:2
16094:N
16088:3
16052:4
16044:2
16030:)
16026:(
16009:4
15986:6
15984:H
15982:2
15974:4
15947:H
15945:5
15935:H
15933:4
15925:8
15923:H
15921:3
15913:6
15911:H
15909:2
15901:4
15871:2
15869:H
15867:2
15842:4
15840:H
15838:2
15806:H
15794:H
15792:9
15782:H
15780:8
15770:H
15768:7
15758:H
15756:6
15746:H
15744:5
15734:H
15732:4
15724:8
15722:H
15720:3
15712:6
15710:H
15708:2
15700:4
15676:H
15674:2
15672:C
15666:3
15658:2
15572:5
15564:6
15562:H
15560:2
15552:3
15538:)
15534:(
15517:6
15515:H
15513:2
15505:3
15482:6
15480:H
15478:2
15470:3
15447:6
15445:H
15443:2
15435:3
15412:6
15410:H
15408:2
15400:3
15375:H
15371:B
15363:H
15359:B
15351:H
15349:6
15347:B
15339:H
15337:6
15335:B
15327:H
15325:5
15323:B
15317:9
15315:H
15313:5
15311:B
15303:H
15301:4
15299:B
15293:4
15291:H
15289:2
15287:B
15281:2
15279:H
15277:2
15275:B
15269:6
15267:H
15265:2
15263:B
15252:3
15216:2
15208:2
15200:2
15192:2
15184:2
15067:e
15060:t
15053:v
15010:3
15000:3
14987:3
14983:2
14974:4
14965:3
14955:2
14951:2
14942:3
14933:2
14920:4
14910:3
14895:3
14885:3
14875:3
14865:3
14855:3
14845:3
14835:3
14825:3
14816:2
14806:3
14796:3
14786:3
14776:3
14766:4
14757:3
14747:3
14674:2
14648:3
14638:4
14629:2
14619:3
14605:2
14595:3
14564:3
14551:2
14526:6
14516:3
14506:4
14497:2
14487:3
14477:2
14459:3
14446:4
14430:4
14420:3
14408:2
14380:3
14370:2
14360:3
14350:2
14340:2
14326:2
14316:2
14306:3
14297:2
14287:2
14277:3
14268:2
14258:3
14249:2
14239:4
14230:3
14221:2
14211:3
14199:2
14174:2
14164:3
14154:4
14144:3
14134:2
14109:2
14098:4
14089:3
14079:4
14069:3
14059:2
14013:e
14006:t
13999:v
13920:)
13918:4
13916:O
13912:(
13906:)
13904:4
13902:O
13898:(
13892:)
13890:4
13888:O
13884:(
13878:)
13876:4
13874:O
13870:(
13864:)
13862:4
13860:O
13856:(
13839:)
13837:7
13835:O
13833:2
13828:(
13822:)
13820:7
13818:O
13816:2
13811:(
13805:)
13803:7
13801:O
13799:2
13794:(
13788:)
13786:7
13784:O
13782:2
13777:(
13760:)
13758:3
13756:O
13752:(
13746:)
13744:3
13742:O
13740:U
13738:(
13732:)
13730:3
13728:O
13726:W
13724:(
13718:)
13716:3
13714:O
13710:(
13704:)
13702:3
13700:O
13698:S
13696:(
13690:)
13688:3
13686:O
13682:(
13676:)
13674:3
13672:O
13668:(
13662:)
13660:3
13658:O
13654:(
13648:)
13646:3
13644:O
13640:(
13634:)
13632:3
13630:O
13626:(
13609:)
13607:5
13605:O
13603:2
13600:V
13598:(
13592:)
13590:5
13588:O
13586:2
13581:(
13575:)
13573:5
13571:O
13569:2
13564:(
13558:)
13556:5
13554:O
13552:2
13549:P
13547:(
13541:)
13539:5
13537:O
13535:2
13530:(
13524:)
13522:5
13520:O
13518:2
13515:N
13513:(
13507:)
13505:5
13503:O
13501:2
13496:(
13490:)
13488:5
13486:O
13484:2
13479:(
13473:)
13471:5
13469:O
13467:2
13462:(
13445:)
13443:2
13441:O
13437:(
13431:)
13429:2
13427:O
13425:V
13423:(
13417:)
13415:2
13413:O
13411:U
13409:(
13403:)
13401:2
13399:O
13397:W
13395:(
13389:)
13387:2
13385:O
13381:(
13375:)
13373:2
13371:O
13367:(
13361:)
13359:2
13357:O
13353:(
13347:)
13345:2
13343:O
13339:(
13333:)
13331:2
13329:O
13325:(
13319:)
13317:2
13315:O
13311:(
13305:)
13303:2
13301:O
13299:S
13297:(
13291:)
13289:2
13287:O
13283:(
13277:)
13275:2
13273:O
13269:(
13263:)
13261:2
13259:O
13255:(
13249:)
13247:2
13245:O
13241:(
13235:)
13233:2
13231:O
13227:(
13221:)
13219:2
13217:O
13213:(
13207:)
13205:2
13203:O
13199:(
13193:)
13191:2
13189:O
13185:(
13179:)
13177:2
13175:O
13171:(
13165:)
13163:2
13161:O
13157:(
13151:)
13149:2
13147:O
13145:N
13143:(
13137:)
13135:2
13133:O
13129:(
13123:)
13121:2
13119:O
13115:(
13109:)
13107:2
13105:O
13101:(
13095:)
13093:2
13091:O
13087:(
13081:)
13079:2
13077:O
13073:(
13067:)
13065:2
13063:O
13061:I
13059:(
13053:)
13051:2
13049:O
13045:(
13039:)
13037:4
13035:O
13033:2
13030:N
13028:(
13022:)
13020:2
13018:O
13014:(
13008:)
13006:2
13004:O
13000:(
12994:)
12992:2
12990:O
12986:(
12980:)
12978:2
12976:O
12972:(
12966:)
12964:3
12962:O
12960:C
12958:(
12952:)
12950:2
12948:O
12946:C
12944:(
12938:)
12936:2
12934:O
12930:(
12924:)
12922:2
12920:O
12916:(
12910:)
12908:2
12906:O
12902:(
12896:)
12894:2
12892:O
12888:(
12871:)
12869:3
12867:O
12865:2
12862:Y
12860:(
12854:)
12852:3
12850:O
12848:2
12843:(
12837:)
12835:3
12833:O
12831:2
12828:V
12826:(
12820:)
12818:3
12816:O
12814:2
12811:W
12809:(
12803:)
12801:3
12799:O
12797:2
12792:(
12786:)
12784:3
12782:O
12780:2
12775:(
12769:)
12767:3
12765:O
12763:2
12758:(
12752:)
12750:3
12748:O
12746:2
12741:(
12735:)
12733:3
12731:O
12729:2
12724:(
12718:)
12716:3
12714:O
12712:2
12707:(
12701:)
12699:3
12697:O
12695:2
12690:(
12684:)
12682:3
12680:O
12678:2
12673:(
12667:)
12665:3
12663:O
12661:2
12656:(
12650:)
12648:6
12646:O
12644:4
12641:P
12639:(
12633:)
12631:3
12629:O
12627:2
12622:(
12616:)
12614:3
12612:O
12610:2
12605:(
12599:)
12597:3
12595:O
12593:2
12588:(
12582:)
12580:3
12578:O
12576:2
12571:(
12565:)
12563:3
12561:O
12559:2
12554:(
12548:)
12546:3
12544:O
12542:2
12537:(
12531:)
12529:3
12527:O
12525:2
12520:(
12514:)
12512:3
12510:O
12508:2
12503:(
12497:)
12495:3
12493:O
12491:2
12486:(
12480:)
12478:3
12476:O
12474:2
12469:(
12463:)
12461:3
12459:O
12457:2
12452:(
12446:)
12444:3
12442:O
12440:2
12435:(
12429:)
12427:3
12425:O
12423:2
12418:(
12412:)
12410:3
12408:O
12406:2
12401:(
12395:)
12393:3
12391:O
12389:2
12384:(
12378:)
12376:3
12374:O
12372:2
12369:N
12367:(
12361:)
12359:3
12357:O
12355:2
12350:(
12344:)
12342:3
12340:O
12338:2
12333:(
12327:)
12325:3
12323:O
12321:2
12316:(
12310:)
12308:3
12306:O
12304:2
12299:(
12293:)
12291:3
12289:O
12287:2
12282:(
12276:)
12274:3
12272:O
12270:2
12267:B
12265:(
12259:)
12257:3
12255:O
12253:2
12248:(
12242:)
12240:3
12238:O
12236:2
12231:(
12225:)
12223:3
12221:O
12219:2
12214:(
12208:)
12206:3
12204:O
12202:2
12197:(
12191:)
12189:3
12187:O
12185:2
12180:(
12174:)
12172:3
12170:O
12168:2
12163:(
12157:)
12155:3
12153:O
12151:2
12146:(
12125:(
12117:Y
12115:(
12107:V
12105:(
12095:(
12085:(
12075:(
12069:)
12067:2
12065:O
12063:2
12060:S
12058:(
12050:S
12048:(
12038:(
12028:(
12018:(
12008:(
11998:(
11990:P
11988:(
11978:(
11970:N
11968:(
11958:(
11948:(
11938:(
11928:(
11918:(
11910:I
11908:(
11898:(
11888:(
11878:(
11872:)
11870:2
11868:O
11866:2
11863:N
11861:(
11851:(
11841:(
11831:(
11821:(
11813:C
11811:(
11801:(
11791:(
11781:(
11771:(
11761:(
11751:(
11741:(
11722:2
11719:H
11717:(
11709:2
11704:(
11696:2
11691:(
11683:2
11678:(
11670:2
11665:(
11657:2
11654:K
11652:(
11644:2
11641:N
11639:(
11631:2
11626:(
11618:2
11613:(
11605:2
11602:I
11600:(
11592:2
11587:(
11579:2
11574:(
11566:2
11563:C
11561:(
11553:2
11548:(
11540:2
11535:(
11527:2
11522:(
11505:)
11503:8
11501:O
11499:3
11496:U
11494:(
11488:)
11486:8
11484:O
11482:3
11477:(
11471:)
11469:7
11467:O
11465:4
11460:(
11454:)
11452:2
11450:O
11448:2
11443:(
11437:)
11433:O
11431:6
11426:(
11420:)
11418:9
11416:O
11411:C
11409:(
11403:)
11401:4
11399:O
11397:3
11392:(
11386:)
11384:4
11382:O
11380:3
11375:(
11369:)
11367:4
11365:O
11363:3
11358:(
11352:)
11350:5
11348:O
11346:2
11341:(
11335:)
11333:4
11331:O
11329:3
11324:(
11318:)
11316:6
11314:O
11312:2
11307:(
11301:)
11299:4
11297:O
11295:2
11290:(
11284:)
11282:2
11280:O
11278:3
11275:C
11273:(
11267:)
11265:2
11263:O
11258:B
11256:(
11250:)
11248:4
11246:O
11244:2
11239:(
11215:e
11208:t
11201:v
11174:6
11170:2
11168:O
11160:2
11158:F
11156:6
11154:O
11146:2
11144:F
11142:5
11140:O
11132:2
11130:F
11128:4
11126:O
11118:2
11116:F
11114:3
11112:O
11104:2
11102:F
11100:2
11098:O
11090:2
11074:F
11072:2
11070:O
11062:3
11060:O
11058:2
11049:2
11036:2
11034:O
11032:2
11025:O
11023:2
11015:2
11007:5
11005:O
11003:2
11001:N
10995:4
10993:O
10991:2
10989:N
10983:3
10981:O
10979:2
10977:N
10972:O
10970:2
10968:N
10962:2
10949:3
10947:O
10945:2
10937:3
10929:2
10921:7
10919:O
10917:2
10909:3
10907:O
10905:2
10897:2
10884:3
10882:O
10880:2
10867:3
10865:O
10863:2
10855:2
10853:O
10851:2
10844:O
10842:2
10834:3
10832:O
10830:2
10822:2
10820:O
10818:2
10816:K
10810:2
10802:2
10794:3
10792:O
10790:2
10782:9
10780:O
10778:4
10776:I
10770:6
10768:O
10766:2
10764:I
10758:5
10756:O
10754:2
10752:I
10746:4
10744:O
10742:2
10740:I
10729:3
10727:O
10725:2
10718:O
10716:2
10703:2
10695:2
10693:O
10691:2
10689:H
10684:O
10682:2
10680:H
10675:O
10673:2
10671:H
10666:O
10664:2
10662:H
10657:O
10655:2
10653:H
10647:2
10634:3
10632:O
10630:2
10623:O
10621:2
10613:4
10611:O
10609:3
10601:3
10599:O
10597:2
10584:3
10582:O
10580:2
10572:3
10570:O
10568:2
10560:3
10558:O
10556:2
10543:3
10541:O
10539:2
10531:2
10521:O
10519:5
10511:5
10509:O
10507:2
10499:3
10497:O
10495:2
10487:3
10479:4
10477:O
10475:3
10467:3
10465:O
10463:2
10450:7
10448:O
10446:2
10438:6
10436:O
10434:2
10426:4
10424:O
10422:2
10414:3
10412:O
10410:2
10402:2
10400:O
10398:2
10391:O
10389:2
10381:2
10373:3
10371:O
10369:2
10361:4
10359:O
10357:3
10349:2
10336:2
10323:2
10321:O
10319:3
10317:C
10311:2
10295:8
10290:O
10286:3
10274:5
10272:O
10270:2
10262:3
10260:O
10258:2
10250:2
10242:5
10240:O
10238:2
10230:2
10222:3
10220:O
10218:2
10200:3
10198:O
10196:2
10194:B
10188:3
10186:O
10184:2
10176:5
10174:O
10172:2
10164:3
10162:O
10160:2
10152:3
10150:O
10148:2
10140:2
10132:3
10130:O
10128:2
10120:4
10118:O
10116:4
10095:e
10088:t
10081:v
10060:]
10058:4
10047:2
10045:H
10037:4
10033:4
10031:H
10023:6
10019:6
10017:H
10009:3
10005:2
10003:H
9994:2
9992:H
9985:H
9983:3
9979:3
9969:8
9967:O
9965:2
9963:S
9961:2
9959:H
9951:7
9949:O
9947:2
9945:S
9943:2
9941:H
9933:6
9931:O
9929:2
9927:S
9925:2
9923:H
9916:O
9914:3
9912:H
9904:3
9902:O
9900:2
9898:S
9896:2
9894:H
9886:5
9882:2
9880:H
9872:4
9868:2
9866:H
9858:3
9854:2
9852:H
9835:]
9833:6
9829:2
9827:H
9819:4
9815:4
9813:H
9805:4
9801:2
9799:H
9791:3
9787:2
9785:H
9776:2
9774:H
9766:2
9764:S
9762:2
9760:H
9753:S
9751:2
9749:H
9742:]
9740:6
9736:2
9734:H
9724:O
9722:3
9720:P
9718:5
9716:H
9708:7
9706:O
9704:2
9702:P
9700:4
9698:H
9690:4
9686:3
9684:H
9676:3
9672:3
9670:H
9662:2
9658:3
9656:H
9648:5
9646:O
9644:2
9642:H
9634:4
9632:O
9630:2
9628:H
9620:3
9618:O
9616:2
9614:H
9606:2
9604:O
9602:2
9600:H
9593:O
9591:2
9589:H
9581:3
9577:3
9575:H
9568:S
9566:5
9556:2
9554:O
9552:2
9550:N
9548:2
9546:H
9538:3
9528:2
9508:3
9493:4
9489:2
9487:H
9479:4
9475:2
9473:H
9465:4
9455:4
9445:3
9435:2
9405:3
9401:2
9399:H
9391:3
9387:2
9385:H
9377:7
9375:O
9373:2
9369:2
9367:H
9365:/
9362:4
9358:2
9356:H
9338:4
9328:3
9318:2
9298:4
9288:3
9278:2
9259:]
9257:4
9248:F
9246:3
9226:4
9222:3
9220:H
9212:3
9208:3
9206:H
9186:e
9179:t
9172:v
8783:e
8776:t
8769:v
8722:.
8700::
8672:.
8622:.
8599:.
8585::
8569:.
8539:.
8516:.
8497:.
8413:.
8372:.
8346:.
8325:.
8293:.
8270:.
8249:.
8235::
8212:.
8169:.
8097:.
8093::
8085::
8079:1
8062:.
8033:.
7915:.
7831:.
7750:.
7730::
7707:.
7680:.
7654:.
7650::
7642::
7623:.
7619::
7611::
7588:.
7478:.
7472:2
7468:3
7464:2
7427:.
7405::
7397::
7370:.
7350::
7342::
7319:.
7294::
7286::
7259:.
7237::
7229::
7202:.
7179::
7091:)
7056:.
7034::
7026::
7020:5
6963:.
6959::
6951::
6928:.
6898:.
6886::
6880:8
6851:.
6804:.
6786::
6778::
6748:.
6723::
6715::
6689:.
6676:.
6626:.
6573:.
6543:.
6520::
6512::
6496:h
6492:2
6477:.
6440:.
6436::
6428::
6405:.
6347:.
6335::
6327::
6321:3
6301:.
6273:.
6234:.
6194:.
6160::
6152::
6142::
6000:.
5980::
5972::
5966:3
5949:.
5925::
5917::
5891:.
5878:.
5853::
5790:.
5667:.
5655::
5647::
5624:.
5620::
5612::
5589:.
5524:.
5481:.
5465::
5444:.
5440::
5412:.
5386::
5378::
5343:.
5228:.
5185:(
5172:.
5149:n
5139:2
5126:O
5122:3
5117:H
5106:H
5079:a
4933:2
4904:H
4711:e
4693:H
4686:2
4681:O
4677:O
4673:2
4668:H
4654:2
4649:H
4638:e
4620:H
4595:2
4590:O
4586:O
4582:2
4577:H
4570:2
4553:O
4549:2
4540:2
4535:O
4477:2
4472:H
4468:O
4464:2
4459:H
4417:N
4415:S
4359:2
4355:4
4339:O
4335:2
4326:4
4301:6
4292:2
4260:3
4251:O
4247:2
4242:H
4235:3
4226:2
4196:6
4191:)
4189:O
4185:2
4180:H
4165:O
4161:2
4156:H
4122:6
4117:)
4115:O
4111:2
4106:H
4091:O
4087:2
4082:H
4044:O
4040:3
4035:H
4022:O
4018:2
4013:H
3990:H
3947:4
3929:O
3925:2
3920:H
3901:3
3879:H
3872:3
3838:O
3834:3
3829:H
3816:O
3812:2
3807:H
3772:H
3760:H
3707:×
3696:2
3691:H
3666:O
3662:2
3657:D
3650:O
3646:2
3641:D
3637:O
3633:2
3628:D
3624:O
3620:2
3615:H
3607:O
3603:2
3598:D
3594:O
3590:2
3585:H
3578:O
3574:2
3569:D
3537:O
3533:2
3528:D
3521:O
3510:O
3499:O
3488:H
3479:(
3469:H
3460:(
3406:O
3402:2
3393:4
3328:]
3317:H
3313:O
3308:[
3305:]
3298:+
3294:O
3288:3
3284:H
3278:[
3275:=
3269:w
3264:K
3254:q
3251:e
3246:K
3235:2
3231:3
3210:2
3204:O
3199:2
3195:H
3189:a
3175:H
3171:O
3166:a
3154:+
3150:O
3144:3
3140:H
3134:a
3127:=
3121:q
3118:e
3113:K
3081:H
3048:]
3037:H
3033:O
3028:[
3025:]
3018:+
3014:O
3008:3
3004:H
2998:[
2995:=
2989:w
2984:K
2944:O
2940:3
2935:H
2931:O
2927:2
2922:H
2526:.
2441:S
2437:2
2432:H
2428:S
2424:2
2419:H
2342:×
2324:/
2277:O
2273:3
2268:H
2164:h
2118:h
2102:h
2036:h
1994:×
1986:×
1978:×
1638:K
1519:O
1515:2
1510:H
1467:H
1434:H
1349:O
1347:2
1345:H
1342:(
1311:Y
1163:0
1156:0
1149:0
1074:)
1072:G
1069:f
1052:)
1047:H
1044:f
1025:)
1020:S
1018:(
1001:)
999:C
997:(
981:D
950:C
901:)
899:D
896:n
894:(
873:b
856:a
852:3
850:H
832:b
829:K
814:a
811:K
518:O
514:2
509:H
487:O
229:)
41:.
34:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.