257:
136:
44:
1725:
206:
191:
1719:
1731:
457:: The π systems form two parallel rings overlap in a "face-to-face" orientation. Aromatic molecules are also able to interact with each other in an "edge-to-face" orientation: The slight positive charge of the substituents on the ring atoms of one molecule are attracted to the slight negative charge of the aromatic system on another molecule.
473:
was discovered to adopt an asymmetric, rectangular configuration in which single and double bonds indeed alternate; there is no resonance and the single bonds are markedly longer than the double bonds, reducing unfavorable p-orbital overlap. This reduction of symmetry lifts the degeneracy of the two
414:
is not, since the number of π delocalized electrons is 4, which of course is a multiple of 4. The cyclobutadienide (2−) ion, however, is aromatic (6 electrons). An atom in an aromatic system can have other electrons that are not part of the system, and are therefore ignored for the 4n + 2 rule. In
196:
Since they are out of the plane of the atoms, these orbitals can interact with each other freely, and become delocalized. This means that, instead of being tied to one atom of carbon, each electron is shared by all six in the ring. Thus, there are not enough electrons to form double bonds on all the
478:
forces the two unpaired electrons into a new, weakly bonding orbital (and also creates a weakly antibonding orbital). Hence, cyclobutadiene is non-aromatic; the strain of the asymmetric configuration outweighs the anti-aromatic destabilization that would afflict the symmetric, square configuration.
422:
Aromatic molecules typically display enhanced chemical stability, compared to similar non-aromatic molecules. A molecule that can be aromatic will tend to alter its electronic or conformational structure to be in this situation. This extra stability changes the chemistry of the molecule. Aromatic
450:. The NMR signal of protons in the plane of an aromatic ring are shifted substantially further down-field than those on non-aromatic sp² carbons. This is an important way of detecting aromaticity. By the same mechanism, the signals of protons located near the ring axis are shifted up-field.
438:
Many of the earliest-known examples of aromatic compounds, such as benzene and toluene, have distinctive pleasant smells. This property led to the term "aromatic" for this class of compounds, and hence the term "aromaticity" for the eventually discovered electronic property.
887:. A π system with 4n electrons in a flat (non-twisted) ring would be anti-aromatic, and therefore highly unstable, due to the symmetry of the combinations of p atomic orbitals. By twisting the ring, the symmetry of the system changes and becomes allowed (see also
1259:
Alexander Kuhn, Puravankara
Sreeraj, Rainer Pöttgen, Hans-Dieter Wiemhöfer, Martin Wilkening,Paul Heitjans (2011). "Li NMR Spectroscopy on Crystalline Li12Si7: Experimental Evidence for the Aromaticity of the Planar Cyclopentadienyl-Analogous Si56− Rings".
572:. About 35 million tonnes are produced worldwide every year. They are extracted from complex mixtures obtained by the refining of oil or by distillation of coal tar, and are used to produce a range of important chemicals and polymers, including
151:
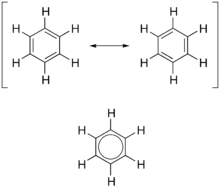
compound, which is best represented by a hybrid (average) of these structures, which can be seen at right. A C=C bond is shorter than a C−C bond, but benzene is perfectly hexagonal—all six carbon-carbon bonds have the same
848:) are structurally analogous to benzene, with the carbon atoms replaced by another element or elements. In borazine, the boron and nitrogen atoms alternate around the ring. Quite recently, the aromaticity of planar Si
126:
forms, which corresponds to the double and single bonds superimposing to give rise to six one-and-a-half bonds. Benzene is a more stable molecule than would be expected without accounting for charge delocalization.
464:
and are, in general, destabilized. Molecules that could be antiaromatic will tend to alter their electronic or conformational structure to avoid this situation, thereby becoming non-aromatic. For example,
931:
cation. Guanidinium does not have a ring structure but has six π-electrons which are delocalized over the molecule. However, this concept is controversial and some authors have stressed different effects.
305:) ... and when an additive compound is formed, the inner cycle of affinity suffers disruption, the contiguous carbon-atoms to which nothing has been attached of necessity acquire the ethylenic condition".
419:, the oxygen atom is sp² hybridized. One lone pair is in the π system and the other in the plane of the ring (analogous to C-H bond on the other positions). There are 6 π electrons, so furan is aromatic.
340:, since he recognized that his affinities had direction, not merely being point particles, and collectively having a distribution that could be altered by introducing substituents onto the benzene ring (
110:
to one another. These bonds may be seen as a hybrid of a single bond and a double bond, each bond in the ring identical to every other. This commonly seen model of aromatic rings, namely the idea that
1349:
264:
In the 19th century chemists found it puzzling that benzene could be so unreactive toward addition reactions, given its presumed high degree of unsaturation. The cyclohexatriene structure for
820:
the aromaticity is still retained. Aromaticity also occurs in compounds that are not carbon-based at all. Inorganic 6-membered-ring compounds analogous to benzene have been synthesized.
1485:
R. Caminiti, A. Pieretti, L. Bencivenni, F. Ramondo, N. Sanna (1996). "Amidine N−C(N)−N Skeleton: Its
Structure in Isolated and Hydrogen-Bonded Guanidines from ab Initio Calculations".
3016:
1431:
Alberto Gobbi, Gemot
Frenking (1993). "Y-Conjugated compounds: the equilibrium geometries and electronic structures of guanidine, guanidinium cation, urea, and 1,1-diaminoethylene".
147:, a double-headed arrow is used to indicate that the two structures are not distinct entities, but merely hypothetical possibilities. Neither is an accurate representation of the
244:, which are not aromatic in the chemical sense. But terpenes and benzenoid substances do have a chemical characteristic in common, namely higher unsaturation indices than many
272:
in 1865. Over the next few decades, most chemists readily accepted this structure, since it accounted for most of the known isomeric relationships of aromatic chemistry.
5231:
240:
character to apply to a group of chemical substances only some of which have notable aromas. Also, many of the most odoriferous organic substances known are
2030:
236:
in 1855. If this is indeed the earliest introduction of the term, it is curious that
Hofmann says nothing about why he introduced an adjective indicating
647:), one or more of the atoms in the aromatic ring is of an element other than carbon. This can lessen the ring's aromaticity, and thus (as in the case of
79:
exhibits a stabilization stronger than would be expected by the stabilization of conjugation alone. The earliest use of the term was in an article by
4347:
4292:
821:
5060:
1818:
1512:
1382:
1336:
1287:
1350:
Claire Castro, Zhongfang Chen, Chaitanya S. Wannere, Haijun Jiao, William L. Karney, Michael
Mauksch, Ralph Puchta, Nico J. R. van Eikema Hommes,
4402:
4552:
3186:
5281:
5055:
2881:
908:
1258:
4157:
2078:
1974:
4727:
3927:
2671:
907:. As of 2012, there is no proof that a Möbius aromatic molecule was synthesized. Aromatics with two half-twists corresponding to the
4892:
4822:
4802:
4297:
3464:
2926:
115:
3345:
2901:
1936:
1863:
1544:
961:
2114:
2010:
4647:
1354:(2005). "Investigation of a Putative Möbius Aromatic Hydrocarbon. The Effect of Benzannelation on Möbius Annulene Aromaticity".
1150:
Armit, James Wilson; Robinson, Robert (1925). "CCXI.?Polynuclear heterocyclic aromatic types. Part II. Some anhydronium bases".
5125:
5075:
3479:
1061:
A. T. Balaban, P. v. R. Schleyer and H. S. Rzepa (2005). "Crocker, Not Armit and
Robinson, Begat the Six Aromatic Electrons".
4582:
5221:
5030:
4687:
4667:
4627:
3434:
2377:
694:
are molecules containing two or more simple aromatic rings fused together by sharing two neighboring carbon atoms (see also
5216:
5146:
5045:
4702:
4557:
4187:
4032:
3642:
3269:
3046:
175:
above and below the ring. This model more correctly represents the location of electron density within the aromatic ring.
5380:
5296:
5080:
4392:
3882:
3557:
1858:
424:
313:
4102:
114:
was formed from a six-membered carbon ring with alternating single and double bonds (cyclohexatriene), was developed by
5291:
5005:
4867:
4657:
4622:
428:
5181:
5120:
4652:
4567:
4537:
4517:
4382:
4377:
3752:
3677:
3320:
3274:
3141:
2402:
283:
5286:
5246:
5196:
4872:
4672:
4422:
4352:
2841:
2412:
1783:
308:
Here, Armstrong is describing at least four modern concepts. First, his "affinity" is better known nowadays as the
4487:
3380:
3101:
5040:
4882:
4752:
4747:
4562:
4037:
3947:
3537:
3469:
3360:
2936:
2691:
2616:
2071:
1967:
1607:
956:
691:
4797:
1798:
5326:
5211:
5110:
5050:
4697:
4492:
4452:
4427:
4337:
3797:
2831:
2761:
2397:
2327:
1634:
1595:
1585:
927:
Y-aromaticity is a concept which was developed to explain the extraordinary stability and high basicity of the
888:
469:(COT) distorts itself out of planarity, breaking π overlap between adjacent double bonds. Relatively recently,
3917:
5316:
4902:
4792:
4412:
4137:
3922:
3867:
3712:
3672:
3504:
3259:
2976:
2826:
1590:
135:
5276:
4837:
4282:
1232:
Merino, Gabriel; Heine, Thomas; Seifert, Gotthard (2004). "The
Induced Magnetic Field in Cyclic Molecules".
600:
The overwhelming majority of aromatic compounds are compounds of carbon, but they need not be hydrocarbons.
5311:
5226:
5201:
5176:
5161:
5085:
5000:
4897:
4857:
4722:
4677:
4442:
3987:
3972:
3837:
3627:
3295:
3051:
2731:
2706:
2676:
2267:
1537:
1458:
Kenneth B. Wiberg (1990). "Resonance interactions in acyclic systems. 2. Y-Conjugated anions and cations".
487:
Aromatic compounds play key roles in the biochemistry of all living things. The four aromatic amino acids
279:, the discoverer of the electron, proposed three equivalent electrons between each carbon atom in benzene.
205:
5375:
5261:
5206:
5151:
4862:
4782:
4682:
4397:
4362:
4207:
4097:
3812:
3807:
3632:
3592:
3489:
3300:
3264:
3116:
3106:
2961:
2821:
2681:
2631:
2626:
2601:
2561:
2507:
2272:
2262:
2237:
342:
much as the distribution of the electric charge in a body is altered by bringing it near to another body
297:, who in 1890 wrote "the (six) centric affinities act within a cycle...benzene may be represented by a
5236:
4937:
4742:
4177:
4062:
3742:
3717:
3657:
3514:
3249:
2956:
2736:
2701:
2606:
2297:
2232:
2064:
1960:
197:
carbon atoms, but the "extra" electrons strengthen all of the bonds on the ring equally. The resulting
4527:
2791:
186:
above and below the plane of the ring. The following diagram shows the positions of these p-orbitals:
5336:
5241:
5095:
4975:
4947:
4917:
4832:
4762:
4717:
4692:
4612:
4512:
4472:
4167:
3787:
3777:
3702:
3226:
3086:
3081:
3061:
2746:
2543:
2522:
2482:
2407:
2045:
1853:
1843:
1833:
1808:
1778:
190:
2227:
5301:
5191:
5171:
5035:
4877:
4787:
4757:
4737:
4632:
4587:
4417:
4327:
4257:
4142:
4132:
3962:
3519:
3459:
3424:
3231:
3211:
3171:
2946:
2816:
2781:
2741:
2492:
2332:
2322:
2252:
1624:
256:
17:
4767:
5271:
5130:
4980:
4922:
4847:
4827:
4547:
4497:
4357:
4322:
4262:
4192:
3747:
3494:
3474:
3206:
3126:
3021:
2981:
2951:
2886:
2771:
2756:
2666:
2656:
2317:
2242:
2197:
1885:
1788:
1760:
1530:
1351:
912:
2532:
1301:
D. Ajami, O. Oeckler, A. Simon, R. Herges (2003). "Synthesis of a Möbius aromatic hydrocarbon".
312:, which was to be discovered only seven years later by J. J. Thomson. Second, he is describing
178:
The single bonds are formed with electrons in line between the carbon nuclei — these are called
5010:
4732:
4482:
4462:
4437:
4387:
4302:
4277:
4232:
4202:
4182:
4152:
4117:
4072:
4047:
4022:
3907:
3832:
3612:
3305:
3241:
3041:
2766:
2686:
2372:
2347:
2124:
2119:
1177:
Ernest C. Crocker (1922). "Application Of The Octet Theory To Single-Ring
Aromatic Compounds".
628:
432:
294:
233:
80:
83:
in 1855. There is no general relationship between aromaticity as a chemical property and the
5346:
4932:
4887:
4602:
4572:
4542:
4477:
4457:
4372:
4367:
4332:
4287:
4272:
4267:
4247:
4237:
4172:
4162:
4092:
4042:
3562:
3365:
2941:
2896:
2726:
2716:
2462:
2387:
2182:
2144:
1929:
1890:
1506:
1484:
1376:
1330:
1281:
904:
790:
640:
443:
144:
123:
95:
48:
2392:
1924:
869:
5115:
5065:
5015:
4995:
4985:
4842:
4817:
4532:
4522:
4407:
4222:
4217:
4147:
3932:
3732:
3692:
3622:
3587:
3542:
3509:
3375:
3350:
3330:
3151:
3111:
3071:
3036:
2966:
2721:
2591:
2566:
2104:
1848:
1739:
1602:
1561:
966:
941:
770:
dianion (10e). Aromatic properties have been attributed to non-benzenoid compounds such as
695:
547:
454:
402:. That is, 4n + 2 number of π electrons, where n=0, 1, 2, 3, and so on. This is known as
376:
317:
8:
5321:
5306:
4952:
4927:
4912:
4907:
4637:
4592:
4577:
4467:
4447:
4342:
4227:
4212:
4057:
4002:
3992:
3957:
3722:
3597:
3572:
3484:
3340:
3325:
3310:
3131:
3076:
2846:
2696:
2641:
2512:
2427:
2287:
2212:
1750:
1614:
1580:
355:
in 1931. He was the first to separate the bonding electrons into sigma and pi electrons.
229:
5331:
3982:
3166:
2357:
1914:
403:
282:
An explanation for the exceptional stability of benzene is conventionally attributed to
182:. Double bonds consist of a σ-bond and a π-bond. The π-bonds are formed from overlap of
5070:
5020:
4990:
4852:
4642:
4432:
4317:
4252:
4242:
4007:
3937:
3902:
3897:
3877:
3872:
3817:
3727:
3577:
3439:
3429:
3335:
3121:
3066:
2996:
2916:
2811:
2711:
2646:
2571:
2417:
2282:
2217:
2202:
1669:
348:
245:
4807:
4127:
4012:
3977:
3942:
3887:
3842:
3757:
3737:
3687:
3682:
3652:
3637:
3547:
3454:
3390:
3355:
3181:
3056:
2931:
2856:
2836:
2751:
2586:
2581:
2527:
2437:
2342:
2302:
2257:
2139:
2134:
2099:
2025:
1984:
1900:
1689:
1649:
1639:
1413:
1356:
1318:
1179:
1043:
863:
767:
719:
616:
466:
321:
198:
64:
56:
3802:
5341:
5186:
5156:
5100:
5025:
4957:
4712:
4662:
4507:
4312:
4087:
4082:
4027:
4017:
3792:
3602:
3582:
3552:
3449:
3385:
3370:
3201:
3156:
3146:
3136:
3031:
3011:
3006:
2991:
2986:
2866:
2861:
2801:
2786:
2776:
2621:
2611:
2477:
2467:
2367:
2362:
2337:
2277:
2129:
2088:
2020:
2015:
2005:
1941:
1681:
1654:
1494:
1467:
1440:
1405:
1396:
Rzepa, Henry S. (2005). "A Double-Twist Möbius-Aromatic
Conformation of Annulene".
1364:
1310:
1269:
1241:
1214:
1187:
1159:
1132:
1103:
1072:
1063:
1035:
1008:
976:
883:
by 4n (n is an integer) electrons, is given a single half-twist to correspond to a
759:
723:
68:
2457:
293:
In fact, this concept can be traced further back, via Ernest
Crocker in 1922, to
269:
5251:
4942:
4777:
4772:
4067:
4052:
3997:
3952:
3912:
3862:
3827:
3822:
3767:
3762:
3697:
3647:
3567:
3395:
3279:
3254:
3216:
3191:
3176:
3161:
3096:
2971:
2921:
2911:
2891:
2851:
2661:
2651:
2636:
2432:
2352:
2222:
2192:
2177:
2172:
2040:
1919:
1793:
1664:
951:
876:
782:
727:
461:
248:, and Hofmann may not have been making a distinction between the two categories.
1952:
1060:
884:
5256:
5166:
5105:
4197:
4107:
4077:
3852:
3707:
3444:
3221:
3091:
2906:
2876:
2576:
2472:
2247:
2109:
1828:
1629:
971:
946:
805:
503:
each serve as one of the 20 basic building-blocks of proteins. Further, all 5
475:
470:
411:
183:
91:
43:
31:
2056:
5369:
5266:
4967:
4812:
4707:
4502:
3892:
3857:
3847:
3782:
3772:
3662:
3499:
3315:
3026:
3001:
2871:
2517:
2502:
2487:
2382:
2312:
2292:
2207:
2000:
1877:
1837:
1770:
1724:
1699:
1572:
1553:
1136:
916:
817:
809:
774:. Aromatic properties are tested to the limit in a class of compounds called
755:
680:
676:
492:
384:
368:
276:
107:
76:
527:) that make up the sequence of the genetic code in DNA and RNA are aromatic
4307:
3667:
3419:
3196:
2796:
2596:
2447:
2442:
2307:
2162:
2035:
1823:
1417:
1322:
1273:
1245:
1047:
1012:
896:
880:
707:
352:
2806:
2452:
2422:
1300:
1218:
1163:
1123:
August Kekulé (1872). "Ueber einige Condensationsproducte des Aldehyds".
892:
813:
801:
797:
735:
699:
540:
532:
399:
380:
161:
157:
153:
1471:
1444:
1314:
1191:
5090:
4617:
3967:
1205:
Henry Edward Armstrong (1890). "The structure of cycloid hydrocarbon".
775:
703:
504:
496:
333:
179:
51:
forms of benzene (top) combine to produce an average structure (bottom)
1498:
1409:
1368:
1108:
1091:
1076:
1039:
383:
system, most commonly an arrangement of alternating single and double
1644:
1619:
928:
763:
672:
660:
585:
488:
453:
Aromatic molecules are able to interact with each other in so-called
237:
84:
72:
35:
167:
A better representation is that of the circular π bond (Armstrong's
2497:
2167:
1718:
833:
664:
656:
652:
612:
516:
500:
389:
351:
origins of this stability, or aromaticity, were first modelled by
309:
241:
99:
2157:
1522:
872:
occurs when a cyclic system of molecular orbitals, formed from p
771:
731:
668:
608:
581:
573:
555:
551:
528:
520:
512:
508:
460:
Planar monocyclic molecules containing 4n π electrons are called
336:'s notation. It is argued that he also anticipated the nature of
265:
172:
171:), in which the electron density is evenly distributed through a
111:
410:
Whereas benzene is aromatic (6 electrons, from 3 double bonds),
577:
566:
559:
524:
358:
226:
1026:
Schleyer, Paul von Ragué (2001). "Introduction: Aromaticity".
1430:
866:
is believed to exist in certain metal clusters of aluminium.
648:
589:
416:
90:
Aromaticity can also be considered a manifestation of cyclic
30:"Aromatic" redirects here. For meanings related to odor, see
442:
The circulating π electrons in an aromatic molecule produce
392:
structure, with all the contributing atoms in the same plane
919:
the ring bonds are extended with alkyne and allene groups.
536:
364:
337:
103:
1730:
286:, who was apparently the first (in 1925) to coin the term
751:
739:
447:
796:
When carbon in benzene is replaced by other elements in
1204:
435:
reactions as happens with carbon-carbon double bonds.
225:
term — namely, to apply to compounds that contain the
122:
section below). The model for benzene consists of two
290:
as a group of six electrons that resists disruption.
63:
is a chemical property describing the way in which a
5232:
Erlenmeyer–Plöchl azlactone and amino-acid synthesis
860:
was experimentally evidenced by Li solid state NMR.
251:
546:Aromatic compounds are important in industry. Key
482:
1231:
651:) increase its reactivity. Other examples include
474:formerly non-bonding molecular orbitals, which by
102:are free to cycle around circular arrangements of
4293:Divinylcyclopropane-cycloheptadiene rearrangement
1982:
1457:
1176:
539:contains an aromatic system with 22 π electrons.
5367:
395:Contributing atoms arranged in one or more rings
324:of the ring is broken. He introduced the symbol
260:Historic benzene formulae as proposed by Kekulé.
221:The first known use of the word "aromatic" as a
216:
2086:
4553:Thermal rearrangement of aromatic hydrocarbons
3187:Thermal rearrangement of aromatic hydrocarbons
745:
595:
5282:Lectka enantioselective beta-lactam synthesis
2542:
2072:
1968:
1538:
1152:Journal of the Chemical Society, Transactions
1149:
1122:
998:
994:
992:
785:where conjugation is interrupted by a single
5061:Inverse electron-demand Diels–Alder reaction
2882:Heterogeneous metal catalyzed cross-coupling
1511:: CS1 maint: multiple names: authors list (
1381:: CS1 maint: multiple names: authors list (
1335:: CS1 maint: multiple names: authors list (
1286:: CS1 maint: multiple names: authors list (
1207:Proceedings of the Chemical Society (London)
1116:
627:where n ≥ 4 and is an even number, such as
398:A number of π delocalized electrons that is
359:Characteristics of aromatic (aryl) compounds
328:centered on the ring as a shorthand for the
4403:Lobry de Bruyn–Van Ekenstein transformation
1092:"Introduction: Delocalization Pi and Sigma"
999:A. W. Hofmann (1855). "On Insolinic Acid".
98:. This is usually considered to be because
34:. For the lack of romantic attraction, see
2079:
2065:
1975:
1961:
1545:
1531:
989:
781:A special case of aromaticity is found in
446:that oppose the applied magnetic field in
4893:Petrenko-Kritschenko piperidone synthesis
4348:Fritsch–Buttenberg–Wiechell rearrangement
1107:
5056:Intramolecular Diels–Alder cycloaddition
1937:Polyhedral skeletal electron pair theory
1460:Journal of the American Chemical Society
1433:Journal of the American Chemical Society
1089:
1025:
713:
255:
106:that are alternately single- and double-
42:
891:for details). Because the twist can be
14:
5368:
5076:Metal-centered cycloaddition reactions
4728:Debus–Radziszewski imidazole synthesis
2672:Bodroux–Chichibabin aldehyde synthesis
899:, the resulting Möbius aromatics are
603:
5222:Diazoalkane 1,3-dipolar cycloaddition
5126:Vinylcyclopropane (5+2) cycloaddition
5031:Diazoalkane 1,3-dipolar cycloaddition
4803:Hurd–Mori 1,2,3-thiadiazole synthesis
4298:Dowd–Beckwith ring-expansion reaction
3465:Hurd–Mori 1,2,3-thiadiazole synthesis
2541:
2378:LFER solvent coefficients (data page)
2060:
1956:
1526:
1395:
852:rings occurring in the Zintl phase Li
371:atoms with specific characteristics:
4033:Sharpless asymmetric dihydroxylation
3270:Methoxymethylenetriphenylphosphorane
911:topologies were first suggested by
543:also has a similar aromatic system.
4158:Allen–Millar–Trippett rearrangement
425:electrophilic aromatic substitution
314:electrophilic aromatic substitution
24:
5297:Nitrone-olefin (3+2) cycloaddition
5292:Niementowski quinazoline synthesis
5081:Nitrone-olefin (3+2) cycloaddition
5006:Azide-alkyne Huisgen cycloaddition
4868:Niementowski quinazoline synthesis
4623:Azide-alkyne Huisgen cycloaddition
3928:Meerwein–Ponndorf–Verley reduction
3480:Leimgruber–Batcho indole synthesis
1552:
429:nucleophilic aromatic substitution
134:
25:
5392:
5121:Trimethylenemethane cycloaddition
4823:Johnson–Corey–Chaykovsky reaction
4688:Cadogan–Sundberg indole synthesis
4668:Bohlmann–Rahtz pyridine synthesis
4628:Baeyer–Emmerling indole synthesis
3435:Cadogan–Sundberg indole synthesis
2927:Johnson–Corey–Chaykovsky reaction
1487:The Journal of Physical Chemistry
1090:Schleyer, Paul von Ragué (2005).
316:, proceeding (third) through a
252:The structure of the benzene ring
156:, intermediate between that of a
5217:Cook–Heilbron thiazole synthesis
5046:Hexadehydro Diels–Alder reaction
4873:Niementowski quinoline synthesis
4703:Cook–Heilbron thiazole synthesis
4648:Bischler–Möhlau indole synthesis
4558:Tiffeneau–Demjanov rearrangement
4188:Baker–Venkataraman rearrangement
3346:Horner–Wadsworth–Emmons reaction
3017:Mizoroki-Heck vs. Reductive Heck
2902:Horner–Wadsworth–Emmons reaction
2413:Neighbouring group participation
1729:
1723:
1717:
1001:Proceedings of the Royal Society
922:
692:Polycyclic aromatic hydrocarbons
634:
483:Importance of aromatic compounds
204:
189:
4753:Fiesselmann thiophene synthesis
4583:Westphalen–Lettré rearrangement
4563:Vinylcyclopropane rearrangement
4393:Kornblum–DeLaMare rearrangement
4038:Epoxidation of allylic alcohols
3948:Noyori asymmetric hydrogenation
3883:Kornblum–DeLaMare rearrangement
3558:Gallagher–Hollander degradation
1478:
1451:
1424:
1389:
1343:
1294:
1252:
5212:Chichibabin pyridine synthesis
4698:Chichibabin pyridine synthesis
4658:Blum–Ittah aziridine synthesis
4493:Ring expansion and contraction
2762:Cross dehydrogenative coupling
1234:Chemistry – A European Journal
1225:
1198:
1170:
1143:
1083:
1054:
1019:
722:are aromatic rings with other
686:
87:properties of such compounds.
13:
1:
5182:Bischler–Napieralski reaction
5140:Heterocycle forming reactions
4793:Hemetsberger indole synthesis
4653:Bischler–Napieralski reaction
4568:Wagner–Meerwein rearrangement
4538:Sommelet–Hauser rearrangement
4518:Seyferth–Gilbert homologation
4383:Ireland–Claisen rearrangement
4378:Hofmann–Martius rearrangement
4138:2,3-sigmatropic rearrangement
3753:Corey–Winter olefin synthesis
3678:Barton–McCombie deoxygenation
3321:Corey–Winter olefin synthesis
3275:Seyferth–Gilbert homologation
3142:Seyferth–Gilbert homologation
982:
400:even, but not a multiple of 4
367:) compound contains a set of
5287:Lehmstedt–Tanasescu reaction
5247:Gabriel–Colman rearrangement
5202:Bucherer carbazole synthesis
5197:Borsche–Drechsel cyclization
5177:Bernthsen acridine synthesis
5162:Bamberger triazine synthesis
5147:Algar–Flynn–Oyamada reaction
4858:Nazarov cyclization reaction
4723:De Kimpe aziridine synthesis
4678:Bucherer carbazole synthesis
4673:Borsche–Drechsel cyclization
4443:Nazarov cyclization reaction
4423:Meyer–Schuster rearrangement
4353:Gabriel–Colman rearrangement
4103:Wolffenstein–Böters reaction
3988:Reduction of nitro compounds
3838:Grundmann aldehyde synthesis
3643:Algar–Flynn–Oyamada reaction
3052:Olefin conversion technology
3047:Nozaki–Hiyama–Kishi reaction
2842:Gabriel–Colman rearrangement
2732:Claisen-Schmidt condensation
2677:Bouveault aldehyde synthesis
726:attached. Examples include
619:excepted) with the formula C
119:
7:
5262:Hantzsch pyridine synthesis
5041:Enone–alkene cycloadditions
4863:Nenitzescu indole synthesis
4783:Hantzsch pyridine synthesis
4748:Ferrario–Ackermann reaction
4398:Kowalski ester homologation
4363:Halogen dance rearrangement
4208:Benzilic acid rearrangement
3633:Akabori amino-acid reaction
3593:Von Braun amide degradation
3538:Barbier–Wieland degradation
3490:Nenitzescu indole synthesis
3470:Kharasch–Sosnovsky reaction
3361:Julia–Kocienski olefination
3265:Kowalski ester homologation
2962:Kowalski ester homologation
2937:Julia–Kocienski olefination
2692:Cadiot–Chodkiewicz coupling
2617:Aza-Baylis–Hillman reaction
2562:Acetoacetic ester synthesis
2273:Dynamic binding (chemistry)
2263:Conrotatory and disrotatory
2238:Charge remote fragmentation
935:
746:Atypical aromatic compounds
596:Types of aromatic compounds
550:of commercial interest are
139:Modern depiction of benzene
10:
5397:
5381:Physical organic chemistry
5327:Robinson–Gabriel synthesis
5277:Kröhnke pyridine synthesis
5111:Retro-Diels–Alder reaction
5051:Imine Diels–Alder reaction
4838:Kröhnke pyridine synthesis
4453:Newman–Kwart rearrangement
4428:Mislow–Evans rearrangement
4338:Fischer–Hepp rearrangement
4283:Di-π-methane rearrangement
4063:Stephen aldehyde synthesis
3798:Eschweiler–Clarke reaction
3515:Williamson ether synthesis
2832:Fujiwara–Moritani reaction
2737:Combes quinoline synthesis
2702:Carbonyl olefin metathesis
2403:More O'Ferrall–Jencks plot
2328:Grunwald–Winstein equation
2298:Electron-withdrawing group
2233:Catalytic resonance theory
1635:Metal–ligand multiple bond
232:— occurs in an article by
211:
29:
5337:Urech hydantoin synthesis
5317:Pomeranz–Fritsch reaction
5242:Fischer oxazole synthesis
5139:
4976:1,3-Dipolar cycloaddition
4966:
4948:Urech hydantoin synthesis
4918:Reissert indole synthesis
4903:Pomeranz–Fritsch reaction
4833:Knorr quinoline synthesis
4763:Fischer oxazole synthesis
4693:Camps quinoline synthesis
4613:1,3-Dipolar cycloaddition
4601:
4513:Semipinacol rearrangement
4488:Ramberg–Bäcklund reaction
4473:Piancatelli rearrangement
4413:McFadyen–Stevens reaction
4168:Alpha-ketol rearrangement
4116:
3923:McFadyen–Stevens reaction
3868:Kiliani–Fischer synthesis
3788:Elbs persulfate oxidation
3713:Bouveault–Blanc reduction
3673:Baeyer–Villiger oxidation
3611:
3528:
3505:Schotten–Baumann reaction
3408:
3381:Ramberg–Bäcklund reaction
3288:
3260:Kiliani–Fischer synthesis
3240:
3102:Ramberg–Bäcklund reaction
3087:Pinacol coupling reaction
3082:Piancatelli rearrangement
2977:Liebeskind–Srogl coupling
2827:Fujimoto–Belleau reaction
2550:
2544:List of organic reactions
2408:Negative hyperconjugation
2153:
2095:
2046:List of organic compounds
1991:
1899:
1876:
1807:
1769:
1749:
1738:
1715:
1698:
1680:
1571:
1560:
738:, and the nucleotides of
320:, in which (fourth) the
130:
5312:Pictet–Spengler reaction
5227:Einhorn–Brunner reaction
5192:Boger pyridine synthesis
5086:Oxo-Diels–Alder reaction
5001:Aza-Diels–Alder reaction
4898:Pictet–Spengler reaction
4798:Hofmann–Löffler reaction
4788:Hegedus indole synthesis
4758:Fischer indole synthesis
4633:Bartoli indole synthesis
4588:Willgerodt rearrangement
4418:McLafferty rearrangement
4328:Ferrier carbocyclization
4143:2,3-Wittig rearrangement
4133:1,2-Wittig rearrangement
3973:Parikh–Doering oxidation
3963:Oxygen rebound mechanism
3628:Adkins–Peterson reaction
3520:Yamaguchi esterification
3460:Hegedus indole synthesis
3425:Bartoli indole synthesis
3296:Bamford–Stevens reaction
3212:Weinreb ketone synthesis
3172:Stork enamine alkylation
2947:Knoevenagel condensation
2817:Ferrier carbocyclization
2707:Castro–Stephens coupling
2333:Hammett acidity function
2323:Free-energy relationship
2268:Curtin–Hammett principle
2253:Conformational isomerism
1137:10.1002/jlac.18721620110
758:cation (2e system), the
750:Aromaticity is found in
611:, as well as most other
5272:Knorr pyrrole synthesis
5207:Bucherer–Bergs reaction
5152:Allan–Robinson reaction
5131:Wagner-Jauregg reaction
4923:Ring-closing metathesis
4848:Larock indole synthesis
4828:Knorr pyrrole synthesis
4683:Bucherer–Bergs reaction
4548:Stieglitz rearrangement
4528:Skattebøl rearrangement
4498:Ring-closing metathesis
4358:Group transfer reaction
4323:Favorskii rearrangement
4263:Cornforth rearrangement
4193:Bamberger rearrangement
4098:Wolff–Kishner reduction
3918:Markó–Lam deoxygenation
3813:Fleming–Tamao oxidation
3808:Fischer–Tropsch process
3495:Oxymercuration reaction
3475:Knorr pyrrole synthesis
3301:Barton–Kellogg reaction
3207:Wagner-Jauregg reaction
3127:Ring-closing metathesis
3117:Reimer–Tiemann reaction
3107:Rauhut–Currier reaction
3022:Nef isocyanide reaction
2982:Malonic ester synthesis
2952:Knorr pyrrole synthesis
2887:High dilution principle
2822:Friedel–Crafts reaction
2757:Cross-coupling reaction
2682:Bucherer–Bergs reaction
2667:Blanc chloromethylation
2657:Blaise ketone synthesis
2632:Baylis–Hillman reaction
2627:Barton–Kellogg reaction
2602:Allan–Robinson reaction
2508:Woodward–Hoffmann rules
2243:Charge-transfer complex
762:anion (6e system), the
275:Between 1897 and 1906,
5237:Feist–Benary synthesis
5011:Bradsher cycloaddition
4981:4+4 Photocycloaddition
4938:Simmons–Smith reaction
4883:Paternò–Büchi reaction
4743:Feist–Benary synthesis
4733:Dieckmann condensation
4483:Pummerer rearrangement
4463:Oxy-Cope rearrangement
4438:Myers allene synthesis
4388:Jacobsen rearrangement
4303:Electrocyclic reaction
4278:Demjanov rearrangement
4233:Buchner ring expansion
4203:Beckmann rearrangement
4183:Aza-Cope rearrangement
4178:Arndt–Eistert reaction
4153:Alkyne zipper reaction
4073:Transfer hydrogenation
4048:Sharpless oxyamination
4023:Selenoxide elimination
3908:Lombardo methylenation
3833:Griesbaum coozonolysis
3743:Corey–Itsuno reduction
3718:Boyland–Sims oxidation
3658:Angeli–Rimini reaction
3306:Boord olefin synthesis
3250:Arndt–Eistert reaction
3242:Homologation reactions
3042:Nitro-Mannich reaction
2957:Kolbe–Schmitt reaction
2767:Cross-coupling partner
2687:Buchner ring expansion
2607:Arndt–Eistert reaction
2373:Kinetic isotope effect
2120:Rearrangement reaction
1274:10.1002/anie.201105081
1246:10.1002/chem.200400457
1013:10.1098/rspl.1856.0002
629:cyclotetradecaheptaene
433:electrophilic addition
295:Henry Edward Armstrong
268:was first proposed by
261:
234:August Wilhelm Hofmann
140:
81:August Wilhelm Hofmann
52:
5096:Pauson–Khand reaction
4933:Sharpless epoxidation
4888:Pechmann condensation
4768:Friedländer synthesis
4718:Davis–Beirut reaction
4573:Wallach rearrangement
4543:Stevens rearrangement
4478:Pinacol rearrangement
4458:Overman rearrangement
4373:Hofmann rearrangement
4368:Hayashi rearrangement
4333:Ferrier rearrangement
4288:Dimroth rearrangement
4273:Curtius rearrangement
4268:Criegee rearrangement
4248:Claisen rearrangement
4238:Carroll rearrangement
4173:Amadori rearrangement
4163:Allylic rearrangement
4043:Sharpless epoxidation
3778:Dess–Martin oxidation
3703:Bohn–Schmidt reaction
3563:Hofmann rearrangement
3366:Kauffmann olefination
3289:Olefination reactions
3227:Wurtz–Fittig reaction
3062:Palladium–NHC complex
2942:Kauffmann olefination
2897:Homologation reaction
2747:Corey–House synthesis
2727:Claisen rearrangement
2523:Yukawa–Tsuno equation
2483:Swain–Lupton equation
2463:Spherical aromaticity
2398:Möbius–Hückel concept
2183:Aromatic ring current
2145:Substitution reaction
889:Möbius–Hückel concept
714:Substituted aromatics
696:simple aromatic rings
548:aromatic hydrocarbons
259:
138:
46:
5302:Paal–Knorr synthesis
5172:Barton–Zard reaction
5116:Staudinger synthesis
5066:Ketene cycloaddition
5036:Diels–Alder reaction
5016:Cheletropic reaction
4996:Alkyne trimerisation
4878:Paal–Knorr synthesis
4843:Kulinkovich reaction
4818:Jacobsen epoxidation
4738:Diels–Alder reaction
4533:Smiles rearrangement
4523:Sigmatropic reaction
4408:Lossen rearrangement
4258:Corey–Fuchs reaction
4223:Boekelheide reaction
4218:Bergmann degradation
4148:Achmatowicz reaction
3933:Methionine sulfoxide
3733:Clemmensen reduction
3693:Bergmann degradation
3623:Acyloin condensation
3588:Strecker degradation
3543:Bergmann degradation
3510:Ullmann condensation
3376:Peterson olefination
3351:Hydrazone iodination
3331:Elimination reaction
3232:Zincke–Suhl reaction
3152:Sonogashira coupling
3112:Reformatsky reaction
3072:Peterson olefination
3037:Nierenstein reaction
2967:Kulinkovich reaction
2782:Diels–Alder reaction
2742:Corey–Fuchs reaction
2722:Claisen condensation
2592:Alkyne trimerisation
2567:Acyloin condensation
2533:Σ-bishomoaromaticity
2493:Thorpe–Ingold effect
2105:Elimination reaction
1625:Coordinate (dipolar)
1352:Paul von R. Schleyer
1262:Angew. Chem. Int. Ed
1219:10.1039/PL8900600095
1164:10.1039/CT9252701604
967:Simple aromatic ring
942:Aromatic hydrocarbon
732:acetylsalicylic acid
332:, thus anticipating
318:Wheland intermediate
5322:Prilezhaev reaction
5307:Pellizzari reaction
4986:(4+3) cycloaddition
4953:Van Leusen reaction
4928:Robinson annulation
4913:Pschorr cyclization
4908:Prilezhaev reaction
4638:Bergman cyclization
4593:Wolff rearrangement
4578:Weerman degradation
4468:Pericyclic reaction
4448:Neber rearrangement
4343:Fries rearrangement
4228:Brook rearrangement
4213:Bergman cyclization
4058:Staudinger reaction
4003:Rosenmund reduction
3993:Reductive amination
3958:Oppenauer oxidation
3748:Corey–Kim oxidation
3723:Cannizzaro reaction
3598:Weerman degradation
3573:Isosaccharinic acid
3485:Mukaiyama hydration
3341:Hofmann elimination
3326:Dehydrohalogenation
3311:Chugaev elimination
3132:Robinson annulation
3077:Pfitzinger reaction
2847:Gattermann reaction
2792:Wulff–Dötz reaction
2772:Dakin–West reaction
2697:Carbonyl allylation
2642:Bergman cyclization
2428:Kennedy J. P. Orton
2348:Hammond's postulate
2318:Flippin–Lodge angle
2288:Electromeric effect
2213:Beta-silicon effect
2198:Baker–Nathan effect
1799:C–H···O interaction
1581:Electron deficiency
1472:10.1021/ja00167a011
1445:10.1021/ja00059a035
1315:10.1038/nature02224
1192:10.1021/ja01429a002
879:and populated in a
604:Neutral homocyclics
431:reactions, but not
284:Sir Robert Robinson
246:aliphatic compounds
217:The term "aromatic"
143:As is standard for
5376:Aromatic compounds
5071:McCormack reaction
5021:Conia-ene reaction
4853:Madelung synthesis
4643:Biginelli reaction
4433:Mumm rearrangement
4318:Favorskii reaction
4253:Cope rearrangement
4243:Chan rearrangement
4008:Rubottom oxidation
3938:Miyaura borylation
3903:Lipid peroxidation
3898:Lindgren oxidation
3878:Kornblum oxidation
3873:Kolbe electrolysis
3818:Fukuyama reduction
3728:Carbonyl reduction
3578:Marker degradation
3440:Diazonium compound
3430:Boudouard reaction
3409:Carbon-heteroatom
3336:Grieco elimination
3122:Rieche formylation
3067:Passerini reaction
2997:Meerwein arylation
2917:Hydroxymethylation
2812:Favorskii reaction
2712:Chan rearrangement
2647:Biginelli reaction
2572:Aldol condensation
2418:2-Norbornyl cation
2393:Möbius aromaticity
2388:Markovnikov's rule
2283:Effective molarity
2228:Bürgi–Dunitz angle
2218:Bicycloaromaticity
1784:Resonance-assisted
870:Möbius aromaticity
766:ion (6e), and the
720:chemical compounds
423:compounds undergo
349:quantum mechanical
262:
145:resonance diagrams
141:
53:
5363:
5362:
5359:
5358:
5355:
5354:
5347:Wohl–Aue reaction
4991:6+4 Cycloaddition
4808:Iodolactonization
4128:1,2-rearrangement
4093:Wohl–Aue reaction
4013:Sabatier reaction
3978:Pinnick oxidation
3943:Mozingo reduction
3888:Leuckart reaction
3843:Haloform reaction
3758:Criegee oxidation
3738:Collins oxidation
3688:Benkeser reaction
3683:Bechamp reduction
3653:Andrussow process
3638:Alcohol oxidation
3548:Edman degradation
3455:Haloform reaction
3404:
3403:
3391:Takai olefination
3356:Julia olefination
3182:Takai olefination
3057:Olefin metathesis
2932:Julia olefination
2857:Grignard reaction
2837:Fukuyama coupling
2752:Coupling reaction
2717:Chan–Lam coupling
2587:Alkyne metathesis
2582:Alkane metathesis
2438:Phosphaethynolate
2343:George S. Hammond
2303:Electronic effect
2258:Conjugated system
2140:Stereospecificity
2135:Stereoselectivity
2100:Addition reaction
2089:organic reactions
2054:
2053:
2026:Organic synthesis
2021:Organic reactions
2016:Organic compounds
2006:Functional groups
1985:organic chemistry
1950:
1949:
1901:Electron counting
1872:
1871:
1761:London dispersion
1713:
1712:
1690:Metal aromaticity
1499:10.1021/jp960311p
1410:10.1021/ol0518333
1369:10.1021/ja0458165
1357:J. Am. Chem. Soc.
1180:J. Am. Chem. Soc.
1125:Liebigs Ann. Chem
1109:10.1021/cr030095y
1077:10.1021/cr0300946
1071:(10): 3436–3447.
1040:10.1021/cr0103221
864:Metal aromaticity
768:cyclooctatetraene
724:functional groups
617:cyclodecapentaene
467:cyclooctatetraene
199:molecular orbital
184:atomic p-orbitals
69:unsaturated bonds
57:organic chemistry
27:Chemical property
16:(Redirected from
5388:
5342:Wenker synthesis
5332:Stollé synthesis
5187:Bobbitt reaction
5157:Auwers synthesis
5101:Povarov reaction
5026:Cyclopropanation
4964:
4963:
4958:Wenker synthesis
4713:Darzens reaction
4663:Bobbitt reaction
4508:Schmidt reaction
4313:Enyne metathesis
4088:Whiting reaction
4083:Wharton reaction
4028:Shapiro reaction
4018:Sarett oxidation
3983:Prévost reaction
3793:Emde degradation
3603:Wohl degradation
3583:Ruff degradation
3553:Emde degradation
3450:Grignard reagent
3386:Shapiro reaction
3371:McMurry reaction
3238:
3237:
3202:Ullmann reaction
3167:Stollé synthesis
3157:Stetter reaction
3147:Shapiro reaction
3137:Sakurai reaction
3032:Negishi coupling
3012:Minisci reaction
3007:Michael reaction
2992:McMurry reaction
2987:Mannich reaction
2867:Hammick reaction
2862:Grignard reagent
2802:Enyne metathesis
2787:Doebner reaction
2777:Darzens reaction
2622:Barbier reaction
2612:Auwers synthesis
2539:
2538:
2513:Woodward's rules
2478:Superaromaticity
2468:Spiroaromaticity
2368:Inductive effect
2363:Hyperconjugation
2338:Hammett equation
2278:Edwards equation
2130:Regioselectivity
2081:
2074:
2067:
2058:
2057:
2001:Covalent bonding
1977:
1970:
1963:
1954:
1953:
1942:Jemmis mno rules
1794:Dihydrogen bonds
1747:
1746:
1733:
1727:
1721:
1655:Hyperconjugation
1569:
1568:
1547:
1540:
1533:
1524:
1523:
1517:
1516:
1510:
1502:
1482:
1476:
1475:
1455:
1449:
1448:
1428:
1422:
1421:
1393:
1387:
1386:
1380:
1372:
1363:(8): 2425–2432.
1347:
1341:
1340:
1334:
1326:
1309:(6968): 819–21.
1298:
1292:
1291:
1285:
1277:
1256:
1250:
1249:
1229:
1223:
1222:
1202:
1196:
1195:
1186:(8): 1618–1630.
1174:
1168:
1167:
1147:
1141:
1140:
1120:
1114:
1113:
1111:
1096:Chemical Reviews
1087:
1081:
1080:
1064:Chemical Reviews
1058:
1052:
1051:
1028:Chemical Reviews
1023:
1017:
1016:
996:
977:Avoided crossing
760:cyclopentadienyl
698:). Examples are
683:, for example).
535:. The molecule
369:covalently bound
363:An aromatic (or
208:
201:has π symmetry.
193:
21:
5396:
5395:
5391:
5390:
5389:
5387:
5386:
5385:
5366:
5365:
5364:
5351:
5252:Gewald reaction
5135:
4962:
4943:Skraup reaction
4778:Graham reaction
4773:Gewald reaction
4604:
4597:
4119:
4112:
4068:Swern oxidation
4053:Stahl oxidation
3998:Riley oxidation
3953:Omega oxidation
3913:Luche reduction
3863:Jones oxidation
3828:Glycol cleavage
3823:Ganem oxidation
3768:Davis oxidation
3763:Dakin oxidation
3698:Birch reduction
3648:Amide reduction
3614:
3607:
3568:Hooker reaction
3530:
3524:
3412:
3410:
3400:
3396:Wittig reaction
3284:
3280:Wittig reaction
3255:Hooker reaction
3236:
3217:Wittig reaction
3192:Thorpe reaction
3177:Suzuki reaction
3162:Stille reaction
3097:Quelet reaction
2972:Kumada coupling
2922:Ivanov reaction
2912:Hydrovinylation
2892:Hiyama coupling
2852:Glaser coupling
2662:Blaise reaction
2652:Bingel reaction
2637:Benary reaction
2554:
2552:
2546:
2537:
2433:Passive binding
2353:Homoaromaticity
2203:Baldwin's rules
2178:Antiaromaticity
2173:Anomeric effect
2149:
2091:
2085:
2055:
2050:
2041:Stereochemistry
1987:
1981:
1951:
1946:
1895:
1868:
1811:
1803:
1765:
1752:
1742:
1734:
1728:
1722:
1709:
1694:
1676:
1564:
1556:
1551:
1521:
1520:
1504:
1503:
1493:: 10928–10935.
1483:
1479:
1456:
1452:
1429:
1425:
1398:Organic Letters
1394:
1390:
1374:
1373:
1348:
1344:
1328:
1327:
1299:
1295:
1279:
1278:
1257:
1253:
1230:
1226:
1203:
1199:
1175:
1171:
1148:
1144:
1121:
1117:
1088:
1084:
1059:
1055:
1024:
1020:
997:
990:
985:
952:BTX (chemistry)
938:
925:
877:atomic orbitals
875:
859:
855:
851:
847:
843:
839:
831:
827:
822:Hexasilabenzene
783:homoaromaticity
748:
728:trinitrotoluene
716:
689:
637:
626:
622:
606:
598:
485:
361:
288:aromatic sextet
254:
219:
214:
133:
39:
28:
23:
22:
15:
12:
11:
5:
5394:
5384:
5383:
5378:
5361:
5360:
5357:
5356:
5353:
5352:
5350:
5349:
5344:
5339:
5334:
5329:
5324:
5319:
5314:
5309:
5304:
5299:
5294:
5289:
5284:
5279:
5274:
5269:
5264:
5259:
5257:Hantzsch ester
5254:
5249:
5244:
5239:
5234:
5229:
5224:
5219:
5214:
5209:
5204:
5199:
5194:
5189:
5184:
5179:
5174:
5169:
5167:Banert cascade
5164:
5159:
5154:
5149:
5143:
5141:
5137:
5136:
5134:
5133:
5128:
5123:
5118:
5113:
5108:
5106:Prato reaction
5103:
5098:
5093:
5088:
5083:
5078:
5073:
5068:
5063:
5058:
5053:
5048:
5043:
5038:
5033:
5028:
5023:
5018:
5013:
5008:
5003:
4998:
4993:
4988:
4983:
4978:
4972:
4970:
4961:
4960:
4955:
4950:
4945:
4940:
4935:
4930:
4925:
4920:
4915:
4910:
4905:
4900:
4895:
4890:
4885:
4880:
4875:
4870:
4865:
4860:
4855:
4850:
4845:
4840:
4835:
4830:
4825:
4820:
4815:
4810:
4805:
4800:
4795:
4790:
4785:
4780:
4775:
4770:
4765:
4760:
4755:
4750:
4745:
4740:
4735:
4730:
4725:
4720:
4715:
4710:
4705:
4700:
4695:
4690:
4685:
4680:
4675:
4670:
4665:
4660:
4655:
4650:
4645:
4640:
4635:
4630:
4625:
4620:
4615:
4609:
4607:
4599:
4598:
4596:
4595:
4590:
4585:
4580:
4575:
4570:
4565:
4560:
4555:
4550:
4545:
4540:
4535:
4530:
4525:
4520:
4515:
4510:
4505:
4500:
4495:
4490:
4485:
4480:
4475:
4470:
4465:
4460:
4455:
4450:
4445:
4440:
4435:
4430:
4425:
4420:
4415:
4410:
4405:
4400:
4395:
4390:
4385:
4380:
4375:
4370:
4365:
4360:
4355:
4350:
4345:
4340:
4335:
4330:
4325:
4320:
4315:
4310:
4305:
4300:
4295:
4290:
4285:
4280:
4275:
4270:
4265:
4260:
4255:
4250:
4245:
4240:
4235:
4230:
4225:
4220:
4215:
4210:
4205:
4200:
4198:Banert cascade
4195:
4190:
4185:
4180:
4175:
4170:
4165:
4160:
4155:
4150:
4145:
4140:
4135:
4130:
4124:
4122:
4118:Rearrangement
4114:
4113:
4111:
4110:
4108:Zinin reaction
4105:
4100:
4095:
4090:
4085:
4080:
4078:Wacker process
4075:
4070:
4065:
4060:
4055:
4050:
4045:
4040:
4035:
4030:
4025:
4020:
4015:
4010:
4005:
4000:
3995:
3990:
3985:
3980:
3975:
3970:
3965:
3960:
3955:
3950:
3945:
3940:
3935:
3930:
3925:
3920:
3915:
3910:
3905:
3900:
3895:
3890:
3885:
3880:
3875:
3870:
3865:
3860:
3855:
3853:Hydrogenolysis
3850:
3845:
3840:
3835:
3830:
3825:
3820:
3815:
3810:
3805:
3803:Étard reaction
3800:
3795:
3790:
3785:
3780:
3775:
3770:
3765:
3760:
3755:
3750:
3745:
3740:
3735:
3730:
3725:
3720:
3715:
3710:
3708:Bosch reaction
3705:
3700:
3695:
3690:
3685:
3680:
3675:
3670:
3665:
3660:
3655:
3650:
3645:
3640:
3635:
3630:
3625:
3619:
3617:
3613:Organic redox
3609:
3608:
3606:
3605:
3600:
3595:
3590:
3585:
3580:
3575:
3570:
3565:
3560:
3555:
3550:
3545:
3540:
3534:
3532:
3526:
3525:
3523:
3522:
3517:
3512:
3507:
3502:
3497:
3492:
3487:
3482:
3477:
3472:
3467:
3462:
3457:
3452:
3447:
3445:Esterification
3442:
3437:
3432:
3427:
3422:
3416:
3414:
3406:
3405:
3402:
3401:
3399:
3398:
3393:
3388:
3383:
3378:
3373:
3368:
3363:
3358:
3353:
3348:
3343:
3338:
3333:
3328:
3323:
3318:
3313:
3308:
3303:
3298:
3292:
3290:
3286:
3285:
3283:
3282:
3277:
3272:
3267:
3262:
3257:
3252:
3246:
3244:
3235:
3234:
3229:
3224:
3222:Wurtz reaction
3219:
3214:
3209:
3204:
3199:
3194:
3189:
3184:
3179:
3174:
3169:
3164:
3159:
3154:
3149:
3144:
3139:
3134:
3129:
3124:
3119:
3114:
3109:
3104:
3099:
3094:
3092:Prins reaction
3089:
3084:
3079:
3074:
3069:
3064:
3059:
3054:
3049:
3044:
3039:
3034:
3029:
3024:
3019:
3014:
3009:
3004:
2999:
2994:
2989:
2984:
2979:
2974:
2969:
2964:
2959:
2954:
2949:
2944:
2939:
2934:
2929:
2924:
2919:
2914:
2909:
2907:Hydrocyanation
2904:
2899:
2894:
2889:
2884:
2879:
2877:Henry reaction
2874:
2869:
2864:
2859:
2854:
2849:
2844:
2839:
2834:
2829:
2824:
2819:
2814:
2809:
2804:
2799:
2794:
2789:
2784:
2779:
2774:
2769:
2764:
2759:
2754:
2749:
2744:
2739:
2734:
2729:
2724:
2719:
2714:
2709:
2704:
2699:
2694:
2689:
2684:
2679:
2674:
2669:
2664:
2659:
2654:
2649:
2644:
2639:
2634:
2629:
2624:
2619:
2614:
2609:
2604:
2599:
2594:
2589:
2584:
2579:
2577:Aldol reaction
2574:
2569:
2564:
2558:
2556:
2551:Carbon-carbon
2548:
2547:
2536:
2535:
2530:
2528:Zaitsev's rule
2525:
2520:
2515:
2510:
2505:
2500:
2495:
2490:
2485:
2480:
2475:
2473:Steric effects
2470:
2465:
2460:
2455:
2450:
2445:
2440:
2435:
2430:
2425:
2420:
2415:
2410:
2405:
2400:
2395:
2390:
2385:
2380:
2375:
2370:
2365:
2360:
2355:
2350:
2345:
2340:
2335:
2330:
2325:
2320:
2315:
2310:
2305:
2300:
2295:
2290:
2285:
2280:
2275:
2270:
2265:
2260:
2255:
2250:
2245:
2240:
2235:
2230:
2225:
2220:
2215:
2210:
2205:
2200:
2195:
2190:
2185:
2180:
2175:
2170:
2165:
2160:
2154:
2151:
2150:
2148:
2147:
2142:
2137:
2132:
2127:
2125:Redox reaction
2122:
2117:
2112:
2110:Polymerization
2107:
2102:
2096:
2093:
2092:
2084:
2083:
2076:
2069:
2061:
2052:
2051:
2049:
2048:
2043:
2038:
2033:
2028:
2023:
2018:
2013:
2008:
2003:
1998:
1992:
1989:
1988:
1980:
1979:
1972:
1965:
1957:
1948:
1947:
1945:
1944:
1939:
1934:
1933:
1932:
1927:
1922:
1917:
1906:
1904:
1897:
1896:
1894:
1893:
1888:
1882:
1880:
1874:
1873:
1870:
1869:
1867:
1866:
1861:
1856:
1851:
1846:
1841:
1831:
1826:
1821:
1815:
1813:
1805:
1804:
1802:
1801:
1796:
1791:
1786:
1781:
1775:
1773:
1767:
1766:
1764:
1763:
1757:
1755:
1744:
1740:Intermolecular
1736:
1735:
1716:
1714:
1711:
1710:
1708:
1707:
1704:
1702:
1696:
1695:
1693:
1692:
1686:
1684:
1678:
1677:
1675:
1674:
1673:
1672:
1667:
1657:
1652:
1647:
1642:
1637:
1632:
1627:
1622:
1617:
1612:
1611:
1610:
1600:
1599:
1598:
1593:
1588:
1577:
1575:
1566:
1562:Intramolecular
1558:
1557:
1554:Chemical bonds
1550:
1549:
1542:
1535:
1527:
1519:
1518:
1477:
1450:
1423:
1404:(21): 4637–9.
1388:
1342:
1293:
1251:
1224:
1213:(85): 95–106.
1197:
1169:
1142:
1115:
1082:
1053:
1018:
987:
986:
984:
981:
980:
979:
974:
972:Pi interaction
969:
964:
959:
954:
949:
947:Aromatic amine
944:
937:
934:
924:
921:
913:Johann Listing
873:
857:
853:
849:
845:
841:
837:
829:
825:
818:pyrylium salts
806:germanabenzene
747:
744:
715:
712:
688:
685:
636:
633:
624:
620:
605:
602:
597:
594:
484:
481:
471:cyclobutadiene
412:cyclobutadiene
408:
407:
396:
393:
387:
360:
357:
338:wave mechanics
253:
250:
218:
215:
213:
210:
160:and that of a
132:
129:
92:delocalization
77:empty orbitals
47:Two different
32:aroma compound
26:
9:
6:
4:
3:
2:
5393:
5382:
5379:
5377:
5374:
5373:
5371:
5348:
5345:
5343:
5340:
5338:
5335:
5333:
5330:
5328:
5325:
5323:
5320:
5318:
5315:
5313:
5310:
5308:
5305:
5303:
5300:
5298:
5295:
5293:
5290:
5288:
5285:
5283:
5280:
5278:
5275:
5273:
5270:
5268:
5267:Herz reaction
5265:
5263:
5260:
5258:
5255:
5253:
5250:
5248:
5245:
5243:
5240:
5238:
5235:
5233:
5230:
5228:
5225:
5223:
5220:
5218:
5215:
5213:
5210:
5208:
5205:
5203:
5200:
5198:
5195:
5193:
5190:
5188:
5185:
5183:
5180:
5178:
5175:
5173:
5170:
5168:
5165:
5163:
5160:
5158:
5155:
5153:
5150:
5148:
5145:
5144:
5142:
5138:
5132:
5129:
5127:
5124:
5122:
5119:
5117:
5114:
5112:
5109:
5107:
5104:
5102:
5099:
5097:
5094:
5092:
5089:
5087:
5084:
5082:
5079:
5077:
5074:
5072:
5069:
5067:
5064:
5062:
5059:
5057:
5054:
5052:
5049:
5047:
5044:
5042:
5039:
5037:
5034:
5032:
5029:
5027:
5024:
5022:
5019:
5017:
5014:
5012:
5009:
5007:
5004:
5002:
4999:
4997:
4994:
4992:
4989:
4987:
4984:
4982:
4979:
4977:
4974:
4973:
4971:
4969:
4968:Cycloaddition
4965:
4959:
4956:
4954:
4951:
4949:
4946:
4944:
4941:
4939:
4936:
4934:
4931:
4929:
4926:
4924:
4921:
4919:
4916:
4914:
4911:
4909:
4906:
4904:
4901:
4899:
4896:
4894:
4891:
4889:
4886:
4884:
4881:
4879:
4876:
4874:
4871:
4869:
4866:
4864:
4861:
4859:
4856:
4854:
4851:
4849:
4846:
4844:
4841:
4839:
4836:
4834:
4831:
4829:
4826:
4824:
4821:
4819:
4816:
4814:
4813:Isay reaction
4811:
4809:
4806:
4804:
4801:
4799:
4796:
4794:
4791:
4789:
4786:
4784:
4781:
4779:
4776:
4774:
4771:
4769:
4766:
4764:
4761:
4759:
4756:
4754:
4751:
4749:
4746:
4744:
4741:
4739:
4736:
4734:
4731:
4729:
4726:
4724:
4721:
4719:
4716:
4714:
4711:
4709:
4708:Cycloaddition
4706:
4704:
4701:
4699:
4696:
4694:
4691:
4689:
4686:
4684:
4681:
4679:
4676:
4674:
4671:
4669:
4666:
4664:
4661:
4659:
4656:
4654:
4651:
4649:
4646:
4644:
4641:
4639:
4636:
4634:
4631:
4629:
4626:
4624:
4621:
4619:
4616:
4614:
4611:
4610:
4608:
4606:
4603:Ring forming
4600:
4594:
4591:
4589:
4586:
4584:
4581:
4579:
4576:
4574:
4571:
4569:
4566:
4564:
4561:
4559:
4556:
4554:
4551:
4549:
4546:
4544:
4541:
4539:
4536:
4534:
4531:
4529:
4526:
4524:
4521:
4519:
4516:
4514:
4511:
4509:
4506:
4504:
4503:Rupe reaction
4501:
4499:
4496:
4494:
4491:
4489:
4486:
4484:
4481:
4479:
4476:
4474:
4471:
4469:
4466:
4464:
4461:
4459:
4456:
4454:
4451:
4449:
4446:
4444:
4441:
4439:
4436:
4434:
4431:
4429:
4426:
4424:
4421:
4419:
4416:
4414:
4411:
4409:
4406:
4404:
4401:
4399:
4396:
4394:
4391:
4389:
4386:
4384:
4381:
4379:
4376:
4374:
4371:
4369:
4366:
4364:
4361:
4359:
4356:
4354:
4351:
4349:
4346:
4344:
4341:
4339:
4336:
4334:
4331:
4329:
4326:
4324:
4321:
4319:
4316:
4314:
4311:
4309:
4306:
4304:
4301:
4299:
4296:
4294:
4291:
4289:
4286:
4284:
4281:
4279:
4276:
4274:
4271:
4269:
4266:
4264:
4261:
4259:
4256:
4254:
4251:
4249:
4246:
4244:
4241:
4239:
4236:
4234:
4231:
4229:
4226:
4224:
4221:
4219:
4216:
4214:
4211:
4209:
4206:
4204:
4201:
4199:
4196:
4194:
4191:
4189:
4186:
4184:
4181:
4179:
4176:
4174:
4171:
4169:
4166:
4164:
4161:
4159:
4156:
4154:
4151:
4149:
4146:
4144:
4141:
4139:
4136:
4134:
4131:
4129:
4126:
4125:
4123:
4121:
4115:
4109:
4106:
4104:
4101:
4099:
4096:
4094:
4091:
4089:
4086:
4084:
4081:
4079:
4076:
4074:
4071:
4069:
4066:
4064:
4061:
4059:
4056:
4054:
4051:
4049:
4046:
4044:
4041:
4039:
4036:
4034:
4031:
4029:
4026:
4024:
4021:
4019:
4016:
4014:
4011:
4009:
4006:
4004:
4001:
3999:
3996:
3994:
3991:
3989:
3986:
3984:
3981:
3979:
3976:
3974:
3971:
3969:
3966:
3964:
3961:
3959:
3956:
3954:
3951:
3949:
3946:
3944:
3941:
3939:
3936:
3934:
3931:
3929:
3926:
3924:
3921:
3919:
3916:
3914:
3911:
3909:
3906:
3904:
3901:
3899:
3896:
3894:
3893:Ley oxidation
3891:
3889:
3886:
3884:
3881:
3879:
3876:
3874:
3871:
3869:
3866:
3864:
3861:
3859:
3858:Hydroxylation
3856:
3854:
3851:
3849:
3848:Hydrogenation
3846:
3844:
3841:
3839:
3836:
3834:
3831:
3829:
3826:
3824:
3821:
3819:
3816:
3814:
3811:
3809:
3806:
3804:
3801:
3799:
3796:
3794:
3791:
3789:
3786:
3784:
3783:DNA oxidation
3781:
3779:
3776:
3774:
3773:Deoxygenation
3771:
3769:
3766:
3764:
3761:
3759:
3756:
3754:
3751:
3749:
3746:
3744:
3741:
3739:
3736:
3734:
3731:
3729:
3726:
3724:
3721:
3719:
3716:
3714:
3711:
3709:
3706:
3704:
3701:
3699:
3696:
3694:
3691:
3689:
3686:
3684:
3681:
3679:
3676:
3674:
3671:
3669:
3666:
3664:
3663:Aromatization
3661:
3659:
3656:
3654:
3651:
3649:
3646:
3644:
3641:
3639:
3636:
3634:
3631:
3629:
3626:
3624:
3621:
3620:
3618:
3616:
3610:
3604:
3601:
3599:
3596:
3594:
3591:
3589:
3586:
3584:
3581:
3579:
3576:
3574:
3571:
3569:
3566:
3564:
3561:
3559:
3556:
3554:
3551:
3549:
3546:
3544:
3541:
3539:
3536:
3535:
3533:
3527:
3521:
3518:
3516:
3513:
3511:
3508:
3506:
3503:
3501:
3500:Reed reaction
3498:
3496:
3493:
3491:
3488:
3486:
3483:
3481:
3478:
3476:
3473:
3471:
3468:
3466:
3463:
3461:
3458:
3456:
3453:
3451:
3448:
3446:
3443:
3441:
3438:
3436:
3433:
3431:
3428:
3426:
3423:
3421:
3418:
3417:
3415:
3411:bond forming
3407:
3397:
3394:
3392:
3389:
3387:
3384:
3382:
3379:
3377:
3374:
3372:
3369:
3367:
3364:
3362:
3359:
3357:
3354:
3352:
3349:
3347:
3344:
3342:
3339:
3337:
3334:
3332:
3329:
3327:
3324:
3322:
3319:
3317:
3316:Cope reaction
3314:
3312:
3309:
3307:
3304:
3302:
3299:
3297:
3294:
3293:
3291:
3287:
3281:
3278:
3276:
3273:
3271:
3268:
3266:
3263:
3261:
3258:
3256:
3253:
3251:
3248:
3247:
3245:
3243:
3239:
3233:
3230:
3228:
3225:
3223:
3220:
3218:
3215:
3213:
3210:
3208:
3205:
3203:
3200:
3198:
3195:
3193:
3190:
3188:
3185:
3183:
3180:
3178:
3175:
3173:
3170:
3168:
3165:
3163:
3160:
3158:
3155:
3153:
3150:
3148:
3145:
3143:
3140:
3138:
3135:
3133:
3130:
3128:
3125:
3123:
3120:
3118:
3115:
3113:
3110:
3108:
3105:
3103:
3100:
3098:
3095:
3093:
3090:
3088:
3085:
3083:
3080:
3078:
3075:
3073:
3070:
3068:
3065:
3063:
3060:
3058:
3055:
3053:
3050:
3048:
3045:
3043:
3040:
3038:
3035:
3033:
3030:
3028:
3027:Nef synthesis
3025:
3023:
3020:
3018:
3015:
3013:
3010:
3008:
3005:
3003:
3002:Methylenation
3000:
2998:
2995:
2993:
2990:
2988:
2985:
2983:
2980:
2978:
2975:
2973:
2970:
2968:
2965:
2963:
2960:
2958:
2955:
2953:
2950:
2948:
2945:
2943:
2940:
2938:
2935:
2933:
2930:
2928:
2925:
2923:
2920:
2918:
2915:
2913:
2910:
2908:
2905:
2903:
2900:
2898:
2895:
2893:
2890:
2888:
2885:
2883:
2880:
2878:
2875:
2873:
2872:Heck reaction
2870:
2868:
2865:
2863:
2860:
2858:
2855:
2853:
2850:
2848:
2845:
2843:
2840:
2838:
2835:
2833:
2830:
2828:
2825:
2823:
2820:
2818:
2815:
2813:
2810:
2808:
2805:
2803:
2800:
2798:
2795:
2793:
2790:
2788:
2785:
2783:
2780:
2778:
2775:
2773:
2770:
2768:
2765:
2763:
2760:
2758:
2755:
2753:
2750:
2748:
2745:
2743:
2740:
2738:
2735:
2733:
2730:
2728:
2725:
2723:
2720:
2718:
2715:
2713:
2710:
2708:
2705:
2703:
2700:
2698:
2695:
2693:
2690:
2688:
2685:
2683:
2680:
2678:
2675:
2673:
2670:
2668:
2665:
2663:
2660:
2658:
2655:
2653:
2650:
2648:
2645:
2643:
2640:
2638:
2635:
2633:
2630:
2628:
2625:
2623:
2620:
2618:
2615:
2613:
2610:
2608:
2605:
2603:
2600:
2598:
2595:
2593:
2590:
2588:
2585:
2583:
2580:
2578:
2575:
2573:
2570:
2568:
2565:
2563:
2560:
2559:
2557:
2553:bond forming
2549:
2545:
2540:
2534:
2531:
2529:
2526:
2524:
2521:
2519:
2518:Y-aromaticity
2516:
2514:
2511:
2509:
2506:
2504:
2503:Walsh diagram
2501:
2499:
2496:
2494:
2491:
2489:
2488:Taft equation
2486:
2484:
2481:
2479:
2476:
2474:
2471:
2469:
2466:
2464:
2461:
2459:
2458:Σ-aromaticity
2456:
2454:
2451:
2449:
2446:
2444:
2441:
2439:
2436:
2434:
2431:
2429:
2426:
2424:
2421:
2419:
2416:
2414:
2411:
2409:
2406:
2404:
2401:
2399:
2396:
2394:
2391:
2389:
2386:
2384:
2383:Marcus theory
2381:
2379:
2376:
2374:
2371:
2369:
2366:
2364:
2361:
2359:
2358:Hückel's rule
2356:
2354:
2351:
2349:
2346:
2344:
2341:
2339:
2336:
2334:
2331:
2329:
2326:
2324:
2321:
2319:
2316:
2314:
2313:Evelyn effect
2311:
2309:
2306:
2304:
2301:
2299:
2296:
2294:
2293:Electron-rich
2291:
2289:
2286:
2284:
2281:
2279:
2276:
2274:
2271:
2269:
2266:
2264:
2261:
2259:
2256:
2254:
2251:
2249:
2246:
2244:
2241:
2239:
2236:
2234:
2231:
2229:
2226:
2224:
2221:
2219:
2216:
2214:
2211:
2209:
2208:Bema Hapothle
2206:
2204:
2201:
2199:
2196:
2194:
2191:
2189:
2186:
2184:
2181:
2179:
2176:
2174:
2171:
2169:
2166:
2164:
2161:
2159:
2156:
2155:
2152:
2146:
2143:
2141:
2138:
2136:
2133:
2131:
2128:
2126:
2123:
2121:
2118:
2116:
2113:
2111:
2108:
2106:
2103:
2101:
2098:
2097:
2094:
2090:
2082:
2077:
2075:
2070:
2068:
2063:
2062:
2059:
2047:
2044:
2042:
2039:
2037:
2034:
2032:
2029:
2027:
2024:
2022:
2019:
2017:
2014:
2012:
2009:
2007:
2004:
2002:
1999:
1997:
1994:
1993:
1990:
1986:
1978:
1973:
1971:
1966:
1964:
1959:
1958:
1955:
1943:
1940:
1938:
1935:
1931:
1928:
1926:
1923:
1921:
1918:
1916:
1915:Hückel's rule
1913:
1912:
1911:
1908:
1907:
1905:
1902:
1898:
1892:
1889:
1887:
1884:
1883:
1881:
1879:
1878:Bond cleavage
1875:
1865:
1862:
1860:
1857:
1855:
1852:
1850:
1847:
1845:
1844:Intercalation
1842:
1839:
1835:
1834:Metallophilic
1832:
1830:
1827:
1825:
1822:
1820:
1817:
1816:
1814:
1810:
1806:
1800:
1797:
1795:
1792:
1790:
1787:
1785:
1782:
1780:
1777:
1776:
1774:
1772:
1768:
1762:
1759:
1758:
1756:
1754:
1751:Van der Waals
1748:
1745:
1741:
1737:
1732:
1726:
1720:
1706:
1705:
1703:
1701:
1697:
1691:
1688:
1687:
1685:
1683:
1679:
1671:
1668:
1666:
1663:
1662:
1661:
1658:
1656:
1653:
1651:
1648:
1646:
1643:
1641:
1638:
1636:
1633:
1631:
1628:
1626:
1623:
1621:
1618:
1616:
1613:
1609:
1606:
1605:
1604:
1601:
1597:
1594:
1592:
1589:
1587:
1584:
1583:
1582:
1579:
1578:
1576:
1574:
1570:
1567:
1563:
1559:
1555:
1548:
1543:
1541:
1536:
1534:
1529:
1528:
1525:
1514:
1508:
1500:
1496:
1492:
1488:
1481:
1473:
1469:
1466:: 4177–4182.
1465:
1461:
1454:
1446:
1442:
1439:: 2362–2372.
1438:
1434:
1427:
1419:
1415:
1411:
1407:
1403:
1399:
1392:
1384:
1378:
1370:
1366:
1362:
1359:
1358:
1353:
1346:
1338:
1332:
1324:
1320:
1316:
1312:
1308:
1304:
1297:
1289:
1283:
1275:
1271:
1268:(50): 12099.
1267:
1263:
1255:
1247:
1243:
1239:
1235:
1228:
1220:
1216:
1212:
1208:
1201:
1193:
1189:
1185:
1182:
1181:
1173:
1165:
1161:
1157:
1153:
1146:
1138:
1134:
1131:(1): 77–124.
1130:
1126:
1119:
1110:
1105:
1101:
1097:
1093:
1086:
1078:
1074:
1070:
1066:
1065:
1057:
1049:
1045:
1041:
1037:
1034:(5): 1115–8.
1033:
1029:
1022:
1014:
1010:
1006:
1002:
995:
993:
988:
978:
975:
973:
970:
968:
965:
963:
960:
958:
955:
953:
950:
948:
945:
943:
940:
939:
933:
930:
923:Y-aromaticity
920:
918:
917:carbo-benzene
914:
910:
906:
902:
898:
894:
890:
886:
882:
878:
871:
867:
865:
861:
835:
823:
819:
815:
811:
810:stannabenzene
807:
803:
799:
794:
793:carbon atom.
792:
788:
784:
779:
777:
773:
769:
765:
761:
757:
756:cyclopropenyl
754:as well: the
753:
743:
741:
737:
733:
729:
725:
721:
711:
709:
705:
701:
697:
693:
684:
682:
681:benzimidazole
678:
677:benzannulated
674:
670:
666:
662:
658:
654:
650:
646:
645:heteroaromats
642:
635:Heterocyclics
632:
630:
618:
614:
610:
601:
593:
591:
587:
583:
579:
575:
571:
569:
564:
562:
557:
553:
549:
544:
542:
538:
534:
530:
526:
522:
518:
514:
510:
506:
502:
498:
494:
493:phenylalanine
490:
480:
477:
472:
468:
463:
458:
456:
451:
449:
445:
444:ring currents
440:
436:
434:
430:
426:
420:
418:
413:
405:
404:Hückel's Rule
401:
397:
394:
391:
388:
386:
382:
378:
374:
373:
372:
370:
366:
356:
354:
350:
345:
343:
339:
335:
331:
327:
323:
319:
315:
311:
306:
304:
300:
296:
291:
289:
285:
280:
278:
277:J. J. Thomson
273:
271:
270:August Kekulé
267:
258:
249:
247:
243:
239:
235:
231:
228:
224:
209:
207:
202:
200:
194:
192:
187:
185:
181:
176:
174:
170:
165:
163:
159:
155:
150:
146:
137:
128:
125:
121:
117:
113:
109:
105:
101:
97:
93:
88:
86:
82:
78:
74:
70:
66:
62:
58:
50:
45:
41:
37:
33:
19:
4308:Ene reaction
3668:Autoxidation
3529:Degradation
3420:Azo coupling
3197:Ugi reaction
2797:Ene reaction
2597:Alkynylation
2448:Polyfluorene
2443:Polar effect
2308:Electrophile
2223:Bredt's rule
2193:Baird's rule
2187:
2163:Alpha effect
2036:Spectroscopy
2031:Publications
2011:Nomenclature
1995:
1983:Concepts in
1920:Baird's rule
1909:
1659:
1640:Charge-shift
1603:Hypervalence
1507:cite journal
1490:
1486:
1480:
1463:
1459:
1453:
1436:
1432:
1426:
1401:
1397:
1391:
1377:cite journal
1360:
1355:
1345:
1331:cite journal
1306:
1302:
1296:
1282:cite journal
1265:
1261:
1254:
1240:(17): 4367.
1237:
1233:
1227:
1210:
1206:
1200:
1183:
1178:
1172:
1155:
1151:
1145:
1128:
1124:
1118:
1102:(10): 3433.
1099:
1095:
1085:
1068:
1062:
1056:
1031:
1027:
1021:
1004:
1000:
926:
901:dissymmetric
900:
897:right-handed
885:Möbius strip
881:closed shell
868:
862:
795:
786:
780:
749:
717:
708:phenanthrene
690:
675:, and their
644:
641:heterocyclic
638:
607:
599:
567:
560:
545:
486:
462:antiaromatic
459:
455:π-π stacking
452:
441:
437:
421:
409:
362:
346:
341:
329:
325:
307:
302:
298:
292:
287:
281:
274:
263:
222:
220:
203:
195:
188:
177:
168:
166:
148:
142:
89:
60:
54:
40:
2807:Ethenolysis
2453:Ring strain
2423:Nucleophile
2248:Clar's rule
2188:Aromaticity
1996:Aromaticity
1910:Aromaticity
1886:Heterolysis
1864:Salt bridge
1809:Noncovalent
1779:Low-barrier
1660:Aromaticity
1650:Conjugation
1630:Pi backbond
929:guanidinium
893:left-handed
814:phosphorine
802:silabenzene
798:borabenzene
776:cyclophanes
736:paracetamol
734:(aspirin),
700:naphthalene
687:Polycyclics
643:aromatics (
541:Chlorophyll
533:pyrimidines
505:nucleotides
476:Hund's rule
379:conjugated
377:delocalized
330:inner cycle
322:conjugation
299:double ring
169:inner cycle
162:double bond
61:aromaticity
5370:Categories
5091:Ozonolysis
4618:Annulation
3968:Ozonolysis
2087:Topics in
1838:aurophilic
1819:Mechanical
983:References
909:paradromic
791:hybridized
704:anthracene
497:tryptophan
334:Erich Clar
73:lone pairs
65:conjugated
4605:reactions
4120:reactions
3615:reactions
3531:reactions
3413:reactions
2555:reactions
1930:spherical
1891:Homolysis
1854:Cation–pi
1829:Chalcogen
1789:Symmetric
1645:Hapticity
764:tropylium
679:analogs (
673:thiophene
661:imidazole
613:annulenes
586:polyester
489:histidine
238:olfactory
124:resonance
100:electrons
96:resonance
85:olfactory
49:resonance
36:aromantic
2498:Vinylogy
2168:Annulene
2115:Reagents
1859:Anion–pi
1849:Stacking
1771:Hydrogen
1682:Metallic
1573:Covalent
1565:(strong)
1418:16209498
1323:14685233
1158:: 1604.
1048:11749368
936:See also
834:borazine
665:pyrazole
657:pyrazine
653:pyridine
517:cytosine
501:tyrosine
390:Coplanar
310:electron
242:terpenes
223:chemical
67:ring of
18:Aromatic
2158:A value
1824:Halogen
1670:bicyclo
1615:Agostic
1007:: 1–3.
772:tropone
730:(TNT),
669:oxazole
609:Benzene
582:aniline
574:styrene
570:-xylene
563:-xylene
556:toluene
552:benzene
529:purines
521:guanine
513:thymine
509:adenine
266:benzene
230:radical
212:History
180:σ-bonds
120:History
112:benzene
94:and of
1925:Möbius
1753:forces
1743:(weak)
1416:
1321:
1303:Nature
1046:
905:chiral
832:) and
706:, and
578:phenol
525:uracil
523:, and
499:, and
353:Hückel
227:phenyl
173:π-bond
158:single
154:length
149:actual
131:Theory
116:Kekulé
108:bonded
1903:rules
1812:other
1700:Ionic
1608:3c–4e
1596:8c–2e
1591:4c–2e
1586:3c–2e
915:. In
718:Many
649:furan
590:nylon
561:ortho
417:furan
385:bonds
118:(see
104:atoms
75:, or
1665:homo
1620:Bent
1513:link
1414:PMID
1383:link
1337:link
1319:PMID
1288:link
1044:PMID
962:SARA
895:or
752:ions
588:and
568:para
565:and
537:heme
427:and
365:aryl
347:The
1495:doi
1491:100
1468:doi
1464:112
1441:doi
1437:115
1406:doi
1365:doi
1361:127
1311:doi
1307:426
1270:doi
1242:doi
1215:doi
1188:doi
1160:doi
1156:127
1133:doi
1129:162
1104:doi
1100:105
1073:doi
1069:105
1036:doi
1032:101
1009:doi
957:PAH
903:or
824:(Si
816:or
740:DNA
639:In
531:or
448:NMR
344:).
303:sic
55:In
5372::
1509:}}
1505:{{
1489:.
1462:.
1435:.
1412:.
1400:.
1379:}}
1375:{{
1333:}}
1329:{{
1317:.
1305:.
1284:}}
1280:{{
1266:50
1264:.
1238:10
1236:.
1209:.
1184:44
1154:.
1127:.
1098:.
1094:.
1067:.
1042:.
1030:.
1003:.
991:^
856:Si
854:12
836:(B
812:,
808:,
804:,
800:,
789:³
787:sp
778:.
742:.
710:.
702:,
671:,
667:,
663:,
659:,
655:,
631:.
592:.
584:,
580:,
576:,
558:,
554:,
519:,
515:,
511:,
495:,
491:,
375:A
164:.
71:,
59:,
2080:e
2073:t
2066:v
1976:e
1969:t
1962:v
1840:)
1836:(
1546:e
1539:t
1532:v
1515:)
1501:.
1497::
1474:.
1470::
1447:.
1443::
1420:.
1408::
1402:7
1385:)
1371:.
1367::
1339:)
1325:.
1313::
1290:)
1276:.
1272::
1248:.
1244::
1221:.
1217::
1211:6
1194:.
1190::
1166:.
1162::
1139:.
1135::
1112:.
1106::
1079:.
1075::
1050:.
1038::
1015:.
1011::
1005:8
874:π
858:7
850:5
846:6
844:H
842:3
840:N
838:3
830:6
828:H
826:6
625:n
623:H
621:n
615:(
507:(
406:.
381:π
326:C
301:(
38:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.