20:
124:
In outer-sphere ET reactions, the participating redox centers are not linked via any bridge during the ET event. Instead, the electron "hops" through space from the reducing center to the acceptor. Outer sphere electron transfer can occur between different chemical species or between identical
105:
In inner-sphere ET, the two redox centers are covalently linked during the ET. This bridge can be permanent, in which case the electron transfer event is termed intramolecular electron transfer. More commonly, however, the covalent linkage is transitory, forming just prior to the ET and then
106:
disconnecting following the ET event. In such cases, the electron transfer is termed intermolecular electron transfer. A famous example of an inner sphere ET process that proceeds via a transitory bridged intermediate is the reduction of by . In this case, the chloride
155:
is that the rates of such self-exchange reactions are mathematically related to the rates of "cross reactions". Cross reactions entail partners that differ by more than their oxidation states. One example (of many thousands) is the reduction of permanganate by
287:
435:
Susan B. Piepho, Elmars R. Krausz, P. N. Schatz; J. Am. Chem. Soc., 1978, 100 (10), pp 2996–3005; Vibronic coupling model for calculation of mixed-valence absorption profiles;
279:
271:
222:
Especially in proteins, electron transfer often involves hopping of an electron from one redox-active center to another. The hopping pathway, which is viewed as a
290:. In proteins, ET rates are governed by the bond structures: the electrons, in effect, tunnel through the bonds comprising the chain structure of the proteins.
262:, has guided most discussions of electron transfer ever since. Both theories are, however, semiclassical in nature, although they have been extended to fully
125:
chemical species that differ only in their oxidation state. The latter process is termed self-exchange. As an example, self-exchange describes the
491:
337:
456:
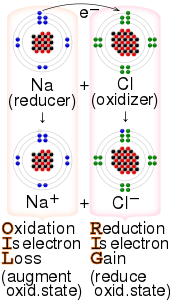
31 May 1991: Vol. 252 no. 5010 pp. 1285–1288; Protein electron transfer rates set by the bridging secondary and tertiary structure;
351:
Piechota, Eric J.; Meyer, Gerald J. (2019). "Introduction to
Electron Transfer: Theoretical Foundations and Pedagogical Examples".
173:
Reactants diffuse together, forming an "encounter complex", out of their solvent shells => precursor complex (requires work =w
145:
In general, if electron transfer is faster than ligand substitution, the reaction will follow the outer-sphere electron transfer.
230:, e.g. the 4Fe-4S ferredoxins. These site are often separated by 7-10 Å, a distance compatible with fast outer-sphere ET.
703:
580:
537:
304:
844:
484:
423:
402:
752:
747:
557:
251:
243:
119:
100:
917:
912:
943:
477:
938:
882:
572:
223:
609:
509:
839:
202:
In heterogeneous electron transfer, an electron moves between a chemical species and a solid-state
75:
887:
688:
247:
126:
642:
275:
872:
804:
662:
652:
867:
595:
360:
329:
148:
Often occurs when one/both reactants are inert or if there is no suitable bridging ligand.
83:
71:
8:
877:
809:
794:
737:
299:
227:
364:
902:
672:
501:
376:
263:
897:
892:
854:
799:
698:
634:
419:
398:
380:
309:
283:
239:
79:
56:
to another such chemical entity. ET is a mechanistic description of certain kinds of
829:
778:
732:
457:
436:
368:
282:. Furthermore, theories have been put forward to take into account the effects of
226:, guides and facilitates ET within an insulating matrix. Typical redox centers are
207:
63:
907:
819:
768:
372:
82:
ET is a step in some commercial polymerization reactions. It is foundational to
614:
603:
267:
250:
approach. The Marcus theory of electron transfer was then extended to include
67:
932:
862:
834:
742:
693:
667:
259:
152:
469:
461:
206:. Theories addressing heterogeneous electron transfer have applications in
814:
620:
527:
517:
130:
773:
708:
440:
211:
824:
255:
203:
186:
Relaxation of bond lengths, solvent molecules => successor complex
134:
110:
is the bridging ligand that covalently connects the redox partners.
53:
45:
180:
Changing bond lengths, reorganize solvent => activated complex
19:
161:
157:
107:
727:
57:
24:
16:
Relocation of an electron from an atom or molecule to another
238:
The first generally accepted theory of ET was developed by
49:
547:
167:
197:
113:
94:
930:
66:are ET reactions. ET reactions are relevant to
89:
27:reaction between sodium and chlorine, with the
499:
485:
350:
217:
60:reactions involving transfer of electrons.
492:
478:
397:(2nd ed.). Oxford: Butterworth-Heinemann.
286:on electron transfer; in particular, the
258:and Marcus. The resultant theory called
18:
393:Greenwood, N. N.; Earnshaw, A. (1997).
931:
518:Unimolecular nucleophilic substitution
189:Diffusion of products (requires work=w
168:Five steps of an outer sphere reaction
133:and its one-electron reduced relative
528:Bimolecular nucleophilic substitution
473:
414:Holleman, A. F.; Wiberg, E. (2001).
581:Electrophilic aromatic substitution
13:
548:Nucleophilic internal substitution
538:Nucleophilic aromatic substitution
452:Beratan DN, Betts JN, Onuchic JN,
305:Electrochemical reaction mechanism
14:
955:
340:from the original on 2022-11-03.
704:Lindemann–Hinshelwood mechanism
288:PKS theory of electron transfer
198:Heterogeneous electron transfer
753:Outer sphere electron transfer
748:Inner sphere electron transfer
558:Nucleophilic acyl substitution
446:
429:
408:
387:
344:
322:
278:and following earlier work in
252:inner-sphere electron transfer
244:outer-sphere electron transfer
120:Outer-sphere electron transfer
114:Outer-sphere electron transfer
101:Inner-sphere electron transfer
95:Inner-sphere electron transfer
1:
918:Diffusion-controlled reaction
418:. San Diego: Academic Press.
353:Journal of Chemical Education
315:
274:, and others proceeding from
443:; Publication Date: May 1978
90:Classes of electron transfer
7:
573:Electrophilic substitutions
373:10.1021/acs.jchemed.9b00489
293:
10:
960:
883:Energy profile (chemistry)
845:More O'Ferrall–Jencks plot
510:Nucleophilic substitutions
218:Vectoral electron transfer
117:
98:
76:transition metal complexes
913:Michaelis–Menten kinetics
853:
787:
761:
717:
681:
633:
594:
571:
508:
395:Chemistry of the Elements
280:non-radiative transitions
233:
64:Electrochemical processes
840:Potential energy surface
719:Electron/Proton transfer
604:Unimolecular elimination
888:Transition state theory
689:Intramolecular reaction
615:Bimolecular elimination
462:10.1126/science.1656523
248:transition-state theory
164:and, again, manganate.
682:Unimolecular reactions
643:Electrophilic addition
272:Alexander M. Kuznetsov
34:
873:Rate-determining step
805:Reactive intermediate
663:Free-radical addition
653:Nucleophilic addition
596:Elimination reactions
74:and commonly involve
22:
868:Equilibrium constant
228:iron-sulfur clusters
84:photoredox catalysis
944:Reaction mechanisms
878:Reaction coordinate
810:Radical (chemistry)
795:Elementary reaction
738:Grotthuss mechanism
502:reaction mechanisms
441:10.1021/ja00478a011
416:Inorganic Chemistry
365:2019JChEd..96.2450P
300:Electron equivalent
276:Fermi's golden rule
246:and was based on a
25:reduction–oxidation
939:Physical chemistry
903:Arrhenius equation
673:Oxidative addition
635:Addition reactions
264:quantum mechanical
260:Marcus-Hush theory
210:and the design of
48:relocates from an
35:
926:
925:
898:Activated complex
893:Activation energy
855:Chemical kinetics
800:Reaction dynamics
699:Photodissociation
359:(11): 2450–2466.
310:Solvated electron
284:vibronic coupling
240:Rudolph A. Marcus
183:Electron transfer
151:A key concept of
129:reaction between
80:organic chemistry
44:) occurs when an
38:Electron transfer
951:
830:Collision theory
779:Matrix isolation
733:Harpoon reaction
610:E1cB-elimination
494:
487:
480:
471:
470:
464:
450:
444:
433:
427:
412:
406:
391:
385:
384:
348:
342:
341:
326:
208:electrochemistry
32:
959:
958:
954:
953:
952:
950:
949:
948:
929:
928:
927:
922:
908:Eyring equation
849:
820:Stereochemistry
783:
769:Solvent effects
757:
713:
677:
658:
648:
629:
624:
590:
586:
567:
563:
553:
543:
533:
523:
504:
498:
468:
467:
451:
447:
434:
430:
413:
409:
392:
388:
349:
345:
328:
327:
323:
318:
296:
236:
220:
200:
192:
176:
170:
122:
116:
103:
97:
92:
28:
17:
12:
11:
5:
957:
947:
946:
941:
924:
923:
921:
920:
915:
910:
905:
900:
895:
890:
885:
880:
875:
870:
865:
859:
857:
851:
850:
848:
847:
842:
837:
832:
827:
822:
817:
812:
807:
802:
797:
791:
789:
788:Related topics
785:
784:
782:
781:
776:
771:
765:
763:
762:Medium effects
759:
758:
756:
755:
750:
745:
740:
735:
730:
724:
722:
715:
714:
712:
711:
706:
701:
696:
691:
685:
683:
679:
678:
676:
675:
670:
665:
660:
656:
650:
646:
639:
637:
631:
630:
628:
627:
622:
618:
612:
607:
600:
598:
592:
591:
589:
588:
584:
577:
575:
569:
568:
566:
565:
561:
555:
551:
545:
541:
535:
531:
525:
521:
514:
512:
506:
505:
497:
496:
489:
482:
474:
466:
465:
445:
428:
407:
386:
343:
320:
319:
317:
314:
313:
312:
307:
302:
295:
292:
268:Joshua Jortner
266:treatments by
235:
232:
219:
216:
199:
196:
195:
194:
190:
187:
184:
181:
178:
174:
169:
166:
143:
142:
118:Main article:
115:
112:
99:Main article:
96:
93:
91:
88:
68:photosynthesis
15:
9:
6:
4:
3:
2:
956:
945:
942:
940:
937:
936:
934:
919:
916:
914:
911:
909:
906:
904:
901:
899:
896:
894:
891:
889:
886:
884:
881:
879:
876:
874:
871:
869:
866:
864:
863:Rate equation
861:
860:
858:
856:
852:
846:
843:
841:
838:
836:
835:Arrow pushing
833:
831:
828:
826:
823:
821:
818:
816:
813:
811:
808:
806:
803:
801:
798:
796:
793:
792:
790:
786:
780:
777:
775:
772:
770:
767:
766:
764:
760:
754:
751:
749:
746:
744:
743:Marcus theory
741:
739:
736:
734:
731:
729:
726:
725:
723:
720:
716:
710:
707:
705:
702:
700:
697:
695:
694:Isomerization
692:
690:
687:
686:
684:
680:
674:
671:
669:
668:Cycloaddition
666:
664:
661:
654:
651:
644:
641:
640:
638:
636:
632:
626:
619:
616:
613:
611:
608:
605:
602:
601:
599:
597:
593:
582:
579:
578:
576:
574:
570:
559:
556:
549:
546:
539:
536:
529:
526:
519:
516:
515:
513:
511:
507:
503:
495:
490:
488:
483:
481:
476:
475:
472:
463:
459:
455:
449:
442:
438:
432:
425:
424:0-12-352651-5
421:
417:
411:
404:
403:0-7506-3365-4
400:
396:
390:
382:
378:
374:
370:
366:
362:
358:
354:
347:
339:
335:
331:
325:
321:
311:
308:
306:
303:
301:
298:
297:
291:
289:
285:
281:
277:
273:
269:
265:
261:
257:
253:
249:
245:
241:
231:
229:
225:
215:
213:
209:
205:
188:
185:
182:
179:
172:
171:
165:
163:
159:
154:
153:Marcus theory
149:
146:
140:
139:
138:
136:
132:
128:
121:
111:
109:
102:
87:
85:
81:
77:
73:
69:
65:
61:
59:
55:
51:
47:
43:
39:
31:
26:
23:Example of a
21:
815:Molecularity
718:
453:
448:
431:
415:
410:
394:
389:
356:
352:
346:
333:
324:
237:
221:
201:
150:
147:
144:
131:permanganate
123:
104:
62:
41:
37:
36:
29:
774:Cage effect
709:RRKM theory
625:elimination
242:to address
212:solar cells
72:respiration
933:Categories
316:References
127:degenerate
825:Catalysis
721:reactions
381:208754569
256:Noel Hush
204:electrode
135:manganate
338:Archived
334:Bitesize
330:"Metals"
294:See also
160:to form
141:+ → +
54:molecule
46:electron
33:mnemonic
454:Science
361:Bibcode
336:. BBC.
30:OIL RIG
500:Basic
422:
401:
379:
234:Theory
224:vector
162:iodine
158:iodide
108:ligand
728:Redox
564:Acyl)
377:S2CID
78:. In
58:redox
617:(E2)
606:(E1)
420:ISBN
399:ISBN
70:and
50:atom
587:Ar)
544:Ar)
458:doi
437:doi
369:doi
254:by
52:or
935::
655:(A
645:(A
583:(S
560:(S
554:i)
550:(S
540:(S
534:2)
530:(S
524:1)
520:(S
375:.
367:.
357:96
355:.
332:.
270:,
214:.
137::
86:.
42:ET
659:)
657:N
649:)
647:E
623:i
621:E
585:E
562:N
552:N
542:N
532:N
522:N
493:e
486:t
479:v
460::
439::
426:.
405:.
383:.
371::
363::
193:)
191:p
177:)
175:r
40:(
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.