709:; Division 5.1(a)1 and Class 5; Division 5.1(a)2. Division 5.1 "means a material that may, generally by yielding oxygen, cause or enhance the combustion of other materials." Division 5.(a)1 of the DOT code applies to solid oxidizers "if, when tested in accordance with the UN Manual of Tests and Criteria (IBR, see § 171.7 of this subchapter), its mean burning time is less than or equal to the burning time of a 3:7 potassium bromate/cellulose mixture." 5.1(a)2 of the DOT code applies to liquid oxidizers "if, when tested in accordance with the UN Manual of Tests and Criteria, it spontaneously ignites or its mean time for a pressure rise from 690 kPa to 2070 kPa gauge is less than the time of a 1:1 nitric acid (65 percent)/cellulose mixture."
169:
22:
37:
151:
1333:
693:
definition of an oxidizing agent is a substance that can cause or contribute to the combustion of other material. By this definition some materials that are classified as oxidizing agents by analytical chemists are not classified as oxidizing agents in a dangerous materials sense. An example is
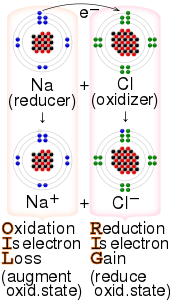
111:, of the oxidizer decreases while that of the reductant increases; this is expressed by saying that oxidizers "undergo reduction" and "are reduced" while reducers "undergo oxidation" and "are oxidized". Common oxidizing agents are
271:
In more common usage, an oxidizing agent transfers oxygen atoms to a substrate. In this context, the oxidizing agent can be called an oxygenation reagent or oxygen-atom transfer (OAT) agent. Examples include
26:
706:
684:
134:(redox) reaction. In the second sense, an oxidizing agent is a chemical species that transfers electronegative atoms, usually oxygen, to a substrate.
1229:
705:
defines oxidizing agents specifically. There are two definitions for oxidizing agents governed under DOT regulations. These two are
260:
1295:
259:
Extensive tabulations of ranking the electron accepting properties of various reagents (redox potentials) are available, see
1337:
702:
195:
1358:
1285:
183:
335:
In some cases, these oxides can also serve as electron acceptors, as illustrated by the conversion of
1363:
1353:
550:
172:
554:
491:
1188:
1034:
658:
580:
139:
1061:
473:
29:
1243:
N. G. Connelly, W. E. Geiger (1996). "Chemical Redox Agents for
Organometallic Chemistry".
1221:
1050:
1043:
695:
629:
8:
1088:
1070:
412:
1197: – Chemical species that donates an electron to another species in a redox reaction
463:
168:
1291:
1262:
1200:
1179:
1161:
935:
647:
558:
528:
450:
396:
179:
127:
116:
74:
65:
1254:
1245:
1173:
1103:
895:
625:
590:
532:
309:
130:
in which it gains one or more electrons. In that sense, it is one component in an
690:
536:
104:
40:
1368:
1194:
1167:
906:
83:
1347:
917:
662:
617:
1318:
49 CFR 172.127 General
Requirements for Shipments and Packagings; Subpart D
1266:
999:
950:
887:
838:
667:
576:
546:
505:
458:
360:
301:
285:
16:
Chemical compound used to oxidize another substance in a chemical reaction
1281:
921:
881:
517:
438:
325:
21:
126:
In one sense, an oxidizing agent is a chemical species that undergoes a
1170: – Chemical entity capable of donating electrons to another entity
1155:
974:
763:
241:
135:
36:
1258:
698:, which does not pass the dangerous goods test of an oxidizing agent.
1014:
364:
108:
1137:
1122:
863:
788:
774:
542:
513:
509:
425:
417:
408:
78:
1176: – Synthesis of chemical compounds in an electrochemical cell
802:
521:
446:
433:
120:
100:
1287:
Advanced
Organic Chemistry: Reactions, Mechanisms, and Structure
150:
1332:
912:
816:
732:
376:
112:
1191: – Redox reaction that takes place with organic compounds
1182: – Redox reaction that takes place with organic compounds
1279:
753:
712:
386:
329:
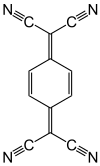
240:. One of the strongest acceptors commercially available is "
155:
131:
70:
1242:
1203: – Free electron in a solution, often liquid ammonia
1143:
Tl(I) thallous compounds, in organic lab scale synthesis
1184:
Pages displaying short descriptions of redirect targets
1164: – Chemical entity capable of accepting electrons
685:
HAZMAT Class 5 Oxidizing agents and organic peroxides
99:). In other words, an oxidizer is any substance that
186:. In this context, the oxidizing agent is called an
1158: – Chemical reaction between a fuel and oxygen
678:
547:chromic and dichromic acids and chromium trioxide
1345:
158:reaction between sodium and chlorine, with the
1290:(6th ed.), New York: Wiley-Interscience,
1309:Australian Dangerous Goods Code, 6th Edition
758:Various, including ketones, aldehydes, and H
924:production, more commonly reducing agent)
713:Common oxidizing agents and their products
370:
266:
261:Standard electrode potential (data page)
228:, which accepts an electron to form Fe(C
167:
149:
107:, which describes the degree of loss of
35:
20:
1346:
1232:from the original on November 3, 2022.
244:", the radical cation derived from N(C
1236:
328:). Notice that these species are all
145:
190:and the reducing agent is called an
194:. A classic oxidizing agent is the
13:
14:
1380:
1325:
703:U.S. Department of Transportation
142:involve atom-transfer reactions.
73:chemical reaction that gains or "
1331:
175:is an organic electron-acceptor.
1127:Various, including oxides and H
1108:in organic lab scale synthesis
737:Various, including the oxides H
1312:
1303:
1273:
1214:
679:Dangerous materials definition
657:Cerium (IV) compounds such as
1:
1207:
7:
1149:
184:electron-transfer reactions
10:
1385:
682:
43:label for oxidizing agents
551:pyridinium chlorochromate
173:Tetracyanoquinodimethane
32:for oxidizing chemicals.
492:Peroxymonosulfuric acid
371:Common oxidizing agents
140:organic redox reactions
138:, many explosives, and
103:another substance. The
1189:Organic redox reaction
1035:antimony pentafluoride
659:ceric ammonium nitrate
581:potassium permanganate
520:, and other analogous
407:) and other inorganic
267:Atom-transfer reagents
176:
165:
69:) is a substance in a
44:
33:
1062:platinum hexafluoride
474:Peroxydisulfuric acid
171:
153:
39:
24:
1340:at Wikimedia Commons
1051:antimony trifluoride
1044:hexafluoroantimonate
696:potassium dichromate
630:Dinitrogen tetroxide
1359:Chemical properties
1280:Smith, Michael B.;
1089:ruthenium tetroxide
1071:hexafluoroplatinate
929:Hexavalent chromium
555:chromate/dichromate
457:), the oxidizer in
156:reduction–oxidation
132:oxidation–reduction
579:compounds such as
557:compounds such as
545:compounds such as
464:Potassium chlorate
449:compounds such as
312:), and especially
180:Electron acceptors
177:
166:
146:Electron acceptors
61:electron recipient
51:(also known as an
45:
34:
1336:Media related to
1297:978-0-471-72091-1
1259:10.1021/cr940053x
1201:Solvated electron
1180:Organic oxidation
1162:Electron acceptor
1147:
1146:
936:chromium trioxide
648:Sodium bismuthate
559:Sodium dichromate
451:potassium nitrate
397:Hydrogen peroxide
188:electron acceptor
128:chemical reaction
117:hydrogen peroxide
66:electron acceptor
1376:
1364:Electrochemistry
1354:Oxidizing agents
1338:Oxidizing agents
1335:
1319:
1316:
1310:
1307:
1301:
1300:
1277:
1271:
1270:
1246:Chemical Reviews
1240:
1234:
1233:
1218:
1185:
1174:Electrosynthesis
1104:osmium tetroxide
1102:
1101:
1100:
1087:
1086:
1085:
1013:
1012:
1011:
998:
997:
996:
973:
972:
971:
963:
962:
949:
948:
947:
896:nitrogen dioxide
862:
861:
860:
832:
831:
830:
717:
716:
626:Nitrogen dioxide
613:
612:
611:
603:
602:
601:
591:Sodium perborate
413:Fenton's reagent
358:
357:
356:
346:
345:
344:
323:
322:
321:
310:osmium tetroxide
299:
298:
297:
283:
282:
281:
227:
226:
225:
217:
216:
208:
207:
163:
77:"/"receives" an
1384:
1383:
1379:
1378:
1377:
1375:
1374:
1373:
1344:
1343:
1328:
1323:
1322:
1317:
1313:
1308:
1304:
1298:
1278:
1274:
1241:
1237:
1220:
1219:
1215:
1210:
1183:
1152:
1130:
1120:
1116:
1099:
1096:
1095:
1094:
1092:
1091:
1084:
1081:
1080:
1079:
1077:
1069:
1060:
1049:
1042:
1033:
1024:
1020:
1010:
1007:
1006:
1005:
1003:
1002:
995:
992:
991:
990:
988:
982:
970:
967:
966:
965:
961:
958:
957:
956:
954:
953:
946:
943:
942:
941:
939:
938:
934:
930:
915:
905:
894:
890:
880:
871:
859:
856:
855:
854:
852:
846:
829:
826:
825:
824:
822:
815:
801:
787:
773:
761:
752:
744:
740:
731:
715:
691:dangerous goods
687:
681:
673:
653:
643:
639:
635:
623:
610:
608:
607:
606:
605:
600:
597:
596:
595:
594:
586:
572:
568:
564:
501:
497:
487:
483:
479:
469:
456:
444:
431:
423:
406:
402:
392:
382:
373:
355:
352:
351:
350:
348:
343:
340:
339:
338:
336:
320:
317:
316:
315:
313:
307:
296:
293:
292:
291:
289:
280:
277:
276:
275:
273:
269:
255:
251:
247:
239:
235:
231:
224:
221:
220:
219:
215:
212:
211:
210:
206:
203:
202:
201:
199:
182:participate in
159:
148:
105:oxidation state
49:oxidizing agent
41:Dangerous goods
17:
12:
11:
5:
1382:
1372:
1371:
1366:
1361:
1356:
1342:
1341:
1327:
1326:External links
1324:
1321:
1320:
1311:
1302:
1296:
1272:
1253:(2): 877–910.
1235:
1212:
1211:
1209:
1206:
1205:
1204:
1198:
1195:Reducing agent
1192:
1186:
1177:
1171:
1168:Electron donor
1165:
1159:
1151:
1148:
1145:
1144:
1141:
1133:
1132:
1128:
1125:
1118:
1114:
1110:
1109:
1106:
1097:
1082:
1074:
1073:
1067:
1064:
1058:
1054:
1053:
1047:
1040:
1037:
1031:
1027:
1026:
1022:
1019:Mn (acidic) or
1017:
1008:
993:
985:
984:
980:
977:
968:
959:
944:
932:
926:
925:
909:
907:sulfur dioxide
903:
899:
898:
892:
884:
878:
874:
873:
869:
866:
857:
849:
848:
844:
841:
834:
833:
827:
819:
813:
809:
808:
805:
799:
795:
794:
791:
785:
781:
780:
777:
771:
767:
766:
759:
756:
750:
746:
745:
742:
738:
735:
729:
725:
724:
721:
714:
711:
680:
677:
676:
675:
671:
665:
655:
651:
645:
641:
637:
633:
621:
615:
609:
598:
588:
584:
574:
570:
566:
562:
539:
525:
503:
499:
495:
489:
485:
481:
477:
471:
467:
461:
454:
442:
436:
429:
421:
415:
404:
400:
394:
390:
384:
380:
372:
369:
353:
341:
318:
305:
294:
278:
268:
265:
253:
249:
245:
237:
233:
229:
222:
213:
204:
192:electron donor
147:
144:
97:electron donor
94:
90:
86:
84:reducing agent
15:
9:
6:
4:
3:
2:
1381:
1370:
1367:
1365:
1362:
1360:
1357:
1355:
1352:
1351:
1349:
1339:
1334:
1330:
1329:
1315:
1306:
1299:
1293:
1289:
1288:
1283:
1276:
1268:
1264:
1260:
1256:
1252:
1248:
1247:
1239:
1231:
1227:
1223:
1217:
1213:
1202:
1199:
1196:
1193:
1190:
1187:
1181:
1178:
1175:
1172:
1169:
1166:
1163:
1160:
1157:
1154:
1153:
1142:
1139:
1135:
1134:
1126:
1124:
1112:
1111:
1107:
1105:
1090:
1076:
1075:
1072:
1065:
1063:
1056:
1055:
1052:
1045:
1038:
1036:
1029:
1028:
1018:
1016:
1001:
987:
986:
978:
976:
952:
937:
928:
927:
923:
919:
918:Claus process
914:
910:
908:
901:
900:
897:
889:
885:
883:
876:
875:
867:
865:
851:
850:
842:
840:
836:
835:
820:
818:
811:
810:
806:
804:
797:
796:
792:
790:
783:
782:
778:
776:
769:
768:
765:
757:
755:
748:
747:
736:
734:
727:
726:
722:
719:
718:
710:
708:
704:
699:
697:
692:
686:
669:
666:
664:
663:ceric sulfate
660:
656:
649:
646:
631:
627:
619:
618:Nitrous oxide
616:
592:
589:
582:
578:
575:
560:
556:
552:
548:
544:
540:
538:
534:
530:
527:Fluorides of
526:
523:
519:
515:
511:
507:
504:
493:
490:
475:
472:
465:
462:
460:
452:
448:
440:
437:
435:
432:), and other
427:
419:
416:
414:
410:
398:
395:
388:
385:
378:
375:
374:
368:
366:
362:
333:
331:
327:
311:
303:
287:
264:
262:
257:
243:
197:
193:
189:
185:
181:
174:
170:
162:
157:
154:Example of a
152:
143:
141:
137:
133:
129:
124:
122:
118:
114:
110:
106:
102:
98:
92:
88:
85:
82:
80:
76:
72:
68:
67:
62:
58:
54:
50:
42:
38:
31:
28:
27:international
23:
19:
1314:
1305:
1286:
1282:March, Jerry
1275:
1250:
1244:
1238:
1225:
1216:
1000:permanganate
888:nitric oxide
839:hypochlorite
700:
688:
668:Lead dioxide
577:Permanganate
506:Hypochlorite
459:black powder
361:permanganate
334:
286:permanganate
270:
258:
191:
187:
178:
160:
125:
96:
87:(called the
64:
60:
56:
52:
48:
46:
18:
922:ultramarine
882:nitric acid
723:Product(s)
553:(PCC), and
541:Hexavalent
518:perchlorate
439:Nitric acid
326:perchlorate
196:ferrocenium
1348:Categories
1208:References
1156:Combustion
1140:compounds
975:dichromate
764:ozonolysis
683:See also:
242:Magic blue
136:Combustion
119:, and the
1123:peroxides
1015:manganate
524:oxyanions
409:peroxides
365:manganate
109:electrons
89:reductant
30:pictogram
1284:(2007),
1267:11848774
1230:Archived
1226:Bitesize
1222:"Metals"
1150:See also
1136:Tl(III)
1121:, other
1025:(basic)
951:chromate
864:chlorate
789:chlorine
775:fluorine
741:O and CO
543:chromium
529:chlorine
514:chlorate
510:chlorite
434:halogens
426:chlorine
418:Fluorine
302:chromate
164:mnemonic
121:halogens
101:oxidizes
79:electron
57:oxidizer
1228:. BBC.
1138:thallic
803:bromine
762:O; see
707:Class 5
533:bromine
522:halogen
447:nitrate
161:OIL RIG
93:reducer
81:from a
75:accepts
53:oxidant
1294:
1265:
1046:or SbF
913:sulfur
817:iodine
733:oxygen
720:Agent
650:(NaBiO
537:iodine
535:, and
445:) and
377:Oxygen
330:oxides
304:), OsO
252:-4-Br)
113:oxygen
1369:Redox
979:Cr, H
868:Cl, H
843:Cl, H
754:ozone
583:(KMnO
466:(KClO
387:Ozone
95:, or
71:redox
63:, or
1292:ISBN
1263:PMID
837:ClO
701:The
689:The
670:(PbO
661:and
624:O),
453:(KNO
441:(HNO
359:,ie
200:Fe(C
198:ion
25:The
1255:doi
1093:OsO
1078:RuO
1066:PtF
1057:PtF
1039:SbF
1030:SbF
1021:MnO
1004:MnO
989:MnO
940:CrO
931:CrO
886:NO
877:HNO
853:ClO
821:I,
807:Br
793:Cl
636:/ N
632:(NO
561:(Na
428:(Cl
424:),
363:to
349:MnO
347:to
337:MnO
314:ClO
290:CrO
288:),
274:MnO
47:An
1350::
1261:.
1251:96
1249:.
1224:.
1131:O
983:O
955:Cr
920:,
911:S
902:SO
891:NO
872:O
847:O
798:Br
784:Cl
779:F
620:(N
565:Cr
549:,
531:,
516:,
512:,
508:,
498:SO
494:(H
476:(H
420:(F
411:,
399:(H
389:(O
379:(O
367:.
332:.
263:.
256:.
123:.
115:,
91:,
59:,
55:,
1269:.
1257::
1129:2
1119:2
1117:O
1115:2
1113:H
1098:4
1083:4
1068:6
1059:6
1048:3
1041:6
1032:5
1023:2
1009:4
994:4
981:2
969:7
964:O
960:2
945:4
933:3
916:(
904:2
893:2
879:3
870:2
858:3
845:2
828:3
823:I
814:2
812:I
800:2
786:2
772:2
770:F
760:2
751:3
749:O
743:2
739:2
730:2
728:O
674:)
672:2
654:)
652:3
644:)
642:4
640:O
638:2
634:2
628:/
622:2
614:)
604:·
599:2
593:(
587:)
585:4
573:)
571:7
569:O
567:2
563:2
502:)
500:5
496:2
488:)
486:8
484:O
482:2
480:S
478:2
470:)
468:3
455:3
443:3
430:2
422:2
405:2
403:O
401:2
393:)
391:3
383:)
381:2
354:4
342:4
324:(
319:4
308:(
306:4
300:(
295:4
284:(
279:4
254:3
250:4
248:H
246:6
238:2
236:)
234:5
232:H
230:5
223:2
218:)
214:5
209:H
205:5
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.