1105:
43:
209:) in molecules with one or more stereocenter. For a chiral molecule with one or more stereocenter, the enantiomer corresponds to the stereoisomer in which every stereocenter has the opposite configuration. An organic compound with only one stereogenic carbon is always chiral. On the other hand, an organic compound with multiple stereogenic carbons is typically, but not always, chiral. In particular, if the stereocenters are configured in such a way that the molecule can take a conformation having a plane of symmetry or an inversion point, then the molecule is achiral and is known as a
706:
580:
27:
604:
566:
785:
646:
629:
552:
380:
defined: 1000 seconds is sometimes employed, as this is regarded as the lower limit for the amount of time required for chemical or chromatographic separation of enantiomers in a practical sense. Molecules that are chiral at room temperature due to restricted rotation about a single bond (barrier to rotation ≥ ca. 23 kcal/mol) are said to exhibit
757:) leads to a stereoisomer of the original, so the central C is a stereocenter. Many chiral molecules have point chirality, namely a single chiral stereogenic center that coincides with an atom. This stereogenic center usually has four or more bonds to different groups, and may be carbon (as in many biological molecules), phosphorus (as in many
1042:, which are chiral, often distinguish between the two enantiomers of a chiral substrate. One could imagine an enzyme as having a glove-like cavity that binds a substrate. If this glove is right-handed, then one enantiomer will fit inside and be bound, whereas the other enantiomer will have a poor fit and is unlikely to bind.
1035:
is the subject of much debate. Most scientists believe that Earth life's "choice" of chirality was purely random, and that if carbon-based life forms exist elsewhere in the universe, their chemistry could theoretically have opposite chirality. However, there is some suggestion that early amino acids
1095:
Chirality is important in context of ordered phases as well, for example the addition of a small amount of an optically active molecule to a nematic phase (a phase that has long range orientational order of molecules) transforms that phase to a chiral nematic phase (or cholesteric phase). Chirality
197:
Chiral molecules will usually have a stereogenic element from which chirality arises. The most common type of stereogenic element is a stereogenic center, or stereocenter. In the case of organic compounds, stereocenters most frequently take the form of a carbon atom with four distinct (different)
379:
However, if the temperature in question is low enough, the process that interconverts the enantiomeric chiral conformations becomes slow compared to a given timescale. The molecule would then be considered to be chiral at that temperature. The relevant timescale is, to some degree, arbitrarily
371:
that are nonidentical mirror images, but the two can interconvert via the cyclohexane chair flip (~10 kcal/mol barrier). As another example, amines with three distinct substituents (RRRN:) are also regarded as achiral molecules because their enantiomeric pyramidal conformers rapidly undergo
294:
are often chiral, but not always. In living organisms, one typically finds only one of the two enantiomers of a chiral compound. For that reason, organisms that consume a chiral compound usually can metabolize only one of its enantiomers. For the same reason, the two enantiomers of a chiral
1036:
could have formed in comet dust. In this case, circularly polarised radiation (which makes up 17% of stellar radiation) could have caused the selective destruction of one chirality of amino acids, leading to a selection bias which ultimately resulted in all life on Earth being homochiral.
443:
axis, which is just an inversion. Any orientation will do, so long as it passes through the center of inversion. Also note that higher symmetries of chiral and achiral molecules also exist, and symmetries that do not include those in the table, such as the chiral
852:
If two enantiomers easily interconvert, the pure enantiomers may be practically impossible to separate, and only the racemic mixture is observable. This is the case, for example, of most amines with three different substituents (NRR′R″), because of the low
344:(the chiral point groups). However, whether the molecule itself is considered to be chiral depends on whether its chiral conformations are persistent isomers that could be isolated as separated enantiomers, at least in principle, or the
1333:
due to their different optical properties. At one time, chirality was thought to be restricted to organic chemistry, but this misconception was overthrown by the resolution of a purely inorganic compound, a cobalt complex called
663:
An example of a molecule that does not have a mirror plane or an inversion and yet would be considered achiral is 1,1-difluoro-2,2-dichlorocyclohexane (or 1,1-difluoro-3,3-dichlorocyclohexane). This may exist in many conformers
198:
groups attached to it in a tetrahedral geometry. Less commonly, other atoms like N, P, S, and Si can also serve as stereocenters, provided they have four distinct substituents (including lone pair electrons) attached to them.
1825:
Meierhenrich Uwe J., Nahon
Laurent, Alcaraz Christian, Hendrik Bredehöft Jan, Hoffmann Søren V., Barbier Bernard, Brack André (2005). "Asymmetric Vacuum UV photolysis of the Amino Acid Leucine in the Solid State".
219:
Molecules with chirality arising from one or more stereocenters are classified as possessing central chirality. There are two other types of stereogenic elements that can give rise to chirality, a stereogenic axis
795:
Similarly, a stereogenic axis (or plane) is defined as an axis (or plane) in the molecule such that the swapping of any two ligands attached to the axis (or plane) gives rise to a stereoisomer. For instance, the
2171:
Researches on the molecular asymmetry of natural organic products, English translation of French original, published by
Alembic Club Reprints (Vol. 14, pp. 1–46) in 1905, facsimile reproduction by SPIE in a 1990
1632:
Lepola U, Wade A, Andersen HF (May 2004). "Do equivalent doses of escitalopram and citalopram have similar efficacy? A pooled analysis of two positive placebo-controlled studies in major depressive disorder".
672:
ring would have to be flat, widening the bond angles and giving the conformation a very high energy. This compound would not be considered chiral because the chiral conformers interconvert easily.
2376:
Ohloff, Günther; Vial, Christian; Wolf, Hans
Richard; Job, Kurt; Jégou, Elise; Polonsky, Judith; Lederer, Edgar (1980). "Stereochemistry-Odor Relationships in Enantiomeric Ambergris Fragrances".
1604:
Theodore J. Leitereg; Dante G. Guadagni; Jean Harris; Thomas R. Mon; Roy
Teranishi (1971). "Chemical and sensory data supporting the difference between the odors of the enantiomeric carvones".
1564:
Gal, Joseph (2012). "The
Discovery of Stereoselectivity at Biological Receptors: Arnaldo Piutti and the Taste of the Asparagine Enantiomers-History and Analysis on the 125th Anniversary".
729:) is an atom such that swapping the positions of two ligands (connected groups) on that atom results in a molecule that is stereoisomeric to the original. For example, a common case is a
1537:
Streitwieser, A. Jr.; Wolfe, J. R. Jr.; Schaeffer, W. D. (1959). "Stereochemistry of the
Primary Carbon. X. Stereochemical Configurations of Some Optically Active Deuterium Compounds".
348:
conformers rapidly interconvert at a given temperature and timescale through low-energy conformational changes (rendering the molecule achiral). For example, despite having chiral
2756:
1141:
complex in which the three bipyridine ligands adopt a chiral propeller-like arrangement. The two enantiomers of complexes such as may be designated as Λ (capital
1228:
crystals in 1849. Sometimes it is possible to seed a racemic solution with a right-handed and a left-handed crystal so that each will grow into a large crystal.
363:
is considered achiral at room temperature because rotation about the central C–C bond rapidly interconverts the enantiomers (3.4 kcal/mol barrier). Similarly,
2761:
1824:
182:
or some other criterion. The two enantiomers have the same chemical properties, except when reacting with other chiral compounds. They also have the same
2473:
1993:
2264:
2092:
2063:
2034:
1494:
1465:
1861:
1118:
Chirality is a symmetry property, not a property of any part of the periodic table. Thus many inorganic materials, molecules, and ions are chiral.
675:
An achiral molecule having chiral conformations could theoretically form a mixture of right-handed and left-handed crystals, as often happens with
2622:
680:
2595:
2489:
2447:
2009:
2574:
2569:
1801:
1263:
when only one of two possible enantiomers is present so that all molecules within a sample have the same chirality sense. Use of
405:) but not always asymmetric (lacking all symmetry elements except the trivial identity). Asymmetric molecules are always chiral.
1138:
1288:
is the difference between how much of one enantiomer is present compared to the other. For example, a sample with 40% e.e. of
2530:
1220:. Some racemic mixtures spontaneously crystallize into right-handed and left-handed crystals that can be separated by hand.
2223:
1208:. This often involves forming crystals of a salt composed of one of the enantiomers and an acid or base from the so-called
2221:
Bentley, Ronald (1995). "From
Optical Activity in Quartz to Chiral Drugs: Molecular Handedness in Biology and Medicine".
1676:
Hyttel, J.; Bøgesø, K. P.; Perregaard, J.; Sánchez, C. (1992). "The pharmacological effect of citalopram resides in the (
2615:
2455:
2417:
2198:
1784:
1922:
Hartwig, J. F. Organotransition Metal
Chemistry, from Bonding to Catalysis; University Science Books: New York, 2010.
1435:
2794:
2412:(2nd ed.). Oxford, UK: Oxford University Press. pp. 319f, 432, 604np, 653, 746int, 803ketals, 839, 846f.
1236:
413:
1183:
1150:
865:
2871:
1927:
1910:
1454:
730:
1145:, the Greek version of "L") for a left-handed twist of the propeller described by the ligands, and Δ (capital
2861:
2779:
2608:
2493:
2013:
765:). However, a stereogenic center can also be a trivalent atom whose bonds are not in the same plane, such as
717:
leads to a molecule that is a stereoisomer of the original. Hence, the central carbon atom is a stereocenter.
398:), which includes planes of symmetry and inversion center. Chiral molecules are always dissymmetric (lacking
1160:
complexes. If the metal exhibits catalytic properties, its combination with a chiral ligand is the basis of
2789:
1358:
1231:
Liquid chromatography (HPLC and TLC) may also be used as an analytical method for the direct separation of
1113:
2596:
http://quantummechanics.ucsd.edu/ph87/ScientificAmerican/Sciam/Hegstrom_The_Handedness_of_the_universe.pdf
2564:
1178:
is derived from the interaction of chiral materials with polarized light. In a solution, the (−)-form, or
416:
of the molecule. In the achiral molecules, X and Y (with no subscript) represent achiral groups, whereas X
1383:
2632:
2554:
1388:
1378:
2124:"Corpuscular Optics and the Wave Theory of Light: The Science and Politics of a Revolution in Physics"
1122:
is an example from the mineral kingdom. Such noncentric materials are of interest for applications in
155:
2751:
388:
2589:
2856:
2677:
1409:
1134:
1104:
368:
105:
201:
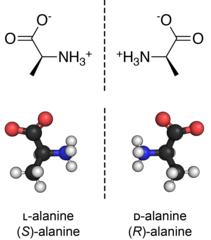
A given stereocenter has two possible configurations (R and S), which give rise to stereoisomers (
2881:
2559:
1363:
42:
2584:
1130:
894:
880:
300:
233:
179:
101:
1523:
Cotton, F. A., "Chemical
Applications of Group Theory," John Wiley & Sons: New York, 1990.
315:
of its conformations. A conformation of a molecule is chiral if and only if it belongs to the
2886:
2876:
2815:
2729:
1855:
1603:
1346:
804:
788:
762:
665:
2825:
2810:
2661:
2334:
1606:
1161:
992:
910:
797:
409:
8:
2723:
2600:
1368:
1281:
1187:
373:
97:
2338:
1449:(3rd Edition) Marye Anne Fox, James K. Whitesell Jones & Bartlett Publishers (2004)
1197:
form, of an optical isomer does the opposite. The rotation of light is measured using a
2866:
2784:
2682:
2358:
2247:
2151:
2143:
1705:
1658:
1310:
868:
for an enantiomer is too low for practical measurement, the species is said to exhibit
858:
770:
700:
312:
229:
1888:
1749:
1724:
2771:
2579:
2536:
2526:
2451:
2442:. Vol. 9 (1st ed.). New York, NY, USA: Wiley & Sons. pp. 428–430.
2413:
2350:
2239:
2194:
2155:
1923:
1906:
1843:
1780:
1754:
1697:
1650:
1646:
1581:
1550:
1450:
1431:
1205:
1096:
in context of such phases in polymeric fluids has also been studied in this context.
988:
854:
237:
2433:
2362:
2274:
2251:
2184:
2102:
2073:
2044:
1709:
1662:
1536:
1504:
1475:
2820:
2746:
2656:
2518:
2485:
2443:
2385:
2342:
2307:
2278:
2269:
2231:
2135:
2106:
2097:
2077:
2068:
2048:
2039:
2005:
1975:
1951:
1943:
1884:
1835:
1744:
1736:
1689:
1642:
1614:
1573:
1546:
1508:
1499:
1479:
1470:
1373:
1329:
in 1894. Different enantiomers or diastereomers of a compound were formerly called
1190:
1174:
1123:
1089:
828:
225:
187:
115:
69:
2346:
408:
The following table shows some examples of chiral and achiral molecules, with the
228:). Finally, the inherent curvature of a molecule can also give rise to chirality (
178:; they are often distinguished as either "right-handed" or "left-handed" by their
2830:
2718:
2698:
2672:
2435:
2407:
2186:
1955:
1404:
1398:
1393:
1309:
The rotation of plane polarized light by chiral substances was first observed by
1012:
Many biologically active molecules are chiral, including the naturally occurring
876:
869:
834:
Chirality can also arise from isotopic differences between atoms, such as in the
827:
derivatives bearing two or more substituents have stereogenic planes and exhibit
812:
758:
684:
251:
221:
191:
20:
2509:
Gal, Joseph (2013). "Molecular
Chirality: Language, History, and Significance".
2295:
2139:
1314:
1194:
1146:
956:
838:
781:(OSRR′), because a lone-pair of electrons is present instead of a fourth bond.
778:
296:
194:, and it usually differs chemically and physically from the pure enantiomers.
2850:
2713:
2565:
Symposium highlights-Session 5: New technologies for small molecule synthesis
2389:
2311:
2273:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
2101:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
2072:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
2043:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
1503:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
1474:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
1339:
1318:
1221:
1028:
981:
668:), but none of them has a mirror plane. In order to have a mirror plane, the
210:
151:
2405:
2282:
2110:
2081:
2052:
1675:
1512:
1483:
1156:
Chiral ligands confer chirality to a metal complex, as illustrated by metal-
190:. A homogeneous mixture of the two enantiomers in equal parts is said to be
2835:
2708:
2651:
2540:
1938:
1847:
1839:
1758:
1654:
1585:
1179:
1088:-(+)-carvone. The two smell different to most people because our olfactory
972:
681:
Chiral resolution#Spontaneous resolution and related specialized techniques
291:
287:
263:
255:
202:
171:
93:
2354:
2243:
2235:
1979:
1701:
1137:, chirality is pervasive and of practical importance. A famous example is
705:
2594:
The Handedness of the Universe by Roger A Hegstrom and Dilip K Kondepudi
2522:
1326:
1209:
1198:
820:
669:
381:
341:
2490:
10.1002/(SICI)1520-636X(1997)9:5/6<428::AID-CHIR5>3.0.CO;2-1
2448:
10.1002/(SICI)1520-636X(1997)9:5/6<428::AID-CHIR5>3.0.CO;2-1
2010:
10.1002/(sici)1520-636x(1997)9:5/6<428::aid-chir5>3.3.co;2-e
1618:
579:
232:). These types of chirality are far less common than central chirality.
2703:
1693:
1577:
1232:
1213:
1157:
1013:
1000:
960:
808:
766:
433:
345:
279:
206:
175:
35:
31:
26:
2325:
Friedman, L.; Miller, J. G. (1971). "Odor Incongruity and Chirality".
2147:
2123:
1740:
1278:
when its enantiomeric ratio is greater than 50:50 but less than 100:0.
784:
624:
perpendicular to line of three C, but not in the plane of the figure.)
247:
possesses helical chirality, which is one type of inherent chirality.
1321:
deduced in 1848 that this phenomenon has a molecular basis. The term
1057:
938:
913:
875:
Chirality is an intrinsic part of the identity of a molecule, so the
835:
824:
603:
565:
275:
164:
59:
645:
1225:
628:
551:
244:
243:
is a commonly cited example of a planar chiral molecule. Finally,
2434:
Eliel, Ernest Ludwig; Wilen, Samuel H.; Mander, Lewis N. (1994).
2185:
Eliel, Ernest Ludwig; Wilen, Samuel H.; Mander, Lewis N. (1994).
1249:
1217:
1075:
1067:
1032:
1017:
964:
946:
934:
676:
283:
259:
183:
1142:
1119:
1039:
774:
688:
360:
271:
267:
2436:"Chirality in Molecules Devoid of Chiral Centers (Chapter 14)"
2187:"Chirality in Molecules Devoid of Chiral Centers (Chapter 14)"
84:
1335:
1021:
2575:
Michigan State University's explanation of R/S nomenclature
1256:. Scalemic materials can be enantiopure or enantioenriched.
1149:, Greek "D") for a right-handed twist (pictured). Also cf.
142:
121:
78:
2630:
1430:(4th Edition) Paula Y. Bruice. Pearson Educational Books.
1224:
used this method to separate left-handed and right-handed
236:
is a typical example of an axially chiral molecule, while
2406:
Clayden, Jonathan; Greeves, Nick; Warren, Stuart (2012).
1722:
436:. Note that there is no meaning to the orientation of an
136:
130:
1345:
In the early 1970s, various groups established that the
19:"L-form" redirects here. For the bacterial strains, see
845:= 0.715°), even though the non-deuterated compound PhCH
2193:(1st ed.). New York, NY, USA: Wiley & Sons.
999:‑penicillamine is toxic as it inhibits the action of
118:
75:
1774:
1050:-forms of amino acids tend to be tasteless, whereas
967:
mixture. However, studies have shown that only the (
791:
is an example of a molecule with a stereogenic axis.
139:
133:
127:
81:
54:)-alanine (right) in zwitterionic form at neutral pH
2580:
Chirality & Odour Perception at leffingwell.com
1875:Srinivasarao, M. (1999). "Chirality and Polymers".
1725:"The Antipyridoxine Effect of Penicillamine in Man"
1313:in 1812, and gained considerable importance in the
975:) is responsible for the drug's beneficial effects.
124:
72:
2590:Chirality and the Search for Extraterrestrial Life
1877:Current Opinion in Colloid & Interface Science
1631:
2762:Ultraviolet–visible spectroscopy of stereoisomers
2570:IUPAC nomenclature for amino acid configurations.
2375:
1802:"Space radiation may select amino acids for life"
1770:
1768:
1212:of naturally occurring chiral compounds, such as
186:properties, except that they often have opposite
2848:
2296:"Zur Kenntnis des asymmetrischen Kobaltatoms. I"
1874:
2220:
1349:is capable of distinguishing chiral compounds.
841:PhCHDOH; which is chiral and optically active (
2324:
2300:Berichte der Deutschen Chemischen Gesellschaft
1765:
1723:JAFFE, IA; ALTMAN, K; MERRYMAN, P (Oct 1964).
900:
2616:
1317:, analytical chemistry, and pharmaceuticals.
1242:
1235:and the control of enantiomeric purity, e.g.
174:that are mirror images of each other, called
16:Geometric property of some molecules and ions
2585:Chirality & Bioactivity I.: Pharmacology
1985:
1868:
1860:: CS1 maint: multiple names: authors list (
1599:
1597:
1595:
311:The chirality of a molecule is based on the
2508:
2474:"Infelicitous Stereochemical Nomenclatures"
2471:
2168:
2074:enantiomerically enriched (enantioenriched)
1994:"Infelicitous Stereochemical Nomenclatures"
1991:
1625:
108:changes. This geometric property is called
2623:
2609:
2318:
1903:Stereochemistry of Coordination Compounds.
1401:for overview of stereochemistry in general
1201:and is expressed as the optical rotation.
761:), silicon, or a metal (as in many chiral
687:is cooled to the point of becoming chiral
2555:21st International Symposium on Chirality
1748:
1592:
1532:
1099:
753:), where swapping any two groups (e.g., C
167:example of an object with this property.
2369:
1669:
1167:
1103:
783:
704:
41:
25:
2121:
2103:enantiomer excess (enantiomeric excess)
1716:
1108:Delta-ruthenium-tris(bipyridine) cation
170:A chiral molecule or ion exists in two
2849:
2293:
2134:(2). Sage Publications Inc.: 147–154.
733:carbon bonded to four distinct groups
694:
250:Chirality is an important concept for
2604:
1799:
1777:Amino acids and the Asymmetry of Life
1729:The Journal of Clinical Investigation
1267:as a synonym is strongly discouraged.
367:-1,2-dichlorocyclohexane consists of
2440:Stereochemistry of Organic Compounds
2191:Stereochemistry of Organic Compounds
1557:
657:Note: This also has a mirror plane.
92:) if it cannot be superposed on its
2560:STEREOISOMERISM - OPTICAL ISOMERISM
1563:
13:
2399:
2270:Compendium of Chemical Terminology
2098:Compendium of Chemical Terminology
2069:Compendium of Chemical Terminology
2040:Compendium of Chemical Terminology
1500:Compendium of Chemical Terminology
1471:Compendium of Chemical Terminology
1007:
811:have stereogenic axes and exhibit
679:mixtures of chiral molecules (see
615:(Note: This molecule has only one
14:
2898:
2757:NMR spectroscopy of stereoisomers
2548:
1237:active pharmaceutical ingredients
709:Here, swapping of the two groups
462:Molecular symmetry and chirality
387:A chiral compound can contain no
2795:Diastereomeric recrystallization
1647:10.1097/00004850-200405000-00005
1204:Enantiomers can be separated by
920:-aspartame tastes sweet whereas
644:
627:
602:
578:
564:
550:
282:that are the building blocks of
114:
68:
2513:. Topics in Current Chemistry.
2287:
2258:
2214:
2178:
2162:
2115:
2086:
2057:
2028:
1961:
1932:
1916:
1895:
1818:
1793:
1682:Journal of Neural Transmission
1526:
1517:
1488:
1459:
1440:
1421:
1114:Complex (chemistry): Isomerism
479:Improper rotational elements (
352:conformers that belong to the
299:usually have vastly different
258:. Most substances relevant to
150:). The terms are derived from
62:, a molecule or ion is called
1:
2347:10.1126/science.172.3987.1044
1889:10.1016/S1359-0294(99)00024-2
1775:Meierhenrich, Uwe J. (2008).
1415:
1139:tris(bipyridine)ruthenium(II)
683:), or as when achiral liquid
306:
2790:Chiral column chromatography
1956:10.1016/j.chroma.2009.12.071
1800:McKee, Maggie (2005-08-24).
1551:10.1016/0040-4020(59)80014-4
1359:Chirality (electromagnetism)
1066:-enantiomer of the chemical
1056:-forms tend to taste sweet.
7:
1352:
1252:chiral substance is called
1182:form, of an optical isomer
901:Manifestations of chirality
224:) and a stereogenic plane (
10:
2903:
2752:Chiral derivatizing agents
2633:enantioselective synthesis
2140:10.1177/030631277600600201
1389:Orientation (vector space)
1379:Enantioselective synthesis
1304:
1243:Miscellaneous nomenclature
1151:dextro- and levo- (laevo-)
1111:
698:
154:
18:
2803:
2770:
2739:
2691:
2639:
2128:Social Studies of Science
1905:Chichester: John Wiley..
1901:von Zelewsky, A. (1995).
1779:. Berlin, GER: Springer.
1239:(APIs) which are chiral.
1003:, an essential B vitamin.
991:and for the treatment of
945:-(+)-carvone smells like
478:
389:improper axis of rotation
2678:Supramolecular chirality
2390:10.1002/hlca.19800630721
2312:10.1002/cber.19110440297
2122:Frankel, Eugene (1976).
1635:Int Clin Psychopharmacol
1410:Supramolecular chirality
1226:sodium ammonium tartrate
1188:linearly polarized light
1135:organometallic chemistry
1016:(the building blocks of
924:-aspartame is tasteless.
879:includes details of the
807:(BINOL) and 1,3-dichloro
2294:Werner, A. (May 1911).
2283:10.1351/goldbook.O04308
2111:10.1351/goldbook.E02070
2082:10.1351/goldbook.E02071
2053:10.1351/goldbook.E02072
1513:10.1351/goldbook.S06144
1484:10.1351/goldbook.C01058
1364:Chirality (mathematics)
1186:the plane of a beam of
163:) 'hand'; which is the
2378:Helvetica Chimica Acta
1840:10.1002/anie.200501311
1270:A chiral substance is
1259:A chiral substance is
1131:coordination chemistry
1109:
1100:In inorganic chemistry
881:absolute configuration
792:
763:coordination compounds
718:
666:conformational isomers
290:. Naturally occurring
278:), all but one of the
180:absolute configuration
96:by any combination of
55:
50:)-Alanine (left) and (
39:
2872:Chemical nomenclature
2816:Chiral pool synthesis
2730:Diastereomeric excess
2236:10.1353/pbm.1995.0069
1980:10.1002/tcr.202100295
1347:human olfactory organ
1325:itself was coined by
1168:Methods and practices
1107:
916:has two enantiomers.
787:
708:
45:
29:
2862:Polarization (waves)
2826:Asymmetric catalysis
2811:Asymmetric induction
2523:10.1007/128_2013_435
2472:Eliel, E.L. (1997).
2224:Perspect. Biol. Med.
2169:Pasteur, L. (1848).
2045:asymmetric synthesis
1992:Eliel, E.L. (1997).
1828:Angew. Chem. Int. Ed
1607:J. Agric. Food Chem.
1162:asymmetric catalysis
993:rheumatoid arthritis
911:artificial sweetener
410:Schoenflies notation
262:are chiral, such as
2724:Enantiomeric excess
2339:1971Sci...172.1044F
2333:(3987): 1044–1046.
1680:)-(+)-enantiomer".
1619:10.1021/jf60176a035
1369:Chirality (physics)
1282:Enantiomeric excess
1193:. The (+)-form, or
1060:leaves contain the
1027:The origin of this
953:Drug effectiveness:
779:S-chiral sulfoxides
771:P-chiral phosphines
695:Stereogenic centers
463:
374:pyramidal inversion
2821:Chiral auxiliaries
2785:Kinetic resolution
2683:Inherent chirality
2668:-symmetric ligands
1694:10.1007/BF01244820
1578:10.1002/chir.22071
1311:Jean-Baptiste Biot
1300:(70% − 30% = 40%).
1110:
1078:seeds contain the
971:)-(+) enantiomer (
895:other designations
859:nitrogen inversion
805:1,1′-bi-2-naphthol
802:-symmetric species
793:
789:1,1′-Bi-2-naphthol
723:stereogenic center
719:
701:Stereogenic center
461:
313:molecular symmetry
230:inherent chirality
188:optical activities
56:
40:
2844:
2843:
2780:Recrystallization
2772:Chiral resolution
2532:978-3-319-03238-2
2409:Organic Chemistry
1834:(35): 5630–5634.
1741:10.1172/JCI105060
1447:Organic Chemistry
1428:Organic Chemistry
1206:chiral resolution
1074:-(−)-carvone and
998:
989:chelation therapy
984:
923:
919:
891:
864:When the optical
661:
660:
2894:
2747:Optical rotation
2692:Chiral molecules
2657:Planar chirality
2625:
2618:
2611:
2602:
2601:
2544:
2505:
2503:
2501:
2492:. Archived from
2484:(5–6): 428–430.
2468:
2466:
2464:
2430:
2428:
2426:
2394:
2393:
2384:(7): 1932–1946.
2373:
2367:
2366:
2322:
2316:
2315:
2306:(2): 1887–1898.
2291:
2285:
2262:
2256:
2255:
2218:
2212:
2211:
2209:
2207:
2182:
2176:
2175:
2166:
2160:
2159:
2119:
2113:
2090:
2084:
2061:
2055:
2032:
2026:
2025:
2023:
2021:
2012:. Archived from
1989:
1983:
1965:
1959:
1944:J. Chromatogr. A
1936:
1930:
1920:
1914:
1899:
1893:
1892:
1872:
1866:
1865:
1859:
1851:
1822:
1816:
1815:
1813:
1812:
1797:
1791:
1790:
1772:
1763:
1762:
1752:
1720:
1714:
1713:
1673:
1667:
1666:
1629:
1623:
1622:
1601:
1590:
1589:
1561:
1555:
1554:
1535:
1530:
1524:
1521:
1515:
1492:
1486:
1463:
1457:
1444:
1438:
1425:
1374:Enantiopure drug
1191:counterclockwise
1175:optical activity
1129:In the areas of
1124:nonlinear optics
1083:
1082:
1065:
1064:
1055:
1054:
1049:
1048:
996:
982:
921:
917:
889:
829:planar chirality
759:organophosphates
648:
631:
606:
582:
568:
554:
526:inversion center
464:
460:
369:chair conformers
226:planar chirality
158:
149:
148:
145:
144:
141:
138:
135:
132:
129:
126:
123:
120:
91:
90:
87:
86:
83:
80:
77:
74:
2902:
2901:
2897:
2896:
2895:
2893:
2892:
2891:
2857:Stereochemistry
2847:
2846:
2845:
2840:
2831:Organocatalysis
2799:
2766:
2735:
2719:Racemic mixture
2687:
2673:Axial chirality
2667:
2640:Chirality types
2635:
2629:
2551:
2533:
2499:
2497:
2496:on 3 March 2016
2462:
2460:
2458:
2424:
2422:
2420:
2402:
2400:Further reading
2397:
2374:
2370:
2323:
2319:
2292:
2288:
2275:Optical isomers
2263:
2259:
2219:
2215:
2205:
2203:
2201:
2183:
2179:
2167:
2163:
2120:
2116:
2091:
2087:
2062:
2058:
2033:
2029:
2019:
2017:
2016:on 3 March 2016
2004:(56): 428–430.
1990:
1986:
1974:, e102100295. (
1966:
1962:
1950:, 1395–1398. (
1937:
1933:
1921:
1917:
1900:
1896:
1873:
1869:
1853:
1852:
1823:
1819:
1810:
1808:
1798:
1794:
1787:
1773:
1766:
1735:(10): 1869–73.
1721:
1717:
1674:
1670:
1630:
1626:
1602:
1593:
1572:(12): 959–976.
1562:
1558:
1531:
1527:
1522:
1518:
1505:Superposability
1493:
1489:
1464:
1460:
1445:
1441:
1426:
1422:
1418:
1405:Stereoisomerism
1399:Stereochemistry
1394:Pfeiffer effect
1355:
1331:optical isomers
1307:
1272:enantioenriched
1245:
1170:
1116:
1102:
1084:-enantiomer or
1080:
1079:
1062:
1061:
1052:
1051:
1046:
1045:
1010:
1008:In biochemistry
903:
877:systematic name
870:cryptochirality
848:
844:
813:axial chirality
801:
703:
697:
685:silicon dioxide
656:
655:
649:
641:
632:
623:
621:
614:
613:
607:
599:
588:
583:
574:
569:
561:
555:
547:
533:
527:
525:
518:
512:
510:
503:
497:
484:
474:
468:
457:
451:or the achiral
450:
442:
431:
427:
423:
419:
403:
396:
358:
327:
320:
309:
252:stereochemistry
222:axial chirality
117:
113:
71:
67:
38:that are chiral
24:
21:L-form bacteria
17:
12:
11:
5:
2900:
2890:
2889:
2884:
2882:Origin of life
2879:
2874:
2869:
2864:
2859:
2842:
2841:
2839:
2838:
2833:
2828:
2823:
2818:
2813:
2807:
2805:
2801:
2800:
2798:
2797:
2792:
2787:
2782:
2776:
2774:
2768:
2767:
2765:
2764:
2759:
2754:
2749:
2743:
2741:
2737:
2736:
2734:
2733:
2727:
2721:
2716:
2711:
2706:
2701:
2695:
2693:
2689:
2688:
2686:
2685:
2680:
2675:
2670:
2665:
2659:
2654:
2649:
2643:
2641:
2637:
2636:
2628:
2627:
2620:
2613:
2605:
2599:
2598:
2592:
2587:
2582:
2577:
2572:
2567:
2562:
2557:
2550:
2549:External links
2547:
2546:
2545:
2531:
2506:
2469:
2457:978-0471016700
2456:
2431:
2419:978-0199270293
2418:
2401:
2398:
2396:
2395:
2368:
2317:
2286:
2257:
2230:(2): 188–229.
2213:
2200:978-0471016700
2199:
2177:
2161:
2114:
2085:
2056:
2027:
1984:
1960:
1931:
1915:
1894:
1883:(5): 369–376.
1867:
1817:
1792:
1786:978-3540768852
1785:
1764:
1715:
1688:(2): 157–160.
1668:
1624:
1613:(4): 785–787.
1591:
1556:
1545:(4): 338–344.
1525:
1516:
1487:
1458:
1439:
1419:
1417:
1414:
1413:
1412:
1407:
1402:
1396:
1391:
1386:
1381:
1376:
1371:
1366:
1361:
1354:
1351:
1315:sugar industry
1306:
1303:
1302:
1301:
1279:
1268:
1257:
1244:
1241:
1195:dextrorotatory
1169:
1166:
1112:Main article:
1101:
1098:
1009:
1006:
1005:
1004:
985:‑penicillamine
976:
957:antidepressant
950:
925:
902:
899:
855:energy barrier
846:
842:
839:benzyl alcohol
799:
699:Main article:
696:
693:
659:
658:
653:
642:
636:
625:
619:
611:
600:
597:
591:
590:
586:
576:
572:
562:
559:
548:
545:
539:
538:
531:
520:
516:
505:
501:
492:
488:
487:
482:
477:
472:
455:
448:
440:
429:
425:
421:
417:
401:
394:
382:atropisomerism
356:
325:
318:
308:
305:
297:pharmaceutical
106:conformational
15:
9:
6:
4:
3:
2:
2899:
2888:
2885:
2883:
2880:
2878:
2875:
2873:
2870:
2868:
2865:
2863:
2860:
2858:
2855:
2854:
2852:
2837:
2834:
2832:
2829:
2827:
2824:
2822:
2819:
2817:
2814:
2812:
2809:
2808:
2806:
2802:
2796:
2793:
2791:
2788:
2786:
2783:
2781:
2778:
2777:
2775:
2773:
2769:
2763:
2760:
2758:
2755:
2753:
2750:
2748:
2745:
2744:
2742:
2738:
2731:
2728:
2725:
2722:
2720:
2717:
2715:
2714:Meso compound
2712:
2710:
2707:
2705:
2702:
2700:
2697:
2696:
2694:
2690:
2684:
2681:
2679:
2676:
2674:
2671:
2669:
2664:
2660:
2658:
2655:
2653:
2650:
2648:
2645:
2644:
2642:
2638:
2634:
2626:
2621:
2619:
2614:
2612:
2607:
2606:
2603:
2597:
2593:
2591:
2588:
2586:
2583:
2581:
2578:
2576:
2573:
2571:
2568:
2566:
2563:
2561:
2558:
2556:
2553:
2552:
2542:
2538:
2534:
2528:
2524:
2520:
2516:
2512:
2507:
2495:
2491:
2487:
2483:
2479:
2475:
2470:
2459:
2453:
2449:
2445:
2441:
2437:
2432:
2421:
2415:
2411:
2410:
2404:
2403:
2391:
2387:
2383:
2379:
2372:
2364:
2360:
2356:
2352:
2348:
2344:
2340:
2336:
2332:
2328:
2321:
2313:
2309:
2305:
2302:(in German).
2301:
2297:
2290:
2284:
2280:
2276:
2272:
2271:
2266:
2261:
2253:
2249:
2245:
2241:
2237:
2233:
2229:
2226:
2225:
2217:
2202:
2196:
2192:
2188:
2181:
2173:
2165:
2157:
2153:
2149:
2145:
2141:
2137:
2133:
2129:
2125:
2118:
2112:
2108:
2104:
2100:
2099:
2094:
2089:
2083:
2079:
2075:
2071:
2070:
2065:
2060:
2054:
2050:
2046:
2042:
2041:
2036:
2031:
2015:
2011:
2007:
2003:
1999:
1995:
1988:
1981:
1977:
1973:
1970:
1967:Ravi Bhushan
1964:
1957:
1953:
1949:
1946:
1945:
1941:; Tanwar, S.
1940:
1935:
1929:
1925:
1919:
1912:
1908:
1904:
1898:
1890:
1886:
1882:
1878:
1871:
1863:
1857:
1849:
1845:
1841:
1837:
1833:
1829:
1821:
1807:
1806:New Scientist
1803:
1796:
1788:
1782:
1778:
1771:
1769:
1760:
1756:
1751:
1746:
1742:
1738:
1734:
1730:
1726:
1719:
1711:
1707:
1703:
1699:
1695:
1691:
1687:
1683:
1679:
1672:
1664:
1660:
1656:
1652:
1648:
1644:
1641:(3): 149–55.
1640:
1636:
1628:
1620:
1616:
1612:
1609:
1608:
1600:
1598:
1596:
1587:
1583:
1579:
1575:
1571:
1567:
1560:
1552:
1548:
1544:
1540:
1534:
1529:
1520:
1514:
1510:
1506:
1502:
1501:
1496:
1491:
1485:
1481:
1477:
1473:
1472:
1467:
1462:
1456:
1452:
1448:
1443:
1437:
1436:9780131407480
1433:
1429:
1424:
1420:
1411:
1408:
1406:
1403:
1400:
1397:
1395:
1392:
1390:
1387:
1385:
1382:
1380:
1377:
1375:
1372:
1370:
1367:
1365:
1362:
1360:
1357:
1356:
1350:
1348:
1343:
1341:
1340:Alfred Werner
1337:
1332:
1328:
1324:
1320:
1319:Louis Pasteur
1316:
1312:
1299:
1295:
1292:contains 70%
1291:
1287:
1283:
1280:
1277:
1273:
1269:
1266:
1262:
1258:
1255:
1251:
1247:
1246:
1240:
1238:
1234:
1229:
1227:
1223:
1222:Louis Pasteur
1219:
1216:or the amine
1215:
1211:
1207:
1202:
1200:
1196:
1192:
1189:
1185:
1181:
1177:
1176:
1165:
1163:
1159:
1154:
1152:
1148:
1144:
1140:
1136:
1132:
1127:
1125:
1121:
1115:
1106:
1097:
1093:
1091:
1087:
1077:
1073:
1069:
1059:
1043:
1041:
1037:
1034:
1030:
1029:homochirality
1025:
1023:
1019:
1015:
1002:
994:
990:
986:
980:
977:
974:
970:
966:
963:is sold as a
962:
958:
954:
951:
948:
944:
940:
936:
932:
929:
926:
915:
912:
908:
905:
904:
898:
896:
892:
886:
882:
878:
873:
871:
867:
862:
860:
856:
850:
840:
837:
832:
830:
826:
822:
818:
814:
810:
806:
803:
790:
786:
782:
780:
776:
773:(PRR′R″) and
772:
768:
764:
760:
756:
752:
748:
744:
740:
736:
732:
728:
724:
716:
712:
707:
702:
692:
690:
686:
682:
678:
673:
671:
667:
652:
647:
643:
640:
635:
630:
626:
618:
610:
605:
601:
596:
593:
592:
589:
581:
577:
575:
567:
563:
558:
553:
549:
544:
541:
540:
537:
530:
524:
521:
515:
509:
506:
504:
496:
493:
490:
489:
485:
475:
466:
465:
459:
454:
447:
439:
435:
415:
411:
406:
404:
397:
390:
385:
383:
377:
375:
370:
366:
362:
359:point group,
355:
351:
347:
343:
340:
336:
332:
328:
321:
314:
304:
302:
298:
293:
292:triglycerides
289:
288:nucleic acids
285:
281:
277:
273:
269:
265:
264:carbohydrates
261:
257:
253:
248:
246:
242:
240:
235:
231:
227:
223:
217:
215:
213:
208:
204:
203:diastereomers
199:
195:
193:
189:
185:
181:
177:
173:
172:stereoisomers
168:
166:
162:
157:
153:
152:Ancient Greek
147:
111:
107:
103:
99:
95:
89:
65:
61:
53:
49:
44:
37:
34:of a generic
33:
28:
22:
2887:Pharmacology
2877:Biochemistry
2836:Biocatalysis
2709:Diastereomer
2699:Stereoisomer
2662:
2652:Stereocenter
2646:
2631:Concepts in
2514:
2510:
2498:. Retrieved
2494:the original
2481:
2477:
2461:. Retrieved
2439:
2423:. Retrieved
2408:
2381:
2377:
2371:
2330:
2326:
2320:
2303:
2299:
2289:
2268:
2260:
2227:
2222:
2216:
2204:. Retrieved
2190:
2180:
2170:
2164:
2131:
2127:
2117:
2096:
2088:
2067:
2059:
2038:
2030:
2018:. Retrieved
2014:the original
2001:
1997:
1987:
1971:
1968:
1963:
1947:
1942:
1934:
1918:
1902:
1897:
1880:
1876:
1870:
1856:cite journal
1831:
1827:
1820:
1809:. Retrieved
1805:
1795:
1776:
1732:
1728:
1718:
1685:
1681:
1677:
1671:
1638:
1634:
1627:
1610:
1605:
1569:
1565:
1559:
1542:
1538:
1533:
1528:
1519:
1498:
1490:
1469:
1461:
1446:
1442:
1427:
1423:
1344:
1330:
1322:
1308:
1297:
1293:
1289:
1285:
1276:heterochiral
1275:
1271:
1264:
1260:
1253:
1230:
1203:
1180:levorotatory
1173:
1171:
1155:
1128:
1117:
1094:
1092:are chiral.
1085:
1071:
1044:
1038:
1026:
1011:
979:Drug safety:
978:
973:escitalopram
968:
952:
942:
937:smells like
930:
927:
906:
888:
884:
874:
863:
851:
833:
816:
794:
754:
750:
746:
742:
738:
734:
727:stereocenter
726:
722:
720:
714:
710:
674:
662:
650:
638:
633:
616:
608:
594:
584:
570:
556:
542:
535:
528:
522:
513:
511:mirror plane
507:
499:
494:
480:
470:
452:
445:
437:
407:
399:
392:
386:
378:
364:
353:
349:
346:enantiomeric
342:point groups
338:
334:
330:
323:
316:
310:
303:or effects.
256:biochemistry
249:
241:-cyclooctene
238:
218:
211:
200:
196:
169:
160:
109:
102:translations
94:mirror image
63:
57:
51:
47:
1939:Bhushan, R.
1539:Tetrahedron
1327:Lord Kelvin
1261:enantiopure
1233:enantiomers
1210:chiral pool
1199:polarimeter
1014:amino acids
987:is used in
849:OH is not.
821:cyclooctene
731:tetrahedral
670:cyclohexane
434:enantiomers
414:point group
280:amino acids
207:enantiomers
176:enantiomers
104:, and some
32:enantiomers
2851:Categories
2704:Enantiomer
2500:5 February
2463:2 February
2425:2 February
2206:2 February
2020:5 February
1969:Chem. Rec.
1928:189138953X
1911:047195599X
1811:2016-02-05
1455:0763721972
1416:References
1384:Handedness
1265:homochiral
1214:malic acid
1158:amino acid
1001:pyridoxine
961:citalopram
836:deuterated
767:phosphorus
467:Rotational
432:represent
307:Definition
286:, and the
36:amino acid
2867:Chirality
2804:Reactions
2647:Chirality
2511:Chirality
2478:Chirality
2156:122887123
1998:Chirality
1566:Chirality
1476:Chirality
1342:in 1911.
1323:chirality
1172:The term
1090:receptors
1058:Spearmint
939:spearmint
914:aspartame
825:ferrocene
823:and many
815:, while (
301:potencies
276:cellulose
165:canonical
110:chirality
98:rotations
60:chemistry
2740:Analysis
2541:23666078
2517:: 1–20.
2363:25725148
2252:46514372
1848:16035020
1759:14236210
1710:20110906
1663:36768144
1655:15107657
1586:23034823
1353:See also
1296:and 30%
1254:scalemic
1248:Any non-
1018:proteins
995:whereas
941:whereas
866:rotation
284:proteins
245:helicene
214:compound
184:physical
2355:5573954
2335:Bibcode
2327:Science
2244:7899056
1702:1632943
1305:History
1250:racemic
1218:brucine
1184:rotates
1076:caraway
1068:carvone
1040:Enzymes
1033:biology
965:racemic
947:caraway
935:carvone
907:Flavor:
677:racemic
523:Achiral
508:Achiral
412:of the
260:biology
192:racemic
2539:
2529:
2454:
2416:
2361:
2353:
2250:
2242:
2197:
2154:
2148:284930
2146:
1926:
1909:
1846:
1783:
1757:
1750:289631
1747:
1708:
1700:
1661:
1653:
1584:
1453:
1434:
1143:lambda
1120:Quartz
1022:sugars
1020:) and
809:allene
775:sulfur
745:, and
689:quartz
495:Chiral
491:
469:axis (
361:butane
350:gauche
274:, and
272:starch
268:sugars
64:chiral
2359:S2CID
2265:IUPAC
2248:S2CID
2152:S2CID
2144:JSTOR
2093:IUPAC
2064:IUPAC
2035:IUPAC
1706:S2CID
1659:S2CID
1495:IUPAC
1466:IUPAC
1338:, by
1336:hexol
1147:delta
959:drug
933:-(–)-
928:Odor:
893:, or
622:axis:
428:and Y
420:and X
239:trans
234:BINOL
161:cheir
2732:(de)
2726:(ee)
2537:PMID
2527:ISBN
2502:2016
2465:2016
2452:ISBN
2427:2016
2414:ISBN
2351:PMID
2240:PMID
2208:2016
2195:ISBN
2172:book
2022:2016
1972:2022
1948:2010
1924:ISBN
1907:ISBN
1862:link
1844:PMID
1781:ISBN
1755:PMID
1698:PMID
1651:PMID
1582:PMID
1451:ISBN
1432:ISBN
1286:e.e.
1133:and
955:the
909:the
857:for
755:bacd
751:abcd
725:(or
713:and
424:or Y
254:and
212:meso
205:and
156:χείρ
30:Two
2519:doi
2515:340
2486:doi
2444:doi
2386:doi
2343:doi
2331:172
2308:doi
2279:doi
2277:".
2232:doi
2136:doi
2107:doi
2105:".
2078:doi
2076:".
2049:doi
2047:".
2006:doi
1976:doi
1952:doi
1885:doi
1836:doi
1745:PMC
1737:doi
1690:doi
1643:doi
1615:doi
1574:doi
1547:doi
1509:doi
1507:".
1480:doi
1478:".
1284:or
1274:or
1070:or
1031:in
897:).
890:D/L
885:R/S
777:in
769:in
519:= σ
498:no
365:cis
58:In
2853::
2535:.
2525:.
2480:.
2476:.
2450:.
2438:.
2382:63
2380:.
2357:.
2349:.
2341:.
2329:.
2304:44
2298:.
2267:,
2246:.
2238:.
2228:38
2189:.
2150:.
2142:.
2130:.
2126:.
2095:,
2066:,
2037:,
2000:.
1996:.
1879:.
1858:}}
1854:{{
1842:.
1832:44
1830:.
1804:.
1767:^
1753:.
1743:.
1733:43
1731:.
1727:.
1704:.
1696:.
1686:88
1684:.
1657:.
1649:.
1639:19
1637:.
1611:19
1594:^
1580:.
1570:24
1568:.
1541:.
1497:,
1468:,
1164:.
1153:.
1126:.
1024:.
887:,
872:.
861:.
831:.
819:)-
749:(C
741:,
737:,
721:A
691:.
654:2h
534:=
486:)
458:.
384:.
376:.
337:,
333:,
329:,
322:,
270:,
216:.
122:aɪ
100:,
85:əl
79:aɪ
2666:2
2663:C
2624:e
2617:t
2610:v
2543:.
2521::
2504:.
2488::
2482:9
2467:.
2446::
2429:.
2392:.
2388::
2365:.
2345::
2337::
2314:.
2310::
2281::
2254:.
2234::
2210:.
2174:.
2158:.
2138::
2132:6
2109::
2080::
2051::
2024:.
2008::
2002:9
1982:)
1978::
1958:)
1954::
1913:.
1891:.
1887::
1881:4
1864:)
1850:.
1838::
1814:.
1789:.
1761:.
1739::
1712:.
1692::
1678:S
1665:.
1645::
1621:.
1617::
1588:.
1576::
1553:.
1549::
1543:6
1511::
1482::
1298:S
1294:R
1290:R
1086:S
1081:D
1072:R
1063:L
1053:D
1047:L
997:L
983:D
969:S
949:.
943:S
931:R
922:D
918:L
883:(
847:2
843:D
817:E
800:2
798:C
747:d
743:c
739:b
735:a
715:b
711:a
664:(
651:C
639:v
637:2
634:C
620:2
617:C
612:2
609:C
598:2
595:C
587:i
585:C
573:s
571:C
560:1
557:C
546:1
543:C
536:i
532:2
529:S
517:1
514:S
502:n
500:S
483:n
481:S
476:)
473:n
471:C
456:4
453:S
449:3
446:C
441:2
438:S
430:S
426:R
422:S
418:R
402:n
400:S
395:n
393:S
391:(
357:2
354:C
339:I
335:O
331:T
326:n
324:D
319:n
317:C
266:(
220:(
159:(
146:/
143:i
140:t
137:ɪ
134:l
131:æ
128:r
125:ˈ
119:k
116:/
112:(
88:/
82:r
76:k
73:ˈ
70:/
66:(
52:R
48:S
46:(
23:.
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.