2891:
3102:
1740:
3703:
3803:
3761:
4077:
503:
3562:
2931:
3451:
4144:
3432:
4235:
4096:
4492:
3879:
4511:
4423:
4255:
4216:
3995:
4171:
4461:
4399:
4035:
3925:
3898:
3860:
3730:
3261:
3143:
748:
4288:
3684:
4371:
4119:
4054:
4012:
3944:
3841:
3661:
3634:
3520:
3972:
1626:
1619:
3539:
3478:
4327:
3822:
3599:
4442:
27:
4307:
3780:
4347:
4190:
3497:
1920:
460:" e.g. as formate, acetate, propionate, and butyrate, as opposed to the IUPAC nomenclature methanoate, ethanoate, propanoate, and butanoate. Esters derived from more complex carboxylic acids are, on the other hand, more frequently named using the systematic IUPAC name, based on the name for the acid followed by the suffix
1546:, carboxylic acid esters are structurally flexible functional groups because rotation about the C–O–C bonds has a low barrier. Their flexibility and low polarity is manifested in their physical properties; they tend to be less rigid (lower melting point) and more volatile (lower boiling point) than the corresponding
3203:
Esters react with nucleophiles at the carbonyl carbon. The carbonyl is weakly electrophilic but is attacked by strong nucleophiles (amines, alkoxides, hydride sources, organolithium compounds, etc.). The C–H bonds adjacent to the carbonyl are weakly acidic but undergo deprotonation with strong bases.
276:
There are compounds in which an acidic hydrogen of acids mentioned in this article are not replaced by an organyl, but by some other group. According to some authors, those compounds are esters as well, especially when the first carbon atom of the organyl group replacing acidic hydrogen, is replaced
4972:
Many mineral and organic acids containing oxygen combine with an alcohol upon elimination of water to neutral, volatile ether compounds, which one can view as coupled compounds of alcohol and acid-water, or, according to the theory of radicals, as salts in which an acid is bonded with an ether.
4958:
Viele mineralische und organische
Sauerstoffsäuren treten mit einer Alkohol-Art unter Ausscheidung von Wasser zu neutralen flüchtigen ätherischen Verbindungen zusammen, welche man als gepaarte Verbindungen von Alkohol und Säuren-Wasser oder, nach der Radicaltheorie, als Salze betrachten kann, in
1654:
as hydrogen-bond acceptors, but cannot act as hydrogen-bond donors, unlike their parent alcohols. This ability to participate in hydrogen bonding confers some water-solubility. Because of their lack of hydrogen-bond-donating ability, esters do not self-associate. Consequently, esters are more
2849:. Transesterification can be either acid- or base-catalyzed, and involves the reaction of an ester with an alcohol. Unfortunately, because the leaving group is also an alcohol, the forward and reverse reactions will often occur at similar rates. Using a large excess of the
3215:, the hydrogen atoms on the carbon adjacent ("α to") the carboxyl group in esters are sufficiently acidic to undergo deprotonation, which in turn leads to a variety of useful reactions. Deprotonation requires relatively strong bases, such as
3204:
This process is the one that usually initiates condensation reactions. The carbonyl oxygen in esters is weakly basic, less so than the carbonyl oxygen in amides due to resonance donation of an electron pair from nitrogen in amides, but forms
5097:
A. A. Yakovenko; J. H. Gallegos; M. Yu. Antipin; A. Masunov; T. V. Timofeeva (2011). "Crystal
Morphology as an Evidence of Supramolecular Organization in Adducts of 1,2-Bis(chloromercurio)tetrafluorobenzene with Organic Esters".
2890:
5535:
Yato, Michihisa; Homma, Koichi; Ishida, Akihiko (June 2001). "Reduction of carboxylic esters to ethers with triethyl silane in the combined use of titanium tetrachloride and trimethylsilyl trifluoromethanesulfonate".
3407:
Many esters have distinctive fruit-like odors, and many occur naturally in the essential oils of plants. This has also led to their common use in artificial flavorings and fragrances which aim to mimic those odors.
2886:
values, the forward and reverse reactions compete with each other. As in transesterification, using a large excess of reactant (water) or removing one of the products (the alcohol) can promote the forward reaction.
5563:
Sakai, Norio; Moriya, Toshimitsu; Konakahara, Takeo (July 2007). "An
Efficient One-Pot Synthesis of Unsymmetrical Ethers: A Directly Reductive Deoxygenation of Esters Using an InBr3/Et3SiH Catalytic System".
3101:
1674:. This peak changes depending on the functional groups attached to the carbonyl. For example, a benzene ring or double bond in conjunction with the carbonyl will bring the wavenumber down about 30 cm.
2902:, is not an equilibrium process; a full equivalent of base is consumed in the reaction, which produces one equivalent of alcohol and one equivalent of a carboxylate salt. The saponification of esters of
5253:
Raber, Douglas J.; Gariano, Jr, Patrick; Brod, Albert O.; Gariano, Anne L.; Guida, Wayne C. (1977). "Esterification of
Carboxylic Acids with Trialkyloxonium Salts: Ethyl and Methyl 4-Acetoxybenzoates".
452:
The names of esters that are formed from an alcohol and an acid, are derived from the parent alcohol and the parent acid, where the latter may be organic or inorganic. Esters derived from the simplest
3117:
or
Dieckmann cyclization, since it can be used to form rings. Esters can also undergo condensations with ketone and aldehyde enolates to give β-dicarbonyl compounds. A specific example of this is the
2228:, catalyze the reaction of a recalcitrant alkyl halide. Alternatively, salts of a coordinating metal, such as silver, may improve the reaction rate by easing halide elimination.
5194:
Williams, Roger J.; Gabriel, Alton; Andrews, Roy C. (1928). "The
Relation Between the Hydrolysis Equilibrium Constant of Esters and the Strengths of the Corresponding Acids".
3074:
Esters can undergo a variety of reactions with carbon nucleophiles. They react with an excess of a
Grignard reagent to give tertiary alcohols. Esters also react readily with
908:
298:
5281:
Matsumoto, Kouichi; Shimazaki, Hayato; Miyamoto, Yu; Shimada, Kazuaki; Haga, Fumi; Yamada, Yuki; Miyazawa, Hirotsugu; Nishiwaki, Keiji; Kashimura, Shigenori (2014).
1798:. Esters are common in organic chemistry and biological materials, and often have a pleasant characteristic, fruity odor. This leads to their extensive use in the
135:
are cyclic carboxylic esters; naturally occurring lactones are mainly 5- and 6-membered ring lactones. Lactones contribute to the aroma of fruits, butter, cheese,
5473:
Makhova, Irina V.; Elinson, Michail N.; Nikishin, Gennady I. (1991). "Electrochemical oxidation of ketones in methanol in the presence of alkali metal bromides".
4485:
3872:
4504:
3670:
4028:
3853:
5643:
5332:
5161:
4823:
4281:
4047:
3796:
5446:
Neumeister, Joachim; Keul, Helmut; Pratap Saxena, Mahendra; Griesbaum, Karl (1978). "Ozone
Cleavage of Olefins with Formation of Ester Fragments".
4580:
4320:
1848:
The equilibrium constant for such reactions is about 5 for typical esters, e.g., ethyl acetate. The reaction is slow in the absence of a catalyst.
213:
Organyl esters of carboxylic acids typically have a pleasant smell; those of low molecular weight are commonly used as fragrances and are found in
5023:
Diwakar M. Pawar; Abdelnaser A. Khalil; Denise R. Hooks; Kenneth
Collins; Tijuana Elliott; Jefforey Stafford; Lucille Smith; Eric A. Noe (1998). "
4921:
2998:. The introduction of catalytic hydrogenation in the early part of the 20th century was a breakthrough; esters of fatty acids are hydrogenated to
2129:. Since acyl chlorides and acid anhydrides also react with water, anhydrous conditions are preferred. The analogous acylations of amines to give
2028:
Using this diazomethane, mixtures of carboxylic acids can be converted to their methyl esters in near quantitative yields, e.g., for analysis by
1867:
Using a dehydrating agent: sulfuric acid not only catalyzes the reaction but sequesters water (a reaction product). Other drying agents such as
97:
belong to the ester category as well. According to some authors, organyl derivatives of acidic hydrogen of other acids are esters as well (e.g.
2032:. The method is useful in specialized organic synthetic operations but is considered too hazardous and expensive for large-scale applications.
3062:
tends to decompose to give an alcohol and an aldehyde (which is rapidly reduced to give a second alcohol). The reaction can be achieved using
3125:-acyloxy ketone undergoes an intramolecular nucleophilic acyl substitution and subsequent rearrangement to form an aromatic β-diketone. The
2837:
and primary and secondary amines to give amides, although this type of reaction is not often used, since acid halides give better yields.
5513:
5707:
447:
5060:
Christophe Dugave; Luc
Demange (2003). "Cis−Trans Isomerization of Organic Molecules and Biomolecules: Implications and Applications".
5009:
6626:
4907:
1560:
of the alpha-hydrogens on esters of carboxylic acids is around 25 (alpha-hydrogen is a hydrogen bound to the carbon adjacent to the
5033:
6631:
2967:
This reaction is not usually reversible. Hydrazines and hydroxylamine can be used in place of amines. Esters can be converted to
5282:
4891:
2894:
The acid-catalyzed hydrolysis of an ester and Fischer esterification correspond to two directions of an equilibrium process.
3118:
2137:
and react more rapidly than does water. This method is employed only for laboratory-scale procedures, as it is expensive.
5178:
2879:
reaction. Because an alcohol (which acts as the leaving group) and water (which acts as the nucleophile) have similar p
2833:
Esters are less reactive than acid halides and anhydrides. As with more reactive acyl derivatives, they can react with
2265:
Like the hydrolysation, transesterification is catalysed by acids and bases. The reaction is widely used for degrading
2145:
5349:
5283:"Simple and Convenient Synthesis of Esters from Carboxylic Acids and Alkyl Halides Using Tetrabutylammonium Fluoride"
4996:
4864:
3300:
3190:
2995:
1013:
4883:
CRC Handbook of Lubrication and Tribology, Volume III: Monitoring, Materials, Synthetic Lubricants, and Applications
3172:
5407:
2930:
3244:
5700:
670:, regardless of whether they are derived from an organic or inorganic acid. One example of an organic lactone is
3129:
is another example of a rearrangement resulting from an intramolecular nucleophilic acyl substitution reaction.
3109:
Crossed Claisen condensations, in which the enolate and nucleophile are different esters, are also possible. An
5124:
Isolation of triglyceride from nutmeg: G. D. Beal "Trimyristen" Organic Syntheses, Coll. Vol. 1, p.538 (1941).
3282:
3168:
1894:
Reagents are known that drive the dehydration of mixtures of alcohols and carboxylic acids. One example is the
6530:
3373:. Protecting a carboxylic acid is useful in peptide synthesis, to prevent self-reactions of the bifunctional
3033:
2754:
2270:
1001:
3105:
The Claisen condensation involves the reaction of an ester enolate and an ester to form a beta-keto ester.
2858:
1857:
2913:
under acidic and basic conditions. Under acidic conditions, the reaction is the reverse reaction of the
589:
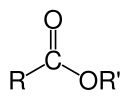
The chemical formulas of organic esters formed from carboxylic acids and alcohols usually take the form
6654:
6103:
5693:
1723:
1551:
965:
3040:
2808:
1596:
1136:
989:
5405:
Ignatyev, Igor; Charlie Van Doorslaer; Pascal G.N. Mertens; Koen Binnemans; Dirk. E. de Vos (2011).
2875:
Acid-catalyzed hydrolysis of esters is also an equilibrium process – essentially the reverse of the
2212:, to give esters. Anion availability can inhibit this reaction, which correspondingly benefits from
6140:
3153:
2770:
1903:
1568:
1469:
1445:, of which many hundreds are known, could be classified as esters of the corresponding acids (e.g.
933:
5022:
1739:
6613:
5675:
5521:
3322:
3271:
3232:
3157:
3110:
3029:
2821:
2748:
2213:
1907:
1895:
1067:
5617:
Wood, J. L.; Khatri, N. A.; Weinreb, S. M. (1979). "A direct conversion of esters to nitriles".
6513:
4575:
3278:
3228:
3164:
3114:
2914:
2876:
2274:
1819:
1204:
1047:
1031:
997:
20:
5404:
4943:
4881:
6620:
6508:
1663:
Esters are generally identified by gas chromatography, taking advantage of their volatility.
1446:
1319:
1039:
993:
961:
937:
702:
183:
6589:
6034:
5398:
5223:"Esterification of Carboxylic Acids with Dicyclohexylcarbodiimide/4-Dimethylaminopyridine:
4678:
4228:
3354:
3350:
3224:
3079:
2976:
2225:
1887:
1823:
1764:
1423:
1162:
1063:
1055:
953:
325:
1856:. Since esterification is highly reversible, the yield of the ester can be improved using
8:
5895:
5680:
5330:
Bienewald, Frank; Leibold, Edgar; Tužina, Pavel; Roscher, Günter (2019). "Vinyl Esters".
4626:
4089:
3567:
3329:
3036:. This method, which is largely obsolete, uses sodium in the presence of proton sources.
2846:
2794:
2700:
2638:
2529:
2236:
2126:
1795:
1465:
969:
925:
889:
671:
195:
5428:
4765:
4674:
4666:
3336:
3126:
3094:. The intermediate collapses, forcing out an alkoxide (R'O) and producing β-keto ester
3044:
2704:
2029:
1927:
1926:
Another method for the dehydration of mixtures of alcohols and carboxylic acids is the
1348:
1303:
1110:
1091:
1023:
771:
254:
5630:
5549:
5486:
1898:, which is a method of forming esters under mild conditions. The method is popular in
1603:
conformation is influenced by the nature of the substituents and solvent, if present.
6579:
6549:
6307:
5929:
5581:
5432:
5371:
5345:
5312:
5304:
5231:
5174:
5096:
5079:
4992:
4887:
4860:
4670:
4415:
4247:
4208:
3988:
3754:
3385:-butyl esters are particularly useful because, under strongly acidic conditions, the
2798:
2446:
2404:
2221:
2071:
1899:
1791:
1731:
1492:
1488:
1371:
1242:
1154:
1128:
686:. One of them are the esters of orthocarboxylic acids. Those esters have the formula
371:
278:
5653:
4833:
3702:
2152:
of carboxylic acids under conditions where acid-catalyzed reactions are infeasible:
1667:
for esters feature an intense sharp band in the range 1730–1750 cm assigned to
6284:
5778:
5716:
5657:
5648:
5626:
5573:
5545:
5482:
5455:
5420:
5380:
5337:
5294:
5263:
5203:
5166:
5107:
5071:
5062:
5042:
4837:
4828:
4454:
4391:
4164:
3918:
3891:
3815:
3723:
3532:
3366:
2983:
2870:
2499:
2205:
1868:
1523:
1401:
1340:
1183:
815:
796:
725:
179:
112:
59:
5341:
5170:
464:. For example, the ester hexyl octanoate, also known under the trivial name hexyl
6659:
6503:
6262:
6257:
6240:
6223:
6024:
5773:
4854:
4682:
4363:
4136:
4112:
4005:
3965:
3937:
3834:
3677:
3654:
3425:
3370:
3343:
3025:
2972:
2760:
2728:
2217:
2197:
Although rarely employed for esterifications, carboxylate salts (often generated
1822:, which involves treating a carboxylic acid with an alcohol in the presence of a
1664:
1643:
1630:
1419:
1344:
1295:
1107:
1087:
885:
881:
837:
763:
453:
187:
175:
163:
31:
5366:
5222:
1852:
is a typical catalyst for this reaction. Many other acids are also used such as
6574:
6569:
6445:
6440:
6435:
6228:
6195:
5979:
5961:
5951:
5125:
4530:
4435:
4313:
4293:
4183:
4070:
3627:
3592:
3578:
3513:
3471:
3444:
3402:
3063:
2922:
2899:
2724:
2491:
2412:
2408:
2149:
2091:
1794:
in which two reactants (typically an alcohol and an acid) form an ester as the
1727:
1561:
1397:
1315:
841:
721:
425:
3802:
3760:
2921:
acts as a nucleophile, while an alkoxide is the leaving group. This reaction,
1902:, where the substrates are sensitive to harsh conditions like high heat. DCC (
502:
6648:
6594:
6542:
6473:
6359:
6349:
6344:
6334:
6329:
6279:
6274:
6190:
6185:
6175:
6029:
5984:
5946:
5934:
5905:
5783:
5652:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
5384:
5308:
5267:
4832:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
4802:
4612:
4545:
4300:
4196:
4076:
3773:
3696:
3555:
3490:
2999:
2732:
2642:
2577:
2537:
2202:
2087:
1849:
1756:
1651:
1179:
1158:
1132:
1083:
929:
856:
792:
788:
610:
506:
465:
457:
435:
270:
266:
214:
159:
155:
128:
5661:
4841:
3561:
1682:
Esters are widespread in nature and are widely used in industry. In nature,
6525:
6412:
6407:
6384:
6135:
5974:
5900:
5837:
5832:
5810:
5766:
5751:
5741:
5585:
5459:
5316:
5083:
4702:
4339:
3981:
3450:
3377:. Methyl and ethyl esters are commonly available for many amino acids; the
3240:
3236:
2854:
2812:
2736:
2716:
2545:
2266:
2239:, which involves changing one ester into another one, is widely practiced:
2209:
2075:
1992:
1875:
1744:
1511:
1200:
1027:
601:
parts of the carboxylic acid and the alcohol, respectively, and R can be a
393:
258:
171:
2937:
The alkoxide group may also be displaced by stronger nucleophiles such as
1314:) (if one classifies thiocyanic acid as an inorganic compound), but forms
6584:
6537:
6498:
6379:
6267:
6252:
6247:
6235:
5800:
5795:
5761:
5756:
5746:
5724:
5299:
4791:
4740:
4696:
4662:
4654:
4631:
4616:
4608:
4570:
4265:
4143:
4125:
4021:
3846:
3608:
3571:
3390:
2857:) will drive the forward reaction towards completion, in accordance with
2777:
2650:
2585:
2495:
2134:
1367:
1299:
1246:
833:
767:
618:
606:
556:
534:
367:
321:
230:
167:
151:
5424:
5207:
3431:
6493:
6484:
6364:
6319:
6215:
6180:
6170:
6110:
6046:
5969:
5917:
4787:
4692:
4658:
4599:
4466:
4234:
4176:
4157:
3911:
3716:
3647:
3544:
3460:
3374:
3285: in this section. Unsourced material may be challenged and removed.
3059:
2968:
2910:
2903:
2786:
2646:
1772:
1715:
1691:
1175:
683:
242:
136:
124:
108:
5577:
5111:
5075:
5046:
4095:
3043:
is used to reduce esters to two primary alcohols. The related reagent
2906:
is an industrially important process, used in the production of soap.
2523:
1393:
Some inorganic acids that are unstable or elusive form stable esters.
6460:
6374:
6339:
6324:
6312:
6155:
6130:
5939:
4783:
4771:
4761:
4724:
4621:
4585:
4550:
4540:
4491:
4470:
4408:
4384:
4269:
4261:
4149:
3957:
3930:
3907:
3878:
3747:
3666:
3643:
3525:
3437:
3212:
2918:
2581:
2541:
1879:
1803:
1799:
1719:
1711:
1235:
614:
363:
317:
286:
246:
218:
206:), where R stands for any group (typically hydrogen or organyl) and R
94:
51:
5685:
5445:
4510:
4422:
4254:
4215:
3994:
3260:
3142:
2081:
6468:
6422:
6389:
6085:
5991:
5865:
5820:
5805:
4777:
4744:
4589:
4555:
4460:
4398:
4170:
4034:
4017:
3924:
3897:
3859:
3729:
3689:
3581:
3538:
3335:
Specific esters are functionalized with an α-hydroxyl group in the
3315:
3216:
2850:
2802:
2712:
2708:
2487:
2365:
2041:
2035:
1911:
1813:
1748:
1687:
1625:
1618:
1535:
1531:
1527:
1442:
1151:) (if one classifies trithiocarbonic acid as an inorganic compound)
602:
582:
574:
226:
116:
74:
67:
35:
4370:
4287:
4118:
4053:
4011:
3971:
3943:
3840:
3683:
3660:
3633:
3519:
3389:-butyl esters undergo elimination to give the carboxylic acid and
747:
174:), but also from acids that do not contain oxygen (e.g. esters of
119:; they are important in biology, being one of the main classes of
6430:
6354:
6205:
6200:
6165:
6150:
6145:
6115:
6098:
5922:
5849:
5815:
5280:
4797:
4755:
4749:
4706:
4688:
4603:
4593:
4565:
4560:
4441:
4376:
4332:
4326:
4189:
4105:
3821:
3735:
3598:
3477:
3464:
3220:
3075:
2938:
2834:
2815:
2764:
2140:
1883:
1807:
1694:. Esters are responsible for the aroma of many fruits, including
1604:
1331:
752:
736:
698:
667:
598:
510:
456:
are commonly named according to the more traditional, so-called "
282:
250:
238:
222:
147:
132:
82:
39:
4306:
3779:
3243:), and is subsequently decarboxylated. Another variation is the
1172:) (if one classifies chloroformic acid as an inorganic compound)
720:) is derived, in terms of its name (but not its synthesis) from
26:
6518:
6450:
6294:
6003:
5996:
5890:
5871:
5860:
5844:
5790:
4644:
4516:
4474:
4356:
4346:
4273:
4221:
4129:
3953:
3903:
3865:
3785:
3739:
3639:
3616:
3205:
2790:
2720:
2589:
1906:) is used to activate the carboxylic acid to further reaction.
1707:
1699:
1647:
1547:
1539:
140:
98:
90:
5159:
Riemenschneider, Wilhelm; Bolt, Hermann M. "Esters, Organic".
5010:"Chemistry of Enols and Enolates – Acidity of alpha-hydrogens"
3496:
3321:
Methyl esters are often susceptible to decarboxylation in the
2845:
Esters can be converted to other esters in a process known as
1542:
atom, which gives rise to 120° C–C–O and O–C–O angles. Unlike
6399:
6369:
6302:
6160:
6125:
6120:
6093:
6008:
5912:
5736:
5408:"Synthesis of glucose esters from cellulose in ionic liquids"
5329:
4710:
4639:
4635:
4535:
4428:
4404:
4352:
4240:
4153:
4101:
4063:
4059:
4040:
3884:
3827:
3808:
3789:
3766:
3743:
3708:
3620:
3612:
3604:
3585:
3574:
3548:
3506:
3502:
3069:
3055:
3048:
2946:
2942:
2130:
1919:
1853:
1722:
are produced industrially annually, important products being
1695:
1543:
1335:
1075:
985:
981:
977:
949:
234:
120:
102:
5059:
3342:
Esters with β-hydrogen atoms can be converted to alkenes in
2269:, e.g. in the production of fatty acid esters and alcohols.
1655:
volatile than carboxylic acids of similar molecular weight.
1464:) could be classified as an ester of aluminic acid which is
1125:) (if one classifies carbonic acid as an inorganic compound)
5827:
5252:
4942:(vol. 1) (Heidelberg, Baden (Germany): Karl Winter, 1848),
4478:
4447:
4380:
4082:
3977:
3949:
3483:
3456:
2540:
is produced industrially by the addition of acetic acid to
1703:
1637:
1071:
1059:
1051:
1043:
1035:
1017:
1005:
973:
957:
945:
941:
693:, where R stands for any group (organic or inorganic) and R
644:. Alternative presentations are common including BuOAc and
571:
294:
71:
63:
2986:
and organolithium compounds, add readily to the carbonyl.
4922:"Trimethyltin acetate | C5H12O2Sn | ChemSpider"
4497:
4200:
3712:
1683:
1009:
290:
262:
2909:
Esterification is a reversible reaction. Esters undergo
2853:
alcohol or removing the leaving group alcohol (e.g. via
2711:
in the presence of an anhydrous base to give an ester.
1864:
Using the alcohol in large excess (i.e., as a solvent).
5472:
5193:
2364:
A subset of transesterification is the alcoholysis of
5681:
Molecule of the month: Ethyl acetate and other esters
5562:
448:
IUPAC nomenclature of organic chemistry § Esters
5031:
Conformations of Esters, Thiol Esters, and Amides".
4786:, a class of natural esters that consist of a large
2824:
exchanges the fatty acid groups of different esters.
1334:
isothiocyanates are not classified as esters by the
3086:) will attack the carbonyl group of another ester (
2524:
Addition of carboxylic acids to alkenes and alkynes
1567:Many carboxylic acid esters have the potential for
5448:Angewandte Chemie International Edition in English
5406:
4959:welchen eine Säure mit einem Aether verbunden ist.
4717:) are replaced by the corresponding ester groups (
4602:, a polymeric ester made of small number of ester
3381:-butyl ester tends to be more expensive. However,
2040:Carboxylic acids are esterified by treatment with
1650:but less polar than alcohols. They participate in
241:, and are one of the largest classes of synthetic
5616:
4879:
3328:Phenyl esters react to hydroxyarylketones in the
3032:, esters were reduced on a large scale using the
2082:Alcoholysis of acyl chlorides and acid anhydrides
759:Esters can also be derived from inorganic acids.
6646:
5220:
2864:
2036:Esterification of carboxylic acids with epoxides
1814:Esterification of carboxylic acids with alcohols
1658:
621:(systematically ethanoic acid) would be written
5534:
5158:
2719:reacts with sodium benzyloxide (generated from
2133:are less sensitive because amines are stronger
2070:This reaction is employed in the production of
751:A phosphoric acid ester, where R stands for an
613:(systematically butyl ethanoate), derived from
5333:Ullmann's Encyclopedia of Industrial Chemistry
5162:Ullmann's Encyclopedia of Industrial Chemistry
4852:
2994:Compared to ketones and aldehydes, esters are
2141:Alkylation of carboxylic acids and their salts
1677:
1615:) conformation due to their cyclic structure.
5701:
2715:are aluminium alkoxides or sodium alkoxides.
1806:industry. Ester bonds are also found in many
4951:
4695:compound composed of two or more monocyclic
1441:In principle, a part of metal and metalloid
742:
429:
273:, are known for their explosive properties.
5637:
4991:4th Ed. J. Wiley and Sons, 1992: New York.
3171:. Unsourced material may be challenged and
3132:
3054:Direct reduction to give the corresponding
2125:The reactions are irreversible simplifying
1874:Removal of water by physical means such as
182:). An example of an ester formation is the
5708:
5694:
5154:
5152:
5150:
5148:
5146:
4953:b. Ester oder sauerstoffsäure Aetherarten.
4914:
3070:Claisen condensation and related reactions
2731:. The method is used in the production of
2415:are produced commercially by this method:
2273:is produced by the transesterification of
428:, probably as a contraction of the German
5298:
4873:
4846:
3396:
3301:Learn how and when to remove this message
3191:Learn how and when to remove this message
2494:, which is the main commercial source of
2224:. An additional iodide salt may, via the
1991:Carboxylic acids can be esterified using
1790:Esterification is the general name for a
5364:
5196:Journal of the American Chemical Society
5034:Journal of the American Chemical Society
3039:Especially for fine chemical syntheses,
2840:
1738:
1686:are, in general, triesters derived from
1638:Physical properties and characterization
1624:
1597:hyperconjugation and dipole minimization
1517:
746:
570:(left side of the picture, orange). The
555:) (right side of the picture, blue) and
501:
25:
5498:
5496:
5143:
4983:
4981:
4900:
4819:
4817:
3419:
3416:
3413:
3227:and its intramolecular equivalent, the
1607:with small rings are restricted to the
1595:) alternative, due to a combination of
424:was coined in 1848 by a German chemist
6647:
5336:. Weinheim: Wiley-VCH. pp. 1–16.
4880:E. Richard Booser (21 December 1993).
3231:. This conversion is exploited in the
2982:Sources of carbon nucleophiles, e.g.,
2532:, alkenes and alkynes insert into the
2368:. This reaction affords 2-ketoesters.
2231:
1747:found in a linseed oil, a triester of
5715:
5689:
5518:Virtual Textbook of Organic Chemistry
5511:
4764:(ascorbic acid), a lactone ester, an
3223:, which can further react, e.g., the
3219:. Deprotonation gives a nucleophilic
2898:Basic hydrolysis of esters, known as
1510:) could be classified as an ester of
1487:) could be classified as an ester of
509:, an ester derived from a residue of
441:
5493:
5358:
4978:
4814:
3360:
3349:Pairs of esters are coupled to give
3314:Esters can be directly converted to
3283:adding citations to reliable sources
3254:
3169:adding citations to reliable sources
3136:
2751:of α-haloketones in presence of base
682:An uncommon class of esters are the
66:(organic or inorganic) in which the
34:. R stands for any group (typically
4856:A worker's guide to solvent hazards
3239:reacts with an electrophile (e.g.,
3090:) to give tetrahedral intermediate
1910:(DMAP) is used as an acyl-transfer
297:); for example, according to them,
13:
5649:Compendium of Chemical Terminology
4829:Compendium of Chemical Terminology
4790:lactone ring to which one or more
3250:
3100:
2889:
2146:Trimethyloxonium tetrafluoroborate
1617:
1422:, which is unstable, forms stable
1400:, which is unstable, forms stable
1238:form two or more types of esters.
926:Pyrophosphoric (diphosphoric) acid
597:or RCOOR', where R and R' are the
14:
6671:
5669:
3113:Claisen condensation is called a
3058:is difficult as the intermediate
2996:relatively resistant to reduction
2694:
2584:-catalyzed reaction of ethylene,
1646:and alcohols are more polar than
81:) of that acid is replaced by an
5566:The Journal of Organic Chemistry
5514:"Carboxyl Derivative Reactivity"
4509:
4490:
4459:
4440:
4421:
4397:
4369:
4345:
4325:
4305:
4286:
4253:
4233:
4214:
4188:
4169:
4142:
4117:
4094:
4075:
4052:
4033:
4010:
3993:
3970:
3942:
3923:
3896:
3877:
3858:
3839:
3820:
3801:
3778:
3759:
3728:
3701:
3682:
3659:
3632:
3597:
3560:
3537:
3518:
3495:
3476:
3449:
3430:
3259:
3141:
3119:Baker–Venkataraman rearrangement
2929:
2742:
2398:
1918:
1599:effects. The preference for the
5610:
5601:
5592:
5556:
5528:
5505:
5466:
5439:
5323:
5274:
5246:
5214:
5187:
5130:
5118:
5090:
5053:
4940:Handbuch der organischen Chemie
4691:, a polymeric ester, a type of
3270:needs additional citations for
3066:with a variety of Lewis acids.
2925:, is the basis of soap making.
2783:Cellulolysis via esterification
2498:. The reaction is catalyzed by
1718:. Several billion kilograms of
1583:) conformation rather than the
585:is replaced by the butyl group.
410:
5365:Kamm, O.; Kamm, W. F. (1922).
5016:
5002:
4928:
4699:units linked by an ester group
3028:. Prior to the development of
2449:is one illustrative example.
1785:
1564:(C=O) of carboxylate esters).
1234:Inorganic acids that exist as
677:
392:is a trimethoxysilyl ester of
1:
5631:10.1016/S0040-4039(01)86746-0
5550:10.1016/S0040-4020(01)00420-3
5487:10.1016/S0040-4020(01)87078-2
5342:10.1002/14356007.a27_419.pub2
5221:B. Neises & W. Steglich.
5171:10.1002/14356007.a09_565.pub2
4808:
3051:reduces esters to aldehydes.
2865:Hydrolysis and saponification
1818:The classic synthesis is the
1659:Characterization and analysis
249:are important plastics, with
221:. They perform as high-grade
46:stands for any organyl group.
16:Compound derived from an acid
5607:Kürti and Czakó 2005, p. 30.
4967:b. Ester or oxy-acid ethers.
4768:for humans and other animals
4709:in which one or more of its
4705:, a type of ester that is a
4649:(R−C(=O)−O−)(R−C(=O)−O−)CH−R
3365:As a class, esters serve as
2989:
2828:
2271:Poly(ethylene terephthalate)
1571:, but they tend to adopt an
415:
101:), but not according to the
7:
5140:. 2003, Scribner, New York.
5100:Crystal Growth & Design
4752:, a cyclic carboxylic ester
4524:
3082:, an enolate of one ester (
1755:(center, black) derived of
1678:Applications and occurrence
1343:forms two types of esters:
909:methyl dihydrogen phosphate
123:and comprising the bulk of
10:
6676:
5393:, vol. 1, p. 104
4989:Advanced Organic Chemistry
4955:Ethers du troisième genre.
3400:
3047:is slow in this reaction.
2917:. Under basic conditions,
2868:
2536:bond of carboxylic acids.
2044:, giving β-hydroxyesters:
1724:polyethylene terephthalate
1633:, distances in picometers.
533:) (the butanol residue is
445:
301:(or trimethyltin acetate)
245:on the commercial market.
210:stands for organyl group.
146:Esters can be formed from
89:). Analogues derived from
18:
6603:
6562:
6482:
6459:
6421:
6398:
6293:
6214:
6084:
6061:
6017:
5960:
5883:
5858:
5723:
5676:An introduction to esters
5502:Wade 2010, pp. 1005–1009.
5241:, vol. 7, p. 93
4969:Ethers of the third type.
4859:. The Group. p. 48.
4780:, a type of lactone ester
4774:, a type of lactone ester
4758:, a type of lactone ester
3245:Fráter–Seebach alkylation
3235:, wherein the diester of
3041:lithium aluminium hydride
3034:Bouveault–Blanc reduction
2818:leading to methyl esters.
2757:of ketones with peroxides
2755:Baeyer–Villiger oxidation
1137:dimethyl trithiocarbonate
743:Esters of inorganic acids
666:Cyclic esters are called
605:in the case of esters of
277:by another atom from the
259:Esters of phosphoric acid
70:atom (H) of at least one
5598:Carey 2006, pp. 919–924.
5385:10.15227/orgsyn.002.0005
5268:10.15227/orgsyn.056.0059
3133:Other ester reactivities
2949:(ammonolysis reaction):
2941:or primary or secondary
2859:Le Chatelier's principle
2771:Nucleophilic abstraction
2580:can also be produced by
2214:phase transfer catalysts
1904:dicyclohexylcarbodiimide
1886:, in conjunction with a
1858:Le Chatelier's principle
1854:polymeric sulfonic acids
1569:conformational isomerism
1470:tetraethyl orthosilicate
934:tetraethyl pyrophosphate
299:trimethylstannyl acetate
6614:chemical classification
5662:10.1351/goldbook.D01604
5287:Journal of Oleo Science
5165:. Weinheim: Wiley-VCH.
4853:Cameron Wright (1986).
4842:10.1351/goldbook.E02219
3393:, simplifying work-up.
3323:Krapcho decarboxylation
3233:malonic ester synthesis
3121:, in which an aromatic
3030:catalytic hydrogenation
2773:of a metal–acyl complex
2749:Favorskii rearrangement
2641:is used to manufacture
1908:4-Dimethylaminopyridine
1896:Steglich esterification
1763:(bottom right, green),
1318:"esters" as well, e.g.
1277:-dimethyl thiosulfate (
1257:-dimethyl thiosulfate (
1133:trithiocarbonate esters
5460:10.1002/anie.197809392
5227:-Butyl ethyl fumarate"
4975:
4963:
4952:
4615:used in refrigeration
3397:List of ester odorants
3229:Dieckmann condensation
3115:Dieckmann condensation
3106:
3024:A typical catalyst is
2915:Fischer esterification
2895:
2877:Fischer esterification
2277:and ethylene glycol:
2275:dimethyl terephthalate
1820:Fischer esterification
1780:
1634:
1622:
1534:group C=O, which is a
756:
586:
430:
47:
21:Ester (disambiguation)
6621:chemical nomenclature
4965:
4949:
4908:"Acetoxytrimethyltin"
4663:orthocarboxylic acids
4634:, an ester that is a
4611:, an ester that is a
4250:(oil of wintergreen)
3104:
2971:through intermediate
2893:
2486:The carbonylation of
2411:catalysts. Esters of
2216:or such highly polar
1742:
1629:Metrical details for
1628:
1621:
1518:Structure and bonding
1447:aluminium triethoxide
1432:−O−C(=O)−O−C(=O)−O−CH
1320:methyl isothiocyanate
750:
703:triethyl orthoformate
505:
267:Esters of nitric acid
261:form the backbone of
225:for a broad array of
202:), forming an ester (
184:substitution reaction
29:
5300:10.5650/jos.ess13199
4886:. CRC. p. 237.
4679:orthophosphoric acid
4229:Methyl phenylacetate
3355:acyloin condensation
3279:improve this article
3225:Claisen condensation
3165:improve this section
3080:Claisen condensation
2977:Lossen rearrangement
2226:Finkelstein reaction
2086:Alcohols react with
1888:Dean-Stark apparatus
1767:alpha-linolenic acid
1642:Esters derived from
1522:Esters derived from
1424:dimethyl dicarbonate
1163:methyl chloroformate
1129:Trithiocarbonic acid
701:group. For example,
326:dibutyltin dilaurate
180:trithiocarbonic acid
19:For other uses, see
6077:not C, H or O)
5619:Tetrahedron Letters
5425:10.1515/hf.2011.161
5208:10.1021/ja01392a005
5138:On Food and Cooking
5012:. 13 February 2011.
4936:Handbuch der Chemie
4627:Transesterification
4139:(methyl butanoate)
4090:Methyl anthranilate
3568:nail polish remover
3420:Odor or occurrence
3330:Fries rearrangement
2962:R″ → RCONHR″ + R'OH
2847:transesterification
2841:Transesterification
2822:Interesterification
2797:in the presence of
2701:Tishchenko reaction
2639:Silicotungstic acid
2544:in the presence of
2530:hydroesterification
2407:in the presence of
2237:Transesterification
2232:Transesterification
1871:are also effective.
1466:aluminium hydroxide
1372:diethyl phosphonate
1245:forms two types of
890:triphenyl phosphate
581:) from acetic acid
6519:Hypervalent iodine
4766:essential nutrient
4675:orthotelluric acid
4667:orthocarbonic acid
4486:Propyl isobutyrate
4211:(methyl valerate)
3873:Geranyl pentanoate
3337:Chan rearrangement
3127:Chan rearrangement
3107:
3045:sodium borohydride
2896:
2705:disproportionation
2099:RCOCl + R'OH → RCO
2030:gas chromatography
1928:Mitsunobu reaction
1781:
1779:(top right, blue).
1635:
1623:
1368:phosphonate esters
1349:triethyl phosphite
1304:methyl thiocyanate
1111:ethylene carbonate
1092:dimethyl carbonate
1024:Triphosphoric acid
772:methyl perchlorate
757:
587:
468:, has the formula
442:IUPAC nomenclature
93:replaced by other
48:
6655:Functional groups
6642:
6641:
6580:Sulfenyl chloride
6558:
6557:
6057:
6056:
5876:(only C, H and O)
5717:Functional groups
5578:10.1021/jo070814z
5572:(15): 5920–5922.
5544:(25): 5353–5359.
5391:Collected Volumes
5372:Organic Syntheses
5367:"Benzyl benzoate"
5256:Organic Syntheses
5239:Collected Volumes
5232:Organic Syntheses
5112:10.1021/cg200547k
5076:10.1021/cr0104375
5047:10.1021/ja9723848
4893:978-1-4200-5045-5
4671:orthosilicic acid
4657:, an ester of an
4522:
4521:
4505:Terpinyl butyrate
4416:Pentyl pentanoate
4342:(pentyl acetate)
4248:Methyl salicylate
4209:Methyl pentanoate
3989:Isopropyl acetate
3755:Ethyl isovalerate
3671:waxy-green banana
3367:protecting groups
3361:Protecting groups
3311:
3310:
3303:
3201:
3200:
3193:
2984:Grignard reagents
2799:hydrochloric acid
2447:methyl propionate
2445:A preparation of
2405:carboalkoxylation
2206:alkylating agents
2072:vinyl ester resin
1900:peptide synthesis
1878:as a low-boiling
1792:chemical reaction
1771:(left, red), and
1732:cellulose acetate
1512:orthotitanic acid
1493:titanium ethoxide
1489:orthosilicic acid
1243:Thiosulfuric acid
1155:Chloroformic acid
1106:) and 5-membered
372:Phillips catalyst
364:dibutylstannylene
279:group 14 elements
143:and other foods.
113:fatty acid esters
6667:
6609:
6514:Trifluoromethoxy
6082:
6081:
6078:
5881:
5880:
5877:
5730:
5710:
5703:
5696:
5687:
5686:
5664:
5641:
5635:
5634:
5614:
5608:
5605:
5599:
5596:
5590:
5589:
5560:
5554:
5553:
5532:
5526:
5525:
5520:. Archived from
5509:
5503:
5500:
5491:
5490:
5481:(4–5): 895–905.
5470:
5464:
5463:
5443:
5437:
5436:
5410:
5402:
5396:
5394:
5387:
5362:
5356:
5355:
5327:
5321:
5320:
5302:
5278:
5272:
5271:
5250:
5244:
5242:
5235:
5218:
5212:
5211:
5202:(5): 1267–1271.
5191:
5185:
5184:
5156:
5141:
5134:
5128:
5122:
5116:
5115:
5106:(9): 3964–3978.
5094:
5088:
5087:
5070:(7): 2475–2932.
5063:Chemical Reviews
5057:
5051:
5050:
5041:(9): 2108–2112.
5020:
5014:
5013:
5006:
5000:
4985:
4976:
4961:
4934:Leopold Gmelin,
4932:
4926:
4925:
4918:
4912:
4911:
4904:
4898:
4897:
4877:
4871:
4870:
4850:
4844:
4821:
4738:
4737:−)(R−C(=O)−O−)CH
4720:
4716:
4661:(e.g. esters of
4650:
4513:
4494:
4463:
4455:Propyl hexanoate
4444:
4425:
4418:(amyl valerate)
4401:
4394:(amyl caproate)
4392:Pentyl hexanoate
4373:
4366:(amyl butyrate)
4349:
4329:
4309:
4290:
4257:
4237:
4218:
4192:
4173:
4165:Methyl cinnamate
4146:
4121:
4098:
4079:
4056:
4037:
4029:Linalyl butyrate
4014:
3997:
3974:
3946:
3927:
3919:Isobutyl formate
3900:
3892:Isobutyl acetate
3881:
3862:
3854:Geranyl butyrate
3843:
3824:
3816:Ethyl pentanoate
3805:
3782:
3763:
3732:
3724:Ethyl heptanoate
3705:
3686:
3663:
3636:
3601:
3564:
3541:
3533:Butyl propionate
3522:
3499:
3480:
3453:
3434:
3411:
3410:
3371:carboxylic acids
3351:α-hydroxyketones
3306:
3299:
3295:
3292:
3286:
3263:
3255:
3196:
3189:
3185:
3182:
3176:
3145:
3137:
3020:
2973:hydroxamic acids
2963:
2933:
2871:Ester hydrolysis
2809:Anodic oxidation
2690:
2634:
2573:
2535:
2519:
2500:sodium methoxide
2482:
2441:
2403:Alkenes undergo
2394:
2360:
2261:
2218:aprotic solvents
2193:
2148:can be used for
2121:
2104:
2094:to give esters:
2066:
2024:
1987:
1922:
1869:molecular sieves
1844:
1796:reaction product
1777:
1769:
1761:
1753:
1644:carboxylic acids
1524:carboxylic acids
1509:
1486:
1463:
1436:
1415:
1402:dimethyl sulfite
1388:
1365:
1345:phosphite esters
1341:Phosphorous acid
1329:
1313:
1291:
1268:
1229:
1196:
1184:trimethyl borate
1171:
1150:
1124:
1105:
1088:carbonate esters
921:
906:
886:phosphate esters
877:
854:
829:
816:methyl bisulfate
813:
797:dimethyl sulfate
784:
734:
726:orthoformic acid
719:
696:
692:
662:
643:
596:
580:
569:
554:
532:
498:
454:carboxylic acids
433:
406:
391:
361:
318:trimethylstannyl
315:
253:linked by ester
209:
205:
201:
193:
150:(e.g. esters of
88:
80:
62:derived from an
60:functional group
45:
6675:
6674:
6670:
6669:
6668:
6666:
6665:
6664:
6645:
6644:
6643:
6638:
6607:
6599:
6554:
6509:Trichloromethyl
6504:Trifluoromethyl
6478:
6455:
6417:
6394:
6289:
6258:Phosphine oxide
6210:
6076:
6074:
6073:
6071:
6069:
6067:
6065:
6063:
6053:
6013:
5956:
5875:
5874:
5869:
5864:
5854:
5728:
5727:
5719:
5714:
5672:
5667:
5642:
5638:
5615:
5611:
5606:
5602:
5597:
5593:
5561:
5557:
5533:
5529:
5510:
5506:
5501:
5494:
5471:
5467:
5454:(12): 939–940.
5444:
5440:
5403:
5399:
5389:
5363:
5359:
5352:
5328:
5324:
5279:
5275:
5251:
5247:
5237:
5219:
5215:
5192:
5188:
5181:
5157:
5144:
5136:McGee, Harold.
5135:
5131:
5123:
5119:
5095:
5091:
5058:
5054:
5021:
5017:
5008:
5007:
5003:
4986:
4979:
4971:
4970:
4968:
4957:
4956:
4954:
4947:
4933:
4929:
4920:
4919:
4915:
4906:
4905:
4901:
4894:
4878:
4874:
4867:
4851:
4847:
4822:
4815:
4811:
4794:may be attached
4739:), an ester of
4736:
4733:−)(R−C(=O)−O−CH
4732:
4728:
4718:
4714:
4683:orthoboric acid
4648:
4527:
4364:Pentyl butyrate
4282:Nonyl caprylate
4276:ointments (UK)
4137:Methyl butyrate
4113:Methyl benzoate
4048:Linalyl formate
4006:Linalyl acetate
3966:Isoamyl formate
3938:Isoamyl acetate
3835:Geranyl acetate
3797:Ethyl nonanoate
3678:Ethyl cinnamate
3655:Ethyl hexanoate
3426:Allyl hexanoate
3405:
3399:
3363:
3344:ester pyrolysis
3307:
3296:
3290:
3287:
3276:
3264:
3253:
3251:Other reactions
3197:
3186:
3180:
3177:
3162:
3146:
3135:
3072:
3026:copper chromite
3018:
3014:
3010:
3006:
2992:
2961:
2957:
2953:
2885:
2873:
2867:
2843:
2831:
2780:in aqueous acid
2767:with an alcohol
2761:Pinner reaction
2745:
2729:benzyl benzoate
2697:
2689:
2685:
2681:
2677:
2673:
2669:
2665:
2661:
2657:
2632:
2628:
2624:
2620:
2616:
2612:
2608:
2604:
2600:
2596:
2572:
2568:
2564:
2560:
2556:
2552:
2533:
2526:
2518:
2514:
2510:
2506:
2481:
2477:
2473:
2469:
2465:
2461:
2457:
2453:
2439:
2435:
2431:
2428:+ ROH + CO → CH
2427:
2423:
2419:
2401:
2392:
2388:
2384:
2380:
2376:
2372:
2358:
2350:
2344:
2340:
2336:
2332:
2328:
2324:
2320:
2316:
2312:
2304:
2300:
2296:
2292:
2288:
2281:
2259:
2255:
2251:
2247:
2243:
2234:
2192:
2188:
2184:
2180:
2176:
2172:
2168:
2164:
2160:
2156:
2143:
2119:
2115:
2111:
2107:
2102:
2098:
2092:acid anhydrides
2084:
2064:
2060:
2056:
2052:
2048:
2038:
2023:
2019:
2015:
2011:
2007:
2003:
1999:
1986:
1982:
1978:
1974:
1970:
1966:
1962:
1958:
1954:
1950:
1946:
1942:
1938:
1934:
1842:
1838:
1834:
1830:
1816:
1788:
1773:
1765:
1757:
1749:
1743:Representative
1728:acrylate esters
1680:
1673:
1661:
1640:
1631:methyl benzoate
1558:
1520:
1508:
1504:
1500:
1496:
1485:
1481:
1477:
1473:
1462:
1458:
1454:
1450:
1435:
1431:
1427:
1420:Dicarbonic acid
1413:
1409:
1405:
1387:
1383:
1379:
1375:
1364:
1360:
1356:
1352:
1327:
1323:
1311:
1307:
1296:Thiocyanic acid
1290:
1286:
1282:
1278:
1266:
1262:
1258:
1228:
1224:
1220:
1216:
1212:
1209:-butyl chromate
1195:
1191:
1187:
1170:
1166:
1148:
1144:
1140:
1122:
1118:
1114:
1103:
1099:
1095:
920:
916:
912:
905:
901:
897:
893:
882:Phosphoric acid
876:
872:
868:
864:
860:
853:
849:
845:
827:
823:
819:
812:
808:
804:
800:
783:
779:
775:
764:Perchloric acid
745:
733:
729:
718:
714:
710:
706:
694:
691:
687:
680:
672:γ-valerolactone
661:
657:
653:
649:
645:
642:
638:
634:
630:
626:
622:
609:. For example,
594:
590:
578:
567:
563:
559:
553:
549:
545:
541:
537:
530:
526:
522:
518:
514:
497:
493:
489:
485:
481:
477:
473:
469:
450:
444:
418:
413:
405:
401:
397:
390:
386:
382:
378:
374:
360:
356:
352:
348:
344:
340:
336:
332:
328:
314:
310:
306:
302:
207:
203:
199:
191:
188:carboxylic acid
176:thiocyanic acid
164:phosphoric acid
86:
78:
43:
32:carboxylic acid
24:
17:
12:
11:
5:
6673:
6663:
6662:
6657:
6640:
6639:
6637:
6636:
6635:
6634:
6629:
6617:
6610:
6604:
6601:
6600:
6598:
6597:
6595:Sulfinylamines
6592:
6587:
6582:
6577:
6575:Phosphoramides
6572:
6570:Isothiocyanate
6566:
6564:
6560:
6559:
6556:
6555:
6553:
6552:
6547:
6546:
6545:
6535:
6534:
6533:
6523:
6522:
6521:
6516:
6511:
6506:
6501:
6490:
6488:
6480:
6479:
6477:
6476:
6471:
6465:
6463:
6457:
6456:
6454:
6453:
6448:
6446:Selenenic acid
6443:
6441:Seleninic acid
6438:
6436:Selenonic acid
6433:
6427:
6425:
6419:
6418:
6416:
6415:
6410:
6404:
6402:
6396:
6395:
6393:
6392:
6387:
6382:
6377:
6372:
6367:
6362:
6357:
6352:
6347:
6342:
6337:
6332:
6327:
6322:
6317:
6316:
6315:
6305:
6299:
6297:
6291:
6290:
6288:
6287:
6282:
6277:
6272:
6271:
6270:
6260:
6255:
6250:
6245:
6244:
6243:
6233:
6232:
6231:
6229:Phosphodiester
6220:
6218:
6212:
6211:
6209:
6208:
6203:
6198:
6193:
6188:
6183:
6178:
6173:
6168:
6163:
6158:
6153:
6148:
6143:
6138:
6133:
6128:
6123:
6118:
6113:
6108:
6107:
6106:
6101:
6090:
6088:
6079:
6075:(one element,
6059:
6058:
6055:
6054:
6052:
6051:
6050:
6049:
6039:
6038:
6037:
6032:
6021:
6019:
6015:
6014:
6012:
6011:
6006:
6001:
6000:
5999:
5989:
5988:
5987:
5982:
5977:
5966:
5964:
5958:
5957:
5955:
5954:
5952:Methylenedioxy
5949:
5944:
5943:
5942:
5937:
5927:
5926:
5925:
5920:
5910:
5909:
5908:
5898:
5893:
5887:
5885:
5878:
5856:
5855:
5853:
5852:
5847:
5842:
5841:
5840:
5835:
5825:
5824:
5823:
5818:
5813:
5808:
5803:
5798:
5788:
5787:
5786:
5781:
5771:
5770:
5769:
5764:
5759:
5754:
5749:
5744:
5733:
5731:
5729:(only C and H)
5721:
5720:
5713:
5712:
5705:
5698:
5690:
5684:
5683:
5678:
5671:
5670:External links
5668:
5666:
5665:
5636:
5609:
5600:
5591:
5555:
5527:
5524:on 2016-05-16.
5504:
5492:
5465:
5438:
5419:(4): 417–425.
5397:
5357:
5350:
5322:
5293:(5): 539–544.
5273:
5245:
5213:
5186:
5180:978-3527306732
5179:
5142:
5129:
5117:
5089:
5052:
5015:
5001:
4977:
4948:Original text:
4927:
4913:
4899:
4892:
4872:
4865:
4845:
4812:
4810:
4807:
4806:
4805:
4800:
4795:
4781:
4775:
4769:
4759:
4753:
4747:
4734:
4730:
4722:
4700:
4686:
4652:
4642:
4629:
4624:
4619:
4606:
4597:
4583:
4581:Tellurocyanate
4578:
4573:
4568:
4563:
4558:
4553:
4548:
4543:
4538:
4533:
4531:List of esters
4526:
4523:
4520:
4519:
4514:
4507:
4501:
4500:
4495:
4488:
4482:
4481:
4464:
4457:
4451:
4450:
4445:
4438:
4436:Propyl acetate
4432:
4431:
4426:
4419:
4412:
4411:
4402:
4395:
4388:
4387:
4374:
4367:
4360:
4359:
4350:
4343:
4336:
4335:
4330:
4323:
4321:Octyl butyrate
4317:
4316:
4310:
4303:
4297:
4296:
4291:
4284:
4278:
4277:
4258:
4251:
4244:
4243:
4238:
4231:
4225:
4224:
4219:
4212:
4205:
4204:
4193:
4186:
4184:Methyl formate
4180:
4179:
4174:
4167:
4161:
4160:
4147:
4140:
4133:
4132:
4122:
4115:
4109:
4108:
4099:
4092:
4086:
4085:
4080:
4073:
4071:Methyl acetate
4067:
4066:
4057:
4050:
4044:
4043:
4038:
4031:
4025:
4024:
4015:
4008:
4002:
4001:
3998:
3991:
3985:
3984:
3975:
3968:
3962:
3961:
3956:(flavoring in
3947:
3940:
3934:
3933:
3928:
3921:
3915:
3914:
3901:
3894:
3888:
3887:
3882:
3875:
3869:
3868:
3863:
3856:
3850:
3849:
3844:
3837:
3831:
3830:
3825:
3818:
3812:
3811:
3806:
3799:
3793:
3792:
3783:
3776:
3770:
3769:
3764:
3757:
3751:
3750:
3733:
3726:
3720:
3719:
3706:
3699:
3693:
3692:
3687:
3680:
3674:
3673:
3664:
3657:
3651:
3650:
3637:
3630:
3628:Ethyl butyrate
3624:
3623:
3602:
3595:
3593:Ethyl benzoate
3589:
3588:
3579:model airplane
3565:
3558:
3552:
3551:
3542:
3535:
3529:
3528:
3523:
3516:
3514:Butyl butyrate
3510:
3509:
3500:
3493:
3487:
3486:
3481:
3474:
3472:Bornyl acetate
3468:
3467:
3454:
3447:
3445:Benzyl acetate
3441:
3440:
3435:
3428:
3422:
3421:
3418:
3415:
3403:List of esters
3398:
3395:
3362:
3359:
3358:
3357:
3347:
3340:
3333:
3326:
3319:
3309:
3308:
3291:September 2024
3267:
3265:
3258:
3252:
3249:
3199:
3198:
3181:September 2024
3149:
3147:
3140:
3134:
3131:
3111:intramolecular
3071:
3068:
3064:triethylsilane
3022:
3021:
3016:
3012:
3008:
3000:fatty alcohols
2991:
2988:
2965:
2964:
2959:
2955:
2935:
2934:
2923:saponification
2900:saponification
2883:
2869:Main article:
2866:
2863:
2842:
2839:
2830:
2827:
2826:
2825:
2819:
2806:
2784:
2781:
2776:Hydrolysis of
2774:
2768:
2758:
2752:
2744:
2741:
2727:) to generate
2725:benzyl alcohol
2696:
2695:From aldehydes
2693:
2692:
2691:
2687:
2683:
2679:
2675:
2671:
2667:
2663:
2659:
2636:
2635:
2630:
2626:
2622:
2618:
2614:
2610:
2606:
2602:
2598:
2575:
2574:
2570:
2566:
2562:
2558:
2554:
2525:
2522:
2521:
2520:
2516:
2512:
2508:
2492:methyl formate
2484:
2483:
2479:
2475:
2471:
2467:
2463:
2459:
2455:
2443:
2442:
2437:
2433:
2429:
2425:
2421:
2413:propanoic acid
2409:metal carbonyl
2400:
2397:
2396:
2395:
2390:
2386:
2382:
2378:
2374:
2362:
2361:
2356:
2346:
2342:
2338:
2334:
2330:
2326:
2322:
2318:
2314:
2310:
2302:
2298:
2294:
2290:
2286:
2263:
2262:
2257:
2253:
2249:
2245:
2233:
2230:
2195:
2194:
2190:
2186:
2182:
2178:
2174:
2170:
2166:
2162:
2158:
2150:esterification
2142:
2139:
2123:
2122:
2117:
2113:
2112:O + R'OH → RCO
2109:
2105:
2100:
2088:acyl chlorides
2083:
2080:
2068:
2067:
2062:
2058:
2054:
2050:
2037:
2034:
2026:
2025:
2021:
2017:
2013:
2009:
2005:
2001:
1989:
1988:
1984:
1980:
1976:
1972:
1968:
1964:
1960:
1956:
1952:
1948:
1944:
1940:
1939:H + R'OH + P(C
1936:
1924:
1923:
1892:
1891:
1872:
1865:
1846:
1845:
1840:
1836:
1835:H + R'OH ⇌ RCO
1832:
1815:
1812:
1787:
1784:
1783:
1782:
1679:
1676:
1671:
1660:
1657:
1652:hydrogen bonds
1639:
1636:
1562:carbonyl group
1556:
1519:
1516:
1506:
1502:
1498:
1483:
1479:
1475:
1460:
1456:
1452:
1439:
1438:
1433:
1429:
1417:
1411:
1407:
1398:Sulfurous acid
1391:
1390:
1385:
1381:
1377:
1362:
1358:
1354:
1338:
1325:
1316:isothiocyanate
1309:
1293:
1288:
1284:
1280:
1264:
1260:
1232:
1231:
1226:
1222:
1218:
1214:
1198:
1193:
1189:
1173:
1168:
1152:
1146:
1142:
1126:
1120:
1116:
1101:
1097:
1081:
1080:
1079:
1021:
918:
914:
903:
899:
895:
879:
874:
870:
866:
862:
851:
847:
842:methyl nitrate
838:nitrate esters
831:
825:
821:
810:
806:
802:
793:sulfate esters
786:
781:
777:
770:esters, e.g.,
744:
741:
731:
722:esterification
716:
712:
708:
689:
679:
676:
659:
655:
651:
647:
640:
636:
632:
628:
624:
592:
565:
561:
551:
547:
543:
539:
528:
524:
520:
516:
495:
491:
487:
483:
479:
475:
471:
446:Main article:
443:
440:
426:Leopold Gmelin
417:
414:
412:
409:
403:
399:
388:
384:
380:
376:
358:
354:
350:
346:
342:
338:
334:
330:
312:
308:
304:
215:essential oils
129:vegetable oils
30:An ester of a
15:
9:
6:
4:
3:
2:
6672:
6661:
6658:
6656:
6653:
6652:
6650:
6633:
6630:
6628:
6625:
6624:
6623:
6622:
6618:
6616:
6615:
6611:
6606:
6605:
6602:
6596:
6593:
6591:
6588:
6586:
6583:
6581:
6578:
6576:
6573:
6571:
6568:
6567:
6565:
6561:
6551:
6548:
6544:
6541:
6540:
6539:
6536:
6532:
6529:
6528:
6527:
6524:
6520:
6517:
6515:
6512:
6510:
6507:
6505:
6502:
6500:
6497:
6496:
6495:
6492:
6491:
6489:
6487:
6486:
6481:
6475:
6474:Telluroketone
6472:
6470:
6467:
6466:
6464:
6462:
6458:
6452:
6449:
6447:
6444:
6442:
6439:
6437:
6434:
6432:
6429:
6428:
6426:
6424:
6420:
6414:
6411:
6409:
6406:
6405:
6403:
6401:
6397:
6391:
6388:
6386:
6383:
6381:
6378:
6376:
6373:
6371:
6368:
6366:
6363:
6361:
6360:Sulfonic acid
6358:
6356:
6353:
6351:
6350:Sulfinic acid
6348:
6346:
6345:Thiosulfonate
6343:
6341:
6338:
6336:
6335:Thiosulfinate
6333:
6331:
6330:Sulfenic acid
6328:
6326:
6323:
6321:
6318:
6314:
6311:
6310:
6309:
6306:
6304:
6301:
6300:
6298:
6296:
6292:
6286:
6285:Phosphaallene
6283:
6281:
6280:Phosphaalkyne
6278:
6276:
6275:Phosphaalkene
6273:
6269:
6266:
6265:
6264:
6261:
6259:
6256:
6254:
6251:
6249:
6246:
6242:
6239:
6238:
6237:
6234:
6230:
6227:
6226:
6225:
6222:
6221:
6219:
6217:
6213:
6207:
6204:
6202:
6199:
6197:
6194:
6192:
6189:
6187:
6184:
6182:
6179:
6177:
6174:
6172:
6169:
6167:
6164:
6162:
6159:
6157:
6154:
6152:
6149:
6147:
6144:
6142:
6139:
6137:
6134:
6132:
6129:
6127:
6124:
6122:
6119:
6117:
6114:
6112:
6109:
6105:
6102:
6100:
6097:
6096:
6095:
6092:
6091:
6089:
6087:
6083:
6080:
6060:
6048:
6045:
6044:
6043:
6040:
6036:
6033:
6031:
6028:
6027:
6026:
6023:
6022:
6020:
6016:
6010:
6007:
6005:
6002:
5998:
5995:
5994:
5993:
5990:
5986:
5983:
5981:
5978:
5976:
5973:
5972:
5971:
5968:
5967:
5965:
5963:
5959:
5953:
5950:
5948:
5947:Ethylenedioxy
5945:
5941:
5938:
5936:
5933:
5932:
5931:
5928:
5924:
5921:
5919:
5916:
5915:
5914:
5911:
5907:
5904:
5903:
5902:
5899:
5897:
5894:
5892:
5889:
5888:
5886:
5882:
5879:
5873:
5867:
5862:
5857:
5851:
5848:
5846:
5843:
5839:
5836:
5834:
5831:
5830:
5829:
5826:
5822:
5819:
5817:
5814:
5812:
5809:
5807:
5804:
5802:
5799:
5797:
5794:
5793:
5792:
5789:
5785:
5782:
5780:
5777:
5776:
5775:
5772:
5768:
5765:
5763:
5760:
5758:
5755:
5753:
5750:
5748:
5745:
5743:
5740:
5739:
5738:
5735:
5734:
5732:
5726:
5722:
5718:
5711:
5706:
5704:
5699:
5697:
5692:
5691:
5688:
5682:
5679:
5677:
5674:
5673:
5663:
5659:
5655:
5654:depsipeptides
5651:
5650:
5645:
5640:
5632:
5628:
5624:
5620:
5613:
5604:
5595:
5587:
5583:
5579:
5575:
5571:
5567:
5559:
5551:
5547:
5543:
5539:
5531:
5523:
5519:
5515:
5508:
5499:
5497:
5488:
5484:
5480:
5476:
5469:
5461:
5457:
5453:
5449:
5442:
5434:
5430:
5426:
5422:
5418:
5414:
5413:Holzforschung
5409:
5401:
5392:
5386:
5382:
5378:
5374:
5373:
5368:
5361:
5353:
5351:9783527303854
5347:
5343:
5339:
5335:
5334:
5326:
5318:
5314:
5310:
5306:
5301:
5296:
5292:
5288:
5284:
5277:
5269:
5265:
5261:
5257:
5249:
5240:
5234:
5233:
5228:
5226:
5217:
5209:
5205:
5201:
5197:
5190:
5182:
5176:
5172:
5168:
5164:
5163:
5155:
5153:
5151:
5149:
5147:
5139:
5133:
5127:
5121:
5113:
5109:
5105:
5101:
5093:
5085:
5081:
5077:
5073:
5069:
5065:
5064:
5056:
5048:
5044:
5040:
5036:
5035:
5030:
5026:
5019:
5011:
5005:
4998:
4997:0-471-60180-2
4994:
4990:
4984:
4982:
4974:
4962:
4960:
4945:
4941:
4937:
4931:
4923:
4917:
4909:
4903:
4895:
4889:
4885:
4884:
4876:
4868:
4866:9780969054542
4862:
4858:
4857:
4849:
4843:
4839:
4835:
4831:
4830:
4825:
4820:
4818:
4813:
4804:
4803:Chloroformate
4801:
4799:
4796:
4793:
4789:
4785:
4782:
4779:
4776:
4773:
4770:
4767:
4763:
4760:
4757:
4754:
4751:
4748:
4746:
4742:
4729:(R−C(=O)−O−CH
4726:
4723:
4712:
4708:
4704:
4701:
4698:
4694:
4690:
4687:
4684:
4680:
4676:
4672:
4668:
4664:
4660:
4656:
4653:
4646:
4643:
4641:
4637:
4633:
4630:
4628:
4625:
4623:
4620:
4618:
4614:
4613:synthetic oil
4610:
4607:
4605:
4601:
4598:
4595:
4591:
4587:
4584:
4582:
4579:
4577:
4576:Selenocyanate
4574:
4572:
4569:
4567:
4564:
4562:
4559:
4557:
4554:
4552:
4549:
4547:
4546:Carboximidate
4544:
4542:
4539:
4537:
4534:
4532:
4529:
4528:
4518:
4515:
4512:
4508:
4506:
4503:
4502:
4499:
4496:
4493:
4489:
4487:
4484:
4483:
4480:
4476:
4472:
4468:
4465:
4462:
4458:
4456:
4453:
4452:
4449:
4446:
4443:
4439:
4437:
4434:
4433:
4430:
4427:
4424:
4420:
4417:
4414:
4413:
4410:
4406:
4403:
4400:
4396:
4393:
4390:
4389:
4386:
4382:
4378:
4375:
4372:
4368:
4365:
4362:
4361:
4358:
4354:
4351:
4348:
4344:
4341:
4338:
4337:
4334:
4331:
4328:
4324:
4322:
4319:
4318:
4315:
4311:
4308:
4304:
4302:
4301:Octyl acetate
4299:
4298:
4295:
4292:
4289:
4285:
4283:
4280:
4279:
4275:
4271:
4267:
4263:
4259:
4256:
4252:
4249:
4246:
4245:
4242:
4239:
4236:
4232:
4230:
4227:
4226:
4223:
4220:
4217:
4213:
4210:
4207:
4206:
4202:
4198:
4194:
4191:
4187:
4185:
4182:
4181:
4178:
4175:
4172:
4168:
4166:
4163:
4162:
4159:
4155:
4151:
4148:
4145:
4141:
4138:
4135:
4134:
4131:
4127:
4123:
4120:
4116:
4114:
4111:
4110:
4107:
4103:
4100:
4097:
4093:
4091:
4088:
4087:
4084:
4081:
4078:
4074:
4072:
4069:
4068:
4065:
4061:
4058:
4055:
4051:
4049:
4046:
4045:
4042:
4039:
4036:
4032:
4030:
4027:
4026:
4023:
4019:
4016:
4013:
4009:
4007:
4004:
4003:
3999:
3996:
3992:
3990:
3987:
3986:
3983:
3979:
3976:
3973:
3969:
3967:
3964:
3963:
3959:
3955:
3951:
3948:
3945:
3941:
3939:
3936:
3935:
3932:
3929:
3926:
3922:
3920:
3917:
3916:
3913:
3909:
3905:
3902:
3899:
3895:
3893:
3890:
3889:
3886:
3883:
3880:
3876:
3874:
3871:
3870:
3867:
3864:
3861:
3857:
3855:
3852:
3851:
3848:
3845:
3842:
3838:
3836:
3833:
3832:
3829:
3826:
3823:
3819:
3817:
3814:
3813:
3810:
3807:
3804:
3800:
3798:
3795:
3794:
3791:
3787:
3784:
3781:
3777:
3775:
3774:Ethyl lactate
3772:
3771:
3768:
3765:
3762:
3758:
3756:
3753:
3752:
3749:
3745:
3741:
3737:
3734:
3731:
3727:
3725:
3722:
3721:
3718:
3714:
3710:
3707:
3704:
3700:
3698:
3697:Ethyl formate
3695:
3694:
3691:
3688:
3685:
3681:
3679:
3676:
3675:
3672:
3668:
3665:
3662:
3658:
3656:
3653:
3652:
3649:
3645:
3641:
3638:
3635:
3631:
3629:
3626:
3625:
3622:
3618:
3615:, medicinal,
3614:
3610:
3606:
3603:
3600:
3596:
3594:
3591:
3590:
3587:
3583:
3580:
3576:
3573:
3569:
3566:
3563:
3559:
3557:
3556:Ethyl acetate
3554:
3553:
3550:
3546:
3543:
3540:
3536:
3534:
3531:
3530:
3527:
3524:
3521:
3517:
3515:
3512:
3511:
3508:
3504:
3501:
3498:
3494:
3492:
3491:Butyl acetate
3489:
3488:
3485:
3482:
3479:
3475:
3473:
3470:
3469:
3466:
3462:
3458:
3455:
3452:
3448:
3446:
3443:
3442:
3439:
3436:
3433:
3429:
3427:
3424:
3423:
3412:
3409:
3404:
3394:
3392:
3388:
3384:
3380:
3376:
3372:
3368:
3356:
3352:
3348:
3345:
3341:
3338:
3334:
3331:
3327:
3324:
3320:
3317:
3313:
3312:
3305:
3302:
3294:
3284:
3280:
3274:
3273:
3268:This section
3266:
3262:
3257:
3256:
3248:
3246:
3242:
3238:
3234:
3230:
3226:
3222:
3218:
3214:
3209:
3207:
3195:
3192:
3184:
3174:
3170:
3166:
3160:
3159:
3155:
3150:This section
3148:
3144:
3139:
3138:
3130:
3128:
3124:
3120:
3116:
3112:
3103:
3099:
3097:
3093:
3089:
3085:
3081:
3077:
3067:
3065:
3061:
3057:
3052:
3050:
3046:
3042:
3037:
3035:
3031:
3027:
3005:
3004:
3003:
3001:
2997:
2987:
2985:
2980:
2978:
2974:
2970:
2952:
2951:
2950:
2948:
2944:
2940:
2932:
2928:
2927:
2926:
2924:
2920:
2916:
2912:
2907:
2905:
2901:
2892:
2888:
2882:
2878:
2872:
2862:
2860:
2856:
2852:
2848:
2838:
2836:
2823:
2820:
2817:
2814:
2810:
2807:
2804:
2800:
2796:
2792:
2788:
2785:
2782:
2779:
2775:
2772:
2769:
2766:
2762:
2759:
2756:
2753:
2750:
2747:
2746:
2743:Other methods
2740:
2738:
2734:
2733:ethyl acetate
2730:
2726:
2722:
2718:
2714:
2710:
2706:
2702:
2656:
2655:
2654:
2653:by ethylene:
2652:
2648:
2644:
2643:ethyl acetate
2640:
2595:
2594:
2593:
2591:
2587:
2583:
2579:
2578:Vinyl acetate
2551:
2550:
2549:
2547:
2543:
2539:
2538:Vinyl acetate
2531:
2511:OH + CO → HCO
2505:
2504:
2503:
2501:
2497:
2493:
2489:
2452:
2451:
2450:
2448:
2418:
2417:
2416:
2414:
2410:
2406:
2399:Carbonylation
2371:
2370:
2369:
2367:
2354:
2349:
2308:
2284:
2280:
2279:
2278:
2276:
2272:
2268:
2267:triglycerides
2242:
2241:
2240:
2238:
2229:
2227:
2223:
2219:
2215:
2211:
2210:alkyl halides
2207:
2204:
2203:electrophilic
2201:) react with
2200:
2155:
2154:
2153:
2151:
2147:
2138:
2136:
2132:
2128:
2106:
2097:
2096:
2095:
2093:
2089:
2079:
2077:
2073:
2047:
2046:
2045:
2043:
2033:
2031:
1998:
1997:
1996:
1994:
1933:
1932:
1931:
1929:
1921:
1917:
1916:
1915:
1913:
1909:
1905:
1901:
1897:
1889:
1885:
1881:
1877:
1873:
1870:
1866:
1863:
1862:
1861:
1859:
1855:
1851:
1850:Sulfuric acid
1829:
1828:
1827:
1825:
1821:
1811:
1809:
1805:
1801:
1797:
1793:
1778:
1776:
1770:
1768:
1762:
1760:
1759:linoleic acid
1754:
1752:
1746:
1741:
1737:
1736:
1735:
1733:
1729:
1725:
1721:
1717:
1713:
1709:
1705:
1701:
1697:
1693:
1689:
1685:
1675:
1670:
1666:
1656:
1653:
1649:
1645:
1632:
1627:
1620:
1616:
1614:
1611:-trans (i.e.
1610:
1606:
1602:
1598:
1594:
1590:
1586:
1582:
1578:
1574:
1570:
1565:
1563:
1559:
1555:
1549:
1545:
1541:
1537:
1533:
1529:
1525:
1515:
1513:
1494:
1490:
1471:
1467:
1448:
1444:
1425:
1421:
1418:
1403:
1399:
1396:
1395:
1394:
1376:H−P(=O)(−O−CH
1373:
1369:
1350:
1346:
1342:
1339:
1337:
1333:
1321:
1317:
1305:
1302:esters, e.g.
1301:
1297:
1294:
1276:
1272:
1256:
1252:
1249:esters, e.g.
1248:
1244:
1241:
1240:
1239:
1237:
1210:
1208:
1202:
1199:
1185:
1181:
1180:borate esters
1177:
1174:
1167:Cl−C(=O)−O−CH
1164:
1161:esters, e.g.
1160:
1159:chloroformate
1156:
1153:
1138:
1134:
1130:
1127:
1112:
1109:
1093:
1089:
1085:
1084:Carbonic acid
1082:
1077:
1073:
1069:
1065:
1061:
1057:
1053:
1049:
1045:
1041:
1037:
1033:
1030:esters, e.g.
1029:
1025:
1022:
1019:
1015:
1011:
1007:
1003:
999:
995:
991:
987:
983:
979:
975:
971:
967:
963:
959:
955:
951:
947:
943:
939:
935:
932:esters, e.g.
931:
930:pyrophosphate
927:
924:
923:
910:
891:
887:
883:
880:
858:
857:nitroglycerin
843:
839:
835:
832:
817:
798:
794:
790:
789:Sulfuric acid
787:
773:
769:
765:
762:
761:
760:
754:
749:
740:
738:
727:
723:
704:
700:
685:
675:
673:
669:
664:
620:
616:
612:
611:butyl acetate
608:
604:
600:
584:
576:
573:
558:
536:
512:
508:
507:Butyl acetate
504:
500:
467:
463:
459:
458:trivial names
455:
449:
439:
437:
432:
427:
423:
408:
395:
373:
369:
365:
327:
323:
319:
300:
296:
292:
288:
284:
280:
274:
272:
271:nitroglycerin
268:
264:
260:
256:
252:
248:
244:
240:
236:
232:
228:
224:
220:
216:
211:
197:
189:
185:
181:
177:
173:
169:
165:
161:
160:sulfuric acid
157:
156:carbonic acid
153:
149:
144:
142:
138:
134:
130:
126:
122:
118:
114:
110:
106:
104:
100:
96:
92:
84:
76:
73:
69:
65:
61:
57:
53:
41:
37:
33:
28:
22:
6619:
6612:
6526:Vinyl halide
6483:
6413:Borinic acid
6408:Boronic acid
6385:Thioxanthate
6041:
5725:Hydrocarbons
5647:
5639:
5625:(51): 4907.
5622:
5618:
5612:
5603:
5594:
5569:
5565:
5558:
5541:
5537:
5530:
5522:the original
5517:
5507:
5478:
5474:
5468:
5451:
5447:
5441:
5416:
5412:
5400:
5390:
5376:
5370:
5360:
5331:
5325:
5290:
5286:
5276:
5259:
5255:
5248:
5238:
5230:
5224:
5216:
5199:
5195:
5189:
5160:
5137:
5132:
5120:
5103:
5099:
5092:
5067:
5061:
5055:
5038:
5032:
5028:
5024:
5018:
5004:
4988:
4966:
4964:Translation:
4950:
4939:
4935:
4930:
4916:
4902:
4882:
4875:
4855:
4848:
4827:
4792:deoxy sugars
4703:Depsipeptide
4693:polyphenolic
4340:Amyl acetate
3982:blackcurrant
3406:
3386:
3382:
3378:
3364:
3297:
3288:
3277:Please help
3272:verification
3269:
3241:alkyl halide
3237:malonic acid
3210:
3202:
3187:
3178:
3163:Please help
3151:
3122:
3108:
3095:
3091:
3087:
3083:
3073:
3053:
3038:
3023:
2993:
2981:
2966:
2936:
2908:
2897:
2880:
2874:
2855:distillation
2844:
2832:
2801:and various
2737:acetaldehyde
2717:Benzaldehyde
2698:
2637:
2576:
2546:zinc acetate
2527:
2485:
2444:
2402:
2363:
2352:
2347:
2306:
2282:
2264:
2235:
2198:
2196:
2144:
2135:nucleophiles
2124:
2085:
2076:acrylic acid
2069:
2039:
2027:
1993:diazomethane
1990:
1925:
1893:
1876:distillation
1847:
1817:
1789:
1774:
1766:
1758:
1750:
1745:triglyceride
1716:strawberries
1681:
1668:
1662:
1641:
1612:
1608:
1600:
1592:
1588:
1584:
1580:
1576:
1572:
1566:
1553:
1521:
1440:
1392:
1330:), although
1274:
1270:
1254:
1250:
1233:
1206:
1201:Chromic acid
1028:triphosphate
758:
681:
665:
588:
461:
451:
436:acetic ether
421:
419:
411:Nomenclature
394:chromic acid
275:
231:plasticizers
212:
204:R−C(=O)−O−R'
172:xanthic acid
145:
107:
55:
49:
6590:Thiocyanate
6585:Sulfonamide
6550:Perchlorate
6538:Acyl halide
6499:Fluoroethyl
6380:Thionoester
6268:Phosphonium
6253:Phosphinate
6248:Phosphonous
6236:Phosphonate
5935:Hydroperoxy
5757:Cyclopropyl
5538:Tetrahedron
5512:W. Reusch.
5475:Tetrahedron
4788:macrocyclic
4741:fatty acids
4655:Ortho ester
4632:Ether lipid
4617:compressors
4609:Polyolester
4571:Thiocyanate
4266:wintergreen
4126:ylang ylang
3609:wintergreen
3414:Ester name
3391:isobutylene
3375:amino acids
2969:isocyanates
2904:fatty acids
2778:orthoesters
2651:acetic acid
2586:acetic acid
2548:catalysts:
2496:formic acid
1824:dehydrating
1786:Preparation
1692:fatty acids
1300:thiocyanate
1247:thiosulfate
834:Nitric acid
768:perchlorate
697:stands for
684:orthoesters
678:Orthoesters
619:acetic acid
607:formic acid
557:acetic acid
535:butyl group
368:lauric acid
322:acetic acid
265:molecules.
168:nitric acid
152:acetic acid
125:animal fats
6649:Categories
6494:Haloalkane
6365:Thioketone
6320:Persulfide
6216:Phosphorus
6181:Isocyanate
6171:Isonitrile
6072:or oxygen
6070:hydrogen,
6066:not being
6047:Orthoester
5940:Dioxiranes
5918:Enol ether
5806:1-Propenyl
4987:March, J.
4938:, vol. 4:
4809:References
4715:−C(=O)−NH−
4659:ortho acid
4600:Oligoester
4467:blackberry
4195:pleasant,
4177:strawberry
4158:strawberry
3958:Pear drops
3912:strawberry
3717:strawberry
3648:strawberry
3545:pear drops
3461:strawberry
3417:Structure
3401:See also:
3060:hemiacetal
2911:hydrolysis
2787:Ozonolysis
2647:alkylation
2553:HC≡CH + CH
2381:+ ROH → CH
2208:, such as
1775:oleic acid
1720:polyesters
1712:pineapples
1665:IR spectra
1530:contain a
1176:Boric acid
431:Essigäther
370:, and the
269:, such as
247:Polyesters
243:lubricants
219:pheromones
192:R−C(=O)−OH
186:between a
137:vegetables
109:Glycerides
95:chalcogens
6627:inorganic
6461:Tellurium
6375:Thioester
6340:Sulfoxide
6325:Disulfide
6313:Sulfonium
6263:Phosphine
6241:Phosphite
6224:Phosphate
6156:Carbamate
6131:Hydrazone
6064:element,
6062:Only one
6035:Anhydride
5774:Methylene
5433:101737591
5309:1345-8957
4784:Macrolide
4772:Phthalide
4762:Vitamin C
4725:Glyceride
4719:−C(=O)−O−
4622:Thioester
4594:polymeric
4586:Polyester
4551:Carbamate
4541:Thioamide
4471:pineapple
4409:pineapple
4385:pineapple
4270:Germolene
4262:root beer
4150:pineapple
3931:raspberry
3908:raspberry
3748:raspberry
3667:pineapple
3644:pineapple
3526:pineapple
3438:pineapple
3217:alkoxides
3213:aldehydes
3152:does not
3078:. In the
3019:OH + R'OH
2990:Reduction
2919:hydroxide
2829:Reactions
2713:Catalysts
2703:involves
2582:palladium
2542:acetylene
2462:+ CO + CH
2321:→ [(C
2053:H + RCHCH
1963:R' + OP(C
1880:azeotrope
1800:fragrance
1538:group at
1443:alkoxides
1287:−S−)S(=O)
1267:S(=O)(=S)
1236:tautomers
913:O=P(−O−CH
780:−O−Cl(=O)
466:caprylate
420:The word
416:Etymology
366:ester of
320:ester of
194:) and an
52:chemistry
6608:See also
6543:Chloride
6469:Tellurol
6423:Selenium
6390:Xanthate
6104:Ammonium
6086:Nitrogen
6068:carbon,
6025:Carboxyl
5992:Aldehyde
5980:Acryloyl
5962:carbonyl
5866:hydrogen
5821:Cumulene
5586:17602594
5317:24770480
5084:12848578
4944:page 182
4778:Coumarin
4745:glycerol
4713:groups (
4697:aromatic
4604:monomers
4592:made of
4590:plastics
4556:Xanthate
4525:See also
4203:, sweet
4197:ethereal
4124:fruity,
4018:lavender
3847:geranium
3690:cinnamon
3316:nitriles
3076:enolates
3011:R' + 2 H
2945:to give
2851:reactant
2803:alcohols
2793:using a
2765:nitriles
2709:aldehyde
2488:methanol
2366:diketene
2252:OH → RCO
2116:R' + RCO
2103:R' + HCl
2042:epoxides
1912:catalyst
1808:polymers
1751:glycerol
1688:glycerol
1605:Lactones
1536:divalent
1532:carbonyl
1528:alcohols
894:O=P(−O−C
861:CH(−O−NO
824:−O−S(=O)
795:, e.g.,
668:lactones
603:hydrogen
583:molecule
575:hydrogen
379:(OSi(OCH
307:COOSn(CH
255:moieties
251:monomers
239:lacquers
227:plastics
223:solvents
148:oxoacids
133:Lactones
117:glycerol
85:group (R
75:hydroxyl
68:hydrogen
36:hydrogen
6632:organic
6431:Selenol
6355:Sulfone
6308:Sulfide
6206:NONOate
6201:Nitroso
6191:Nitrite
6186:Nitrate
6176:Cyanate
6166:Nitrile
6151:Amidine
6146:Imidate
6116:Nitrene
6111:Hydrazo
6099:Enamine
6030:Acetoxy
6018:carboxy
5985:Benzoyl
5923:Epoxide
5906:Methoxy
5896:Alcohol
5850:Carbene
5784:Methine
4798:Formate
4756:Lactide
4750:Lactone
4707:peptide
4689:Depside
4638:and an
4566:Cyanate
4561:Amidine
4377:apricot
4333:parsnip
4312:fruity-
4260:Modern
4222:flowery
4106:jasmine
4000:fruity
3736:apricot
3465:jasmine
3353:in the
3221:enolate
3211:As for
3206:adducts
3173:removed
3158:sources
2975:in the
2958:R' + NH
2939:ammonia
2835:ammonia
2816:ketones
2795:work up
2791:alkenes
2645:by the
2490:yields
2466:OH → CH
2248:R' + CH
2199:in situ
2189:O + HBF
2161:H + (CH
2127:work-up
2065:CH(OH)R
2057:O → RCO
1884:toluene
1826:agent:
1708:bananas
1700:durians
1370:, e.g.
1366:), and
1353:P(−O−CH
1347:, e.g.
1332:organyl
1283:−O−)(CH
1188:B(−O−CH
1182:, e.g.
1135:, e.g.
1090:, e.g.
888:, e.g.
840:, e.g.
753:organyl
737:ethanol
735:) with
699:organyl
688:RC(OR′)
615:butanol
599:organyl
511:butanol
196:alcohol
83:organyl
77:group (
42:) and R
40:organyl
6660:Esters
6531:Iodide
6451:Selone
6295:Sulfur
6004:Ketone
5997:Ketene
5975:Acetyl
5930:Peroxy
5901:Alkoxy
5891:Acetal
5872:oxygen
5861:carbon
5845:Alkyne
5838:Benzyl
5833:Phenyl
5816:Allene
5811:Crotyl
5791:Alkene
5779:Bridge
5767:Pentyl
5752:Propyl
5742:Methyl
5584:
5431:
5348:
5315:
5307:
5262:: 59.
5177:
5082:
4995:
4890:
4863:
4834:esters
4685:, ...)
4645:Acylal
4517:cherry
4475:cheese
4357:banana
4314:orange
4294:orange
4274:Ralgex
4130:feijoa
3954:banana
3904:cherry
3866:cherry
3786:butter
3740:cherry
3640:banana
3617:cherry
3613:fruity
2947:amides
2943:amines
2813:methyl
2721:sodium
2707:of an
2674:H → CH
2617:→ 2 CH
2605:+ 2 CH
2590:oxygen
2588:, and
2561:H → CH
2385:C(O)CH
2260:+ R'OH
2173:→ RCO
2131:amides
2004:H + CH
1839:R' + H
1804:flavor
1730:, and
1714:, and
1696:apples
1648:ethers
1550:. The
1548:amides
1544:amides
1497:Ti(OCH
1491:, and
1474:Si(OCH
1451:Al(OCH
1328:−N=C=S
1312:−S−C≡N
1298:forms
1269:) and
1225:Cr(=O)
1203:forms
1178:forms
1157:forms
1131:forms
1108:cyclic
1086:forms
1026:forms
928:forms
917:)(−OH)
907:) and
884:forms
855:) and
836:forms
814:) and
791:forms
766:forms
755:group.
730:HC(OH)
707:HC(OCH
650:COO(CH
577:atom (
572:acidic
345:Sn((CH
324:, and
237:, and
235:resins
141:celery
121:lipids
99:amides
91:oxygen
72:acidic
6563:Other
6400:Boron
6370:Thial
6303:Thiol
6196:Nitro
6161:Imide
6141:Amide
6126:Oxime
6121:Imine
6094:Amine
6042:Ester
6009:Ynone
5913:Ether
5884:R-O-R
5859:Only
5801:Allyl
5796:Vinyl
5762:Butyl
5747:Ethyl
5737:Alkyl
5644:IUPAC
5429:S2CID
5379:: 5.
4824:IUPAC
4711:amide
4640:ether
4636:lipid
4596:ester
4536:Amide
4429:apple
4405:apple
4353:apple
4241:honey
4154:apple
4102:grape
4064:peach
4060:apple
4041:peach
3885:apple
3828:apple
3809:grape
3790:cream
3767:apple
3744:grape
3709:lemon
3621:grape
3605:sweet
3586:pears
3575:paint
3572:model
3549:apple
3507:honey
3503:apple
3123:ortho
3056:ether
3049:DIBAH
3015:→ RCH
2735:from
2629:+ 2 H
2625:CH=CH
2613:H + O
2569:CH=CH
2181:+ (CH
2108:(RCO)
2074:from
2012:→ RCO
1959:→ RCO
1882:with
1704:pears
1589:trans
1336:IUPAC
1221:C−O−)
1076:AThTP
986:DMAPP
982:HMBPP
978:MEcPP
950:cADPR
869:−O−NO
865:)(−CH
850:−O−NO
809:S(=O)
462:-oate
422:ester
362:is a
316:is a
200:R'−OH
139:like
103:IUPAC
58:is a
56:ester
54:, an
6485:Halo
5970:Acyl
5870:and
5828:Aryl
5582:PMID
5346:ISBN
5313:PMID
5305:ISSN
5225:tert
5175:ISBN
5126:Link
5080:PMID
5027:and
4993:ISBN
4888:ISBN
4861:ISBN
4743:and
4479:wine
4448:pear
4381:pear
4272:and
4083:glue
4022:sage
3978:plum
3950:pear
3582:glue
3484:pine
3457:pear
3369:for
3156:any
3154:cite
2723:and
2699:The
2666:+ CH
2662:C=CH
2601:C=CH
2458:C=CH
2424:C=CH
2329:)(CO
2317:(OH)
2293:)(CO
2090:and
1802:and
1690:and
1684:fats
1591:(or
1579:(or
1526:and
1410:−O−)
1263:−O−)
1213:((CH
1207:tert
1145:−S−)
1119:−O−)
1115:(−CH
1100:−O−)
1072:ThTP
1060:dTTP
1052:dGTP
1044:dCTP
1036:dATP
1018:NADP
1006:ThDP
1002:GGPP
974:dTDP
966:dGDP
958:dCDP
946:ADPR
942:dADP
805:−O−)
617:and
341:COO)
217:and
178:and
127:and
111:are
64:acid
6136:Azo
5658:doi
5656:".
5627:doi
5574:doi
5546:doi
5483:doi
5456:doi
5421:doi
5381:doi
5338:doi
5295:doi
5264:doi
5204:doi
5167:doi
5108:doi
5072:doi
5068:103
5043:doi
5039:120
4838:doi
4836:".
4498:rum
4201:rum
3713:rum
3281:by
3167:by
3007:RCO
2954:RCO
2811:of
2789:of
2763:of
2649:of
2597:2 H
2534:O−H
2528:In
2377:CO)
2373:(CH
2351:+ 2
2305:+ 2
2244:RCO
2222:DMF
2220:as
2169:OBF
2157:RCO
2049:RCO
2020:+ N
2000:RCO
1975:+ R
1951:+ R
1935:RCO
1831:RCO
1672:C=O
1577:cis
1514:).
1414:S=O
1406:(CH
1279:(CH
1259:(CH
1205:di-
1149:C=S
1141:(CH
1123:C=O
1104:C=O
1096:(CH
1068:XTP
1064:ITP
1056:UTP
1048:GTP
1040:CTP
1032:ATP
1014:NAD
1010:FAD
998:FPP
994:GPP
990:IPP
970:UDP
962:GDP
954:CDP
938:ADP
828:−OH
801:(CH
724:of
631:(CH
591:RCO
538:−CH
486:(CH
474:(CH
438:".
434:, "
407:).
402:CrO
375:CrO
333:(CH
329:(CH
263:DNA
115:of
79:−OH
50:In
38:or
6651::
5868:,
5863:,
5646:,
5623:20
5621:.
5580:.
5570:72
5568:.
5542:57
5540:.
5516:.
5495:^
5479:47
5477:.
5452:17
5450:.
5427:.
5417:66
5415:.
5411:.
5388:;
5375:.
5369:.
5344:.
5311:.
5303:.
5291:63
5289:.
5285:.
5260:56
5258:.
5236:;
5229:.
5200:50
5198:.
5173:.
5145:^
5104:11
5102:.
5078:.
5066:.
5037:.
4980:^
4826:,
4816:^
4681:,
4677:,
4673:,
4669:,
4665:,
4588:,
4477:,
4473:,
4469:,
4407:,
4383:,
4379:,
4355:,
4268:,
4264:,
4199:,
4156:,
4152:,
4128:,
4104:,
4062:,
4020:,
3980:,
3960:)
3952:,
3910:,
3906:,
3788:,
3746:,
3742:,
3738:,
3715:,
3711:,
3669:,
3646:,
3642:,
3619:,
3611:,
3607:,
3584:,
3577:,
3570:,
3547:,
3505:,
3463:,
3459:,
3247:.
3208:.
3098:.
3002:.
2979:.
2861:.
2739:.
2686:CH
2682:CH
2678:CO
2670:CO
2621:CO
2609:CO
2592::
2565:CO
2557:CO
2515:CH
2507:CH
2502::
2478:CH
2474:CO
2470:CH
2436:CO
2432:CH
2389:CO
2359:OH
2355:CH
2345:)]
2337:(C
2297:CH
2285:(C
2256:CH
2177:CH
2078:.
2061:CH
2016:CH
1995::
1930::
1914:.
1860::
1810:.
1734:.
1726:,
1710:,
1706:,
1702:,
1698:,
1501:CH
1478:CH
1468:,
1455:CH
1428:CH
1380:CH
1357:CH
1324:CH
1308:CH
1074:,
1070:,
1066:,
1062:,
1058:,
1054:,
1050:,
1046:,
1042:,
1038:,
1034:,
1016:,
1012:,
1008:,
1004:,
1000:,
996:,
992:,
988:,
984:,
980:,
976:,
972:,
968:,
964:,
960:,
956:,
952:,
948:,
944:,
940:,
936:,
922:)
846:CH
820:CH
776:CH
739:.
711:CH
674:.
663:.
658:CH
646:CH
639:CH
627:CO
623:CH
595:R'
579:−H
564:CO
560:CH
550:CH
546:CH
542:CH
531:OH
527:CH
523:CH
519:CH
515:CH
499:.
494:CH
482:CO
470:CH
353:CH
339:10
303:CH
295:Pb
293:,
291:Sn
289:,
287:Ge
285:,
283:Si
257:.
233:,
229:,
170:,
166:,
162:,
158:,
154:,
131:.
105:.
5709:e
5702:t
5695:v
5660::
5633:.
5629::
5588:.
5576::
5552:.
5548::
5489:.
5485::
5462:.
5458::
5435:.
5423::
5395:.
5383::
5377:2
5354:.
5340::
5319:.
5297::
5270:.
5266::
5243:.
5210:.
5206::
5183:.
5169::
5114:.
5110::
5086:.
5074::
5049:.
5045::
5029:Z
5025:E
4999:.
4946:.
4924:.
4910:.
4896:.
4869:.
4840::
4735:2
4731:2
4727:(
4721:)
4651:)
4647:(
3387:t
3383:t
3379:t
3346:.
3339:.
3332:.
3325:.
3318:.
3304:)
3298:(
3293:)
3289:(
3275:.
3194:)
3188:(
3183:)
3179:(
3175:.
3161:.
3096:4
3092:3
3088:2
3084:1
3017:2
3013:2
3009:2
2960:2
2956:2
2884:a
2881:K
2805:.
2688:3
2684:2
2680:2
2676:3
2672:2
2668:3
2664:2
2660:2
2658:H
2633:O
2631:2
2627:2
2623:2
2619:3
2615:2
2611:2
2607:3
2603:2
2599:2
2571:2
2567:2
2563:3
2559:2
2555:3
2517:3
2513:2
2509:3
2480:3
2476:2
2472:2
2468:3
2464:3
2460:2
2456:2
2454:H
2440:R
2438:2
2434:2
2430:3
2426:2
2422:2
2420:H
2393:R
2391:2
2387:2
2383:3
2379:2
2375:2
2357:3
2353:n
2348:n
2343:4
2341:H
2339:2
2335:2
2333:)
2331:2
2327:4
2325:H
2323:6
2319:2
2315:4
2313:H
2311:2
2309:C
2307:n
2303:2
2301:)
2299:3
2295:2
2291:4
2289:H
2287:6
2283:n
2258:3
2254:2
2250:3
2246:2
2191:4
2187:2
2185:)
2183:3
2179:3
2175:2
2171:4
2167:3
2165:)
2163:3
2159:2
2120:H
2118:2
2114:2
2110:2
2101:2
2063:2
2059:2
2055:2
2051:2
2022:2
2018:3
2014:2
2010:2
2008:N
2006:2
2002:2
1985:2
1983:H
1981:2
1979:N
1977:2
1973:3
1971:)
1969:5
1967:H
1965:6
1961:2
1957:2
1955:N
1953:2
1949:3
1947:)
1945:5
1943:H
1941:6
1937:2
1890:.
1843:O
1841:2
1837:2
1833:2
1669:ν
1613:E
1609:s
1601:Z
1593:E
1587:-
1585:S
1581:Z
1575:-
1573:S
1557:a
1554:K
1552:p
1540:C
1507:4
1505:)
1503:3
1499:2
1495:(
1484:4
1482:)
1480:3
1476:2
1472:(
1461:3
1459:)
1457:3
1453:2
1449:(
1437:)
1434:3
1430:3
1426:(
1416:)
1412:2
1408:3
1404:(
1389:)
1386:2
1384:)
1382:3
1378:2
1374:(
1363:3
1361:)
1359:3
1355:2
1351:(
1326:3
1322:(
1310:3
1306:(
1292:)
1289:2
1285:3
1281:3
1275:S
1273:,
1271:O
1265:2
1261:3
1255:O
1253:,
1251:O
1230:)
1227:2
1223:2
1219:3
1217:)
1215:3
1211:(
1197:)
1194:3
1192:)
1190:3
1186:(
1169:3
1165:(
1147:2
1143:3
1139:(
1121:2
1117:2
1113:(
1102:2
1098:3
1094:(
1078:.
1020:.
919:2
915:3
911:(
904:3
902:)
900:5
898:H
896:6
892:(
878:)
875:2
873:)
871:2
867:2
863:2
859:(
852:2
848:3
844:(
830:)
826:2
822:3
818:(
811:2
807:2
803:3
799:(
785:)
782:3
778:3
774:(
732:3
728:(
717:3
715:)
713:3
709:2
705:(
695:′
690:3
660:3
656:3
654:)
652:2
648:3
641:3
637:3
635:)
633:2
629:2
625:3
593:2
568:H
566:2
562:3
552:3
548:2
544:2
540:2
529:2
525:2
521:2
517:3
513:(
496:3
492:5
490:)
488:2
484:2
480:6
478:)
476:2
472:3
404:4
400:2
398:H
396:(
389:2
387:)
385:3
383:)
381:3
377:2
359:2
357:)
355:3
351:3
349:)
347:2
343:2
337:)
335:2
331:3
313:3
311:)
309:3
305:3
281:(
208:′
198:(
190:(
87:′
44:′
23:.
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.