61:
109:
286:
252:
Montavon, M.; Lindlar, H.; Marbet, R.; Rüegg, R.; Ryser, G.; Saucy, G.; Zeller, P.; Isler, O. (1957). "Synthesen in der
Carotinoid-Reihe. 11. Mitteilung. α,β-Ungesättigte Carbonylverbindungen aus Allylhalogeniden mittels Nitroparaffinen".
198:
Hass, Henry B.; Bender, Myron L. (1949). "The
Reaction of Benzyl Halides with the Sodium Salt of 2-Nitropropane. A General Synthesis of Substituted Benzaldehydes".
327:
182:
149:
354:
359:
320:
313:
301:
32:
60:
349:
8:
96:
84:
91:
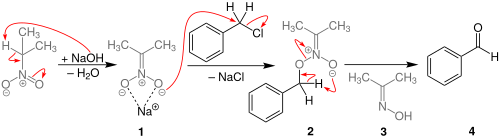
atom, it is instead an oxygen of the nitro itself that attacks the benzylic carbon. The
108:
293:
178:
145:
20:
262:
234:
207:
174:
170:
142:
Organic
Syntheses Based on Name Reactions: A Practical Guide to 750 Transformations
225:
Bersohn, Malcolm (1961). "C versus O Alkylation in the Case of a Stable Cation".
52:
115:
Although originally developed for benzyl compounds, the reaction also works for
297:
140:
Hassner, Alfred; Namboothiri, Irishi (2012). "Hass–Bender carbonyl synthesis".
44:
343:
266:
48:
40:
67:
The reaction process begins with the deprotonation of 2-nitropropane at the
76:
68:
88:
238:
211:
72:
285:
87:, where the deprotonated carbon of the nitroalkyl group is the
36:
120:
116:
100:
251:
139:
167:Comprehensive Organic Name Reactions and Reagents
341:
165:Wang, Zerong (2009). "Hass–Bender oxidation".
321:
83:to displace the benzyl halide. Unlike in the
328:
314:
197:
99:to produce a benzaldehyde, with dimethyl
224:
342:
280:
164:
119:halides, giving the respective α,β-
95:-benzyl structure then undergoes a
13:
75:. This compound then initiates an
14:
371:
55:, who first reported it in 1949.
284:
107:
59:
51:is named for Henry B. Hass and
245:
218:
191:
175:10.1002/9780470638859.conrr297
158:
144:. Elsevier. pp. 203–204.
133:
29:Hass–Bender carbonyl synthesis
1:
169:. Wiley. pp. 1335–1337.
126:
300:. You can help Knowledge by
7:
355:Organic oxidation reactions
10:
376:
279:
33:organic oxidation reaction
43:using the sodium salt of
267:10.1002/hlca.19570400516
360:Organic chemistry stubs
47:as the oxidant. This
25:Hass–Bender oxidation
239:10.1021/ja01470a022
212:10.1021/ja01173a066
97:pericyclic reaction
85:nitroaldol reaction
309:
308:
294:organic chemistry
184:978-0-471-70450-8
151:978-0-08-096630-4
27:(also called the
21:organic chemistry
16:Chemical reaction
367:
330:
323:
316:
288:
281:
271:
270:
261:(5): 1250–1256.
255:Helv. Chim. Acta
249:
243:
242:
233:(9): 2136–2138.
227:J. Am. Chem. Soc
222:
216:
215:
206:(5): 1767–1769.
200:J. Am. Chem. Soc
195:
189:
188:
162:
156:
155:
137:
121:enones and enals
111:
103:as a byproduct.
63:
375:
374:
370:
369:
368:
366:
365:
364:
340:
339:
337:
335:
334:
277:
275:
274:
250:
246:
223:
219:
196:
192:
185:
163:
159:
152:
138:
134:
129:
80:
53:Myron L. Bender
17:
12:
11:
5:
373:
363:
362:
357:
352:
350:Name reactions
333:
332:
325:
318:
310:
307:
306:
289:
273:
272:
244:
217:
190:
183:
157:
150:
131:
130:
128:
125:
113:
112:
78:
65:
64:
45:2-nitropropane
35:that converts
15:
9:
6:
4:
3:
2:
372:
361:
358:
356:
353:
351:
348:
347:
345:
338:
331:
326:
324:
319:
317:
312:
311:
305:
303:
299:
296:article is a
295:
290:
287:
283:
282:
278:
268:
264:
260:
256:
248:
240:
236:
232:
228:
221:
213:
209:
205:
201:
194:
186:
180:
176:
172:
168:
161:
153:
147:
143:
136:
132:
124:
122:
118:
110:
106:
105:
104:
102:
98:
94:
90:
86:
82:
74:
70:
62:
58:
57:
56:
54:
50:
49:name reaction
46:
42:
41:benzaldehydes
39:halides into
38:
34:
30:
26:
22:
336:
302:expanding it
291:
276:
258:
254:
247:
230:
226:
220:
203:
199:
193:
166:
160:
141:
135:
114:
92:
89:nucleophilic
66:
28:
24:
18:
344:Categories
127:References
81:2 reaction
71:to form a
73:nitronate
69:α carbon
31:) is an
181:
148:
37:benzyl
23:, the
292:This
117:allyl
101:oxime
298:stub
179:ISBN
146:ISBN
263:doi
235:doi
208:doi
171:doi
19:In
346::
259:40
257:.
231:83
229:.
204:71
202:.
177:.
123:.
329:e
322:t
315:v
304:.
269:.
265::
241:.
237::
214:.
210::
187:.
173::
154:.
93:O
79:N
77:S
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.