1479:
1113:
111:
162:
1586:
238:
1826:
1107:
20:
1648:
1352:
228:
profiles were evaluated and tested in an attempt to determine the optimal way to deliver the drug, which was especially important given the puzzling failure of an existing extended-release formulation of methylphenidate (Ritalin SR) to act as expected. The zero-order (flat) release profile that the
233:
that an ascending pattern of drug delivery was necessary to maintain clinical effect. Trials designed to test this hypothesis were successful, and ALZA subsequently developed a modified PPOP design that utilized an overcoat of methylphenidate designed to release immediately and rapidly raise serum
234:
levels, followed by 10 hours of first-order (ascending) drug delivery from the modified PPOP design. This design was called the Push-Stick
Osmotic Pump (PSOP), and utilized two separate drug layers with different concentrations of methylphenidate in addition to the (now quite robust) push layer.
140:
later developed the
Controlled-Porosity Osmotic Pump (CPOP) with the intention of addressing some of the issues that led to Osmosin's withdrawal via a new approach to the final stage of the release mechanism. Unlike the EOP, the CPOP had no pre-formed hole in the outer shell for the drug to be
23:
A 54 mg tablet of
Concerta, which uses OROS technology. 22% of the drug is contained in the red overcoat, while the remaining 78% is split between two drug layers of differing concentration. The tablet uses an additional push layer that expands as water enters the tablet via the osmotic
93:, and differing intestinal environments. Using an osmotic pump to deliver drugs has additional inherent advantages regarding control over drug delivery rates. This allows for much more precise drug delivery over an extended period of time, which results in much more predictable
229:
PPOP was optimal at delivering failed to maintain its efficacy over time, which suggested that acute tolerance to methylphenidate formed over the course of the day. This explained why
Ritalin SR was inferior to twice-daily Ritalin IR, and led to the
141:
expelled out of. Instead, the CPOP's semipermeable membrane was designed to form numerous small pores upon contact with water through which the drug would be expelled via osmotic pressure. The pores were formed via the use of a pH insensitive
97:. However, osmotic release systems are relatively complicated, somewhat difficult to manufacture, and may cause irritation or even blockage of the GI tract due to prolonged release of irritating drugs from the non-deformable tablet.
658:
van den Berg, G; van
Steveninck, F; Gubbens-Stibbe, JM; Schoemaker, HC; de Boer, AG; Cohen, AF (1990). "Influence of food on the bioavailability of metoprolol from an OROS system; a study in healthy volunteers".
791:
Auiler, JF; Liu, K; Lynch, JM; Gelotte, CK (2002). "Effect of food on early drug exposure from extended-release stimulants: results from the
Concerta, Adderall XR Food Evaluation (CAFE) Study".
205:
1024:
967:"Development of a new once-a-day formulation of methylphenidate for the treatment of attention-deficit/hyperactivity disorder: proof-of-concept and proof-of-product studies"
122:
in 1974, and was the first practical example of an osmotic pump based drug release system for oral use. It was introduced to the market in the early 1980s in
Osmosin (
1870:
748:
Modi, NB; Wang, B; Hu, WT; Gupta, SK (January 2000). "Effect of food on the pharmacokinetics of osmotic controlled-release methylphenidate HCl in healthy subjects".
521:
Conley, R; Gupta, SK; Sathyan, G (October 2006). "Clinical spectrum of the osmotic-controlled release oral delivery system (OROS), an advanced oral delivery form".
224:
required multiple doses to be administered each day to attain long-lasting coverage, which made it an ideal candidate for the OROS technology. Multiple candidate
85:
Osmotic release systems have a number of major advantages over other controlled-release mechanisms. They are significantly less affected by factors such as
1017:
705:
Bass, DM; Prevo, M; Waxman, DS (2002). "Gastrointestinal safety of an extended-release, nondeformable, oral dosage form (OROS: a retrospective study".
1010:
427:
Malaterre, V; Ogorka, J; Loggia, N; Gurny, R (November 2009). "Oral osmotically driven systems: 30 years of development and clinical use".
217:
197:. The initial design developed in 1982 by ALZA researchers was designated the Push-Pull Osmotic Pump (PPOP), and Procardia XL (
761:
45:
1381:
1964:
181:
polymer for suspension of poorly soluble drugs) out of the exit hole at a controlled rate. Osmotic agents such as
90:
1969:
1462:
467:
Theeuwes, F; Yum, SI; Haak, R; Wong, P (1991). "Systems for triggered, pulsed, and programmed drug delivery".
1559:
1402:
173:
drugs. This led to the development of an additional internal "push layer" composed of material (a swellable
169:
Both the EOP and CPOP were relatively simple designs, and were limited by their inability to deliver poorly
1933:
1895:
1238:
1054:
131:
869:
Eckenhoff, B; Yum, SI (April 1981). "The osmotic pump: novel research tool for optimizing drug regimens".
1847:
965:
Swanson, J; Gupta, S; Lam, A; Shoulson, I; Lerner, M; Modi, N; Lindemulder, E; Wigal, S (February 2003).
208:
An animation illustrating the exterior/interior compositions of a tablet of
Concerta, a PSOP OROS design.
1875:
1444:
1989:
1923:
1852:
1439:
1386:
1220:
1478:
1842:
1216:
1913:
1880:
1834:
1679:
1511:
1243:
1164:
1091:
1033:
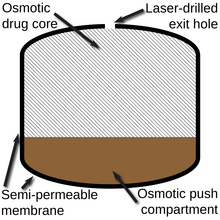
177:) that would expand as it absorbed water, which then pushed the drug layer (which incorporates a
146:
33:
24:
membrane. The drug is expelled via the laser-drilled hole visible on the left side of the tablet.
1908:
1862:
1804:
1710:
1577:
1184:
1058:
65:
49:
241:
An illustration of the different inner components of a tablet of
Concerta, a PSOP OROS design.
1984:
1974:
1918:
1799:
1212:
1979:
1763:
1608:
1526:
1471:
1391:
1376:
1179:
476:
142:
212:
In the early 1990s, an ALZA-funded research program began to develop a new dosage form of
8:
1816:
1553:
1434:
1429:
1149:
1045:
1002:
256:
127:
615:
Verma, RK; Mishra, B; Garg, S (July 2000). "Osmotically controlled oral drug delivery".
480:
1890:
1684:
1639:
1336:
1081:
816:
773:
730:
684:
640:
546:
500:
488:
186:
1740:
1735:
1573:
1314:
1076:
988:
917:
886:
882:
851:
808:
765:
722:
718:
676:
632:
594:
538:
492:
444:
37:
820:
777:
734:
644:
550:
504:
1674:
978:
909:
878:
843:
800:
757:
714:
688:
668:
624:
584:
530:
484:
436:
324:
225:
194:
94:
69:
57:
657:
110:
1564:
1248:
1112:
370:
328:
320:
316:
312:
284:
213:
182:
161:
1625:
1258:
804:
440:
137:
41:
983:
966:
936:
913:
1958:
1768:
1690:
1659:
1424:
1253:
534:
377:
335:
1809:
1745:
1730:
1705:
1543:
1263:
992:
847:
812:
769:
726:
636:
598:
542:
448:
349:
123:
114:
An illustration of the different components of the
Elementary Osmotic Pump.
921:
890:
680:
628:
496:
165:
An illustration of the different components of the Push-Pull Osmotic Pump.
1903:
1791:
1664:
1620:
1598:
1585:
1516:
1486:
1420:
1269:
1208:
1037:
204:
237:
1825:
1787:
1758:
1318:
1292:
1154:
855:
672:
384:
363:
305:
298:
263:
230:
198:
170:
1106:
1397:
1287:
1139:
1129:
342:
291:
277:
221:
178:
19:
589:
572:
1939:
1928:
1720:
1700:
1695:
1669:
1613:
1506:
1496:
1491:
1194:
1159:
1144:
1086:
762:
10.1002/1099-081x(200001)21:1<23::aid-bdd212>3.0.co;2-v
573:"Osmotically controlled drug delivery system with associated drugs"
356:
270:
150:
72:, which pioneered the use of osmotic pumps for oral drug delivery.
130:), but unexpectedly severe issues with GI irritation and cases of
1725:
1654:
1593:
1538:
1533:
1501:
1341:
1297:
1174:
904:
Heimlich, KR (1983). "The evolution of precision drug delivery".
190:
174:
53:
1647:
64:
through the laser drilled opening(s) in the tablet and into the
1715:
1635:
1548:
1368:
1279:
1169:
1134:
1121:
1603:
1328:
1230:
1189:
1068:
571:
Gupta, BP; Thakur, N; Jain, NP; Banweer, J; Jain, S (2010).
193:
are added to both the drug and push layers to increase the
119:
61:
40:
with a semi-permeable outer membrane and one or more small
1351:
1032:
426:
201:) was one of the first drugs to utilize this PPOP design.
1467:
1412:
834:
Theeuwes, F (December 1975). "Elementary osmotic pump".
964:
86:
570:
429:
European Journal of Pharmaceutics and Biopharmaceutics
30:
osmotic-controlled release oral delivery system (OROS)
790:
16:
Advanced controlled release oral drug delivery system
466:
118:The Elementary Osmotic Pump (EOP) was developed by
520:
577:Journal of Pharmacy & Pharmaceutical Sciences
36:oral drug delivery system in the form of a rigid
1956:
960:
958:
747:
704:
614:
422:
100:
700:
698:
610:
608:
566:
564:
562:
560:
516:
514:
420:
418:
416:
414:
412:
410:
408:
406:
404:
402:
1018:
462:
460:
458:
955:
868:
943:. United States Patent and Trademark Office
897:
862:
784:
741:
695:
605:
557:
511:
399:
245:
1025:
1011:
651:
469:Annals of the New York Academy of Sciences
455:
982:
661:European Journal of Clinical Pharmacology
588:
903:
833:
827:
617:Drug Development and Industrial Pharmacy
236:
218:attention deficit hyperactivity disorder
203:
160:
109:
18:
750:Biopharmaceutics & Drug Disposition
1957:
934:
68:. OROS is a trademarked name owned by
1006:
906:Current Medical Research and Opinion
793:Current Medical Research and Opinion
523:Current Medical Research and Opinion
1382:Heated humidified high-flow therapy
216:for the treatment of children with
13:
1646:
1584:
1477:
937:"Controlled porosity osmotic pump"
836:Journal of Pharmaceutical Sciences
489:10.1111/j.1749-6632.1991.tb27262.x
134:led to the withdrawal of Osmosin.
14:
2001:
935:Haslam, John L.; Rork, Gerald S.
1824:
1350:
1111:
1105:
719:10.2165/00002018-200225140-00004
220:(ADHD). Methylphenidate's short
80:
48:, water is absorbed through the
928:
105:
971:Archives of General Psychiatry
156:
1:
1150:Effervescent powder or tablet
392:
1934:Patient-controlled analgesia
1239:Orally disintegrating tablet
883:10.1016/0142-9612(81)90005-3
101:Oral osmotic release systems
75:
7:
60:is used to push the active
44:holes in it. As the tablet
10:
2006:
1445:Relative analgesia machine
805:10.1185/030079902125000840
441:10.1016/j.ejpb.2009.07.002
250:OROS medications include:
1889:
1861:
1833:
1822:
1786:
1753:
1634:
1572:
1525:
1461:
1411:
1367:
1348:
1327:
1313:
1278:
1229:
1207:
1120:
1103:
1067:
1053:
1044:
984:10.1001/archpsyc.60.2.204
914:10.1185/03007998309109821
1034:Routes of administration
535:10.1185/030079906x132613
297:Ditropan XL/Lyrinel XL (
246:List of OROS medications
1965:Pharmaceutical industry
1560:Extra-amniotic infusion
1165:Molecular encapsulation
1097:Osmotic delivery system
1092:Time release technology
46:passes through the body
1863:Central nervous system
1651:
1589:
1517:Mucoadhesive microdisc
1482:
848:10.1002/jps.2600641218
242:
209:
166:
115:
66:gastrointestinal tract
50:semipermeable membrane
25:
1970:Drug delivery devices
1706:Transfersome vesicles
1650:
1588:
1565:Intravesical infusion
1481:
629:10.1081/ddc-100101287
240:
207:
164:
113:
22:
1392:Metered-dose inhaler
1377:Anesthetic vaporizer
908:. 8 Suppl 2: 28–37.
56:, and the resulting
1817:Transdermal implant
1777:(into tissue/blood)
1554:Intrauterine device
1435:Anaesthetic machine
1430:Oxygen concentrator
481:1991NYASA.618..428T
257:phenylpropanolamine
128:phenylpropanolamine
1929:Nanocell injection
1652:
1590:
1483:
1337:Dry-powder inhaler
673:10.1007/bf00315121
243:
210:
187:potassium chloride
167:
116:
34:controlled release
26:
1952:
1951:
1948:
1947:
1779:
1741:Transdermal spray
1736:Transdermal patch
1726:Medicated shampoo
1457:
1456:
1453:
1452:
1440:Medical inhalants
1387:Medical inhalants
1315:Respiratory tract
1309:
1308:
1203:
1202:
334:Exalgo/Jurnista (
149:additive such as
1997:
1990:Pharmacokinetics
1828:
1784:
1783:
1775:
1362:
1358:
1354:
1325:
1324:
1254:Sublingual drops
1227:
1226:
1115:
1109:
1065:
1064:
1051:
1050:
1027:
1020:
1013:
1004:
1003:
997:
996:
986:
962:
953:
952:
950:
948:
932:
926:
925:
901:
895:
894:
866:
860:
859:
831:
825:
824:
788:
782:
781:
745:
739:
738:
702:
693:
692:
655:
649:
648:
612:
603:
602:
592:
568:
555:
554:
518:
509:
508:
464:
453:
452:
424:
325:chlorpheniramine
195:osmotic pressure
95:pharmacokinetics
70:ALZA Corporation
58:osmotic pressure
2005:
2004:
2000:
1999:
1998:
1996:
1995:
1994:
1955:
1954:
1953:
1944:
1924:Intraperitoneal
1896:musculoskeletal
1894:
1885:
1857:
1853:Intra-articular
1829:
1820:
1780:
1774:
1773:
1749:
1630:
1568:
1521:
1466:
1449:
1407:
1363:
1360:
1359:
1356:
1355:
1346:
1305:
1274:
1199:
1116:
1110:
1101:
1055:Digestive tract
1040:
1031:
1001:
1000:
963:
956:
946:
944:
933:
929:
902:
898:
867:
863:
842:(12): 1987–91.
832:
828:
789:
785:
746:
742:
713:(14): 1021–33.
703:
696:
656:
652:
613:
606:
590:10.18433/j38w25
569:
558:
529:(10): 1879–92.
519:
512:
465:
456:
425:
400:
395:
390:
371:pseudoephedrine
329:pseudoephedrine
321:pseudoephedrine
317:brompheniramine
313:pseudoephedrine
285:methylphenidate
248:
226:pharmacokinetic
214:methylphenidate
183:sodium chloride
159:
138:Merck & Co.
126:) and Acutrim (
108:
103:
89:, food intake,
83:
78:
32:is an advanced
17:
12:
11:
5:
2003:
1993:
1992:
1987:
1982:
1977:
1972:
1967:
1950:
1949:
1946:
1945:
1943:
1942:
1937:
1931:
1926:
1921:
1916:
1911:
1906:
1900:
1898:
1887:
1886:
1884:
1883:
1878:
1873:
1867:
1865:
1859:
1858:
1856:
1855:
1850:
1845:
1843:Intracavernous
1839:
1837:
1831:
1830:
1823:
1821:
1819:
1814:
1813:
1812:
1802:
1797:
1795:
1781:
1772:
1771:
1766:
1761:
1755:
1754:
1751:
1750:
1748:
1743:
1738:
1733:
1728:
1723:
1718:
1713:
1708:
1703:
1698:
1693:
1688:
1682:
1677:
1672:
1667:
1662:
1657:
1645:
1643:
1632:
1631:
1629:
1628:
1626:Nutrient enema
1623:
1618:
1617:
1616:
1611:
1601:
1596:
1583:
1581:
1570:
1569:
1567:
1562:
1557:
1551:
1546:
1541:
1536:
1531:
1529:
1523:
1522:
1520:
1519:
1514:
1509:
1504:
1499:
1494:
1489:
1476:
1474:
1459:
1458:
1455:
1454:
1451:
1450:
1448:
1447:
1442:
1437:
1432:
1427:
1417:
1415:
1409:
1408:
1406:
1405:
1400:
1395:
1389:
1384:
1379:
1373:
1371:
1365:
1364:
1349:
1347:
1345:
1344:
1339:
1333:
1331:
1322:
1311:
1310:
1307:
1306:
1304:
1303:
1300:
1295:
1290:
1284:
1282:
1276:
1275:
1273:
1272:
1267:
1261:
1256:
1251:
1246:
1241:
1235:
1233:
1224:
1205:
1204:
1201:
1200:
1198:
1197:
1192:
1187:
1182:
1177:
1172:
1167:
1162:
1157:
1152:
1147:
1142:
1137:
1132:
1126:
1124:
1118:
1117:
1104:
1102:
1100:
1099:
1094:
1089:
1084:
1079:
1073:
1071:
1062:
1048:
1042:
1041:
1030:
1029:
1022:
1015:
1007:
999:
998:
954:
941:Google Patents
927:
896:
861:
826:
783:
740:
694:
650:
623:(7): 695–708.
604:
556:
510:
454:
397:
396:
394:
391:
389:
388:
381:
374:
367:
362:Procardia XL (
360:
355:Minipress XL (
353:
346:
341:Glucotrol XL (
339:
332:
309:
302:
295:
288:
281:
274:
267:
260:
252:
247:
244:
158:
155:
132:GI perforation
107:
104:
102:
99:
82:
79:
77:
74:
15:
9:
6:
4:
3:
2:
2002:
1991:
1988:
1986:
1983:
1981:
1978:
1976:
1973:
1971:
1968:
1966:
1963:
1962:
1960:
1941:
1938:
1935:
1932:
1930:
1927:
1925:
1922:
1920:
1917:
1915:
1914:Intramuscular
1912:
1910:
1907:
1905:
1902:
1901:
1899:
1897:
1892:
1888:
1882:
1879:
1877:
1874:
1872:
1871:Intracerebral
1869:
1868:
1866:
1864:
1860:
1854:
1851:
1849:
1846:
1844:
1841:
1840:
1838:
1836:
1832:
1827:
1818:
1815:
1811:
1808:
1807:
1806:
1803:
1801:
1798:
1796:
1793:
1789:
1785:
1782:
1778:
1770:
1767:
1765:
1762:
1760:
1757:
1756:
1752:
1747:
1744:
1742:
1739:
1737:
1734:
1732:
1729:
1727:
1724:
1722:
1719:
1717:
1714:
1712:
1709:
1707:
1704:
1702:
1699:
1697:
1694:
1692:
1691:Iontophoresis
1689:
1686:
1683:
1681:
1678:
1676:
1673:
1671:
1668:
1666:
1663:
1661:
1660:Topical cream
1658:
1656:
1653:
1649:
1644:
1641:
1637:
1633:
1627:
1624:
1622:
1619:
1615:
1612:
1610:
1607:
1606:
1605:
1602:
1600:
1597:
1595:
1592:
1591:
1587:
1582:
1579:
1575:
1571:
1566:
1563:
1561:
1558:
1555:
1552:
1550:
1547:
1545:
1542:
1540:
1537:
1535:
1532:
1530:
1528:
1524:
1518:
1515:
1513:
1510:
1508:
1505:
1503:
1500:
1498:
1495:
1493:
1490:
1488:
1485:
1484:
1480:
1475:
1473:
1469:
1464:
1460:
1446:
1443:
1441:
1438:
1436:
1433:
1431:
1428:
1426:
1425:Nasal cannula
1422:
1419:
1418:
1416:
1414:
1410:
1404:
1401:
1399:
1396:
1393:
1390:
1388:
1385:
1383:
1380:
1378:
1375:
1374:
1372:
1370:
1366:
1353:
1343:
1340:
1338:
1335:
1334:
1332:
1330:
1326:
1323:
1320:
1316:
1312:
1301:
1299:
1296:
1294:
1291:
1289:
1286:
1285:
1283:
1281:
1277:
1271:
1268:
1265:
1262:
1260:
1257:
1255:
1252:
1250:
1247:
1245:
1242:
1240:
1237:
1236:
1234:
1232:
1228:
1225:
1222:
1218:
1214:
1210:
1206:
1196:
1193:
1191:
1188:
1186:
1183:
1181:
1178:
1176:
1173:
1171:
1168:
1166:
1163:
1161:
1158:
1156:
1153:
1151:
1148:
1146:
1143:
1141:
1138:
1136:
1133:
1131:
1128:
1127:
1125:
1123:
1119:
1114:
1108:
1098:
1095:
1093:
1090:
1088:
1085:
1083:
1080:
1078:
1075:
1074:
1072:
1070:
1066:
1063:
1060:
1056:
1052:
1049:
1047:
1043:
1039:
1035:
1028:
1023:
1021:
1016:
1014:
1009:
1008:
1005:
994:
990:
985:
980:
977:(2): 204–11.
976:
972:
968:
961:
959:
942:
938:
931:
923:
919:
915:
911:
907:
900:
892:
888:
884:
880:
876:
872:
865:
857:
853:
849:
845:
841:
837:
830:
822:
818:
814:
810:
806:
802:
798:
794:
787:
779:
775:
771:
767:
763:
759:
755:
751:
744:
736:
732:
728:
724:
720:
716:
712:
708:
701:
699:
690:
686:
682:
678:
674:
670:
666:
662:
654:
646:
642:
638:
634:
630:
626:
622:
618:
611:
609:
600:
596:
591:
586:
583:(4): 571–88.
582:
578:
574:
567:
565:
563:
561:
552:
548:
544:
540:
536:
532:
528:
524:
517:
515:
506:
502:
498:
494:
490:
486:
482:
478:
475:(1): 428–40.
474:
470:
463:
461:
459:
450:
446:
442:
438:
435:(3): 311–23.
434:
430:
423:
421:
419:
417:
415:
413:
411:
409:
407:
405:
403:
398:
386:
382:
379:
378:carbamazepine
376:Tegretol XR (
375:
372:
368:
365:
361:
358:
354:
351:
347:
344:
340:
337:
336:hydromorphone
333:
330:
326:
322:
318:
314:
310:
307:
304:Dynacirc CR (
303:
300:
296:
293:
289:
286:
282:
279:
275:
272:
268:
265:
262:Adalat OROS (
261:
258:
254:
253:
251:
239:
235:
232:
227:
223:
219:
215:
206:
202:
200:
196:
192:
188:
184:
180:
176:
172:
163:
154:
152:
148:
144:
139:
135:
133:
129:
125:
121:
112:
98:
96:
92:
88:
81:Pros and cons
73:
71:
67:
63:
59:
55:
51:
47:
43:
42:laser drilled
39:
35:
31:
21:
1985:Pharmacology
1975:Dosage forms
1919:Intraosseous
1909:Intracardiac
1848:Intravitreal
1810:Injector pen
1805:Subcutaneous
1776:
1746:Jet injector
1731:Dermal patch
1544:Vaginal ring
1512:Insufflation
1264:Effervescent
1096:
1038:dosage forms
974:
970:
945:. Retrieved
940:
930:
905:
899:
877:(2): 89–97.
874:
871:Biomaterials
870:
864:
839:
835:
829:
799:(5): 311–6.
796:
792:
786:
756:(1): 23–31.
753:
749:
743:
710:
706:
667:(3): 315–6.
664:
660:
653:
620:
616:
580:
576:
526:
522:
472:
468:
432:
428:
369:Sudafed 24 (
350:paliperidone
276:Cardura XL (
269:Alpress LP (
249:
211:
168:
136:
124:indomethacin
117:
106:Single-layer
84:
29:
27:
1980:Alza brands
1904:Intravenous
1891:Circulatory
1876:Intrathecal
1800:Intradermal
1792:transdermal
1759:Parenterals
1665:Topical gel
1621:Murphy drip
1599:Suppository
1487:Nasal spray
1421:Oxygen mask
1270:Chewing gum
1209:Oral mucosa
707:Drug Safety
311:Efidac 24 (
290:Covera HS (
157:Multi-layer
147:dissolvable
91:GI motility
1959:Categories
1764:Injections
1527:Urogenital
1463:Ophthalmic
1319:inhalation
1293:Toothpaste
1221:sublingual
1185:Suspension
1155:Herbal tea
393:References
385:salbutamol
364:nifedipine
306:isradipine
299:oxybutynin
283:Concerta (
264:nifedipine
231:hypothesis
199:nifedipine
1769:infusions
1701:Liposomes
1497:Eye drops
1492:Ear drops
1403:Vaporizer
1398:Nebulizer
1288:Mouthwash
1217:sublabial
1140:Electuary
1130:Decoction
343:glipizide
292:verapamil
278:doxazosin
255:Acutrim (
222:half-life
143:leachable
76:Rationale
1940:PIC line
1881:Epidural
1721:Lip balm
1696:Hydrogel
1687:solution
1670:Liniment
1655:Ointment
1614:Hydrogel
1609:Solution
1594:Ointment
1534:Ointment
1507:Hydrogel
1502:Ointment
1298:Ointment
1259:Lozenges
1249:Lollipop
1195:Tincture
1180:Solution
1160:Hydrogel
1145:Emulsion
1087:Pastille
993:12578439
947:19 March
821:25994524
813:12240794
778:33413277
770:11038435
735:35424637
727:12408733
645:35670161
637:10872087
599:21486532
551:42490425
543:17022845
505:31442663
449:19602438
383:Volmax (
357:prazosin
348:Invega (
271:prazosin
151:sorbitol
1640:topical
1578:enteral
1539:Pessary
1369:Liquids
1342:Smoking
1280:Liquids
1175:Softgel
1122:Liquids
1082:Capsule
1059:enteral
922:6851623
891:7248427
689:1838636
681:2257873
497:2006800
477:Bibcode
191:xylitol
179:viscous
175:polymer
171:soluble
54:osmosis
1835:Organs
1716:Lotion
1636:Dermal
1574:Rectal
1549:Douche
1329:Solids
1266:tablet
1231:Solids
1213:buccal
1170:Powder
1135:Elixir
1077:Tablet
1069:Solids
991:
920:
889:
854:
819:
811:
776:
768:
733:
725:
687:
679:
643:
635:
597:
549:
541:
503:
495:
447:
38:tablet
1711:Cream
1675:Paste
1604:Enema
1556:(IUD)
1472:nasal
1394:(MDI)
1302:Spray
1190:Syrup
817:S2CID
774:S2CID
731:S2CID
685:S2CID
641:S2CID
547:S2CID
501:S2CID
189:, or
1936:pump
1788:Skin
1685:DMSO
1680:Film
1468:otic
1423:and
1244:Film
1046:Oral
989:PMID
949:2016
918:PMID
887:PMID
856:1510
852:PMID
809:PMID
766:PMID
723:PMID
677:PMID
633:PMID
595:PMID
539:PMID
493:PMID
445:PMID
120:ALZA
62:drug
52:via
28:The
1413:Gas
979:doi
910:doi
879:doi
844:doi
801:doi
758:doi
715:doi
669:doi
625:doi
585:doi
531:doi
485:doi
473:618
437:doi
145:or
1961::
1470:,
1219:,
1215:,
1036:,
987:.
975:60
973:.
969:.
957:^
939:.
916:.
885:.
873:.
850:.
840:64
838:.
815:.
807:.
797:18
795:.
772:.
764:.
754:21
752:.
729:.
721:.
711:25
709:.
697:^
683:.
675:.
665:39
663:.
639:.
631:.
621:26
619:.
607:^
593:.
581:13
579:.
575:.
559:^
545:.
537:.
527:22
525:.
513:^
499:.
491:.
483:.
471:.
457:^
443:.
433:73
431:.
401:^
327:,
319:,
185:,
153:.
87:pH
1893:,
1794:)
1790:(
1642:)
1638:(
1580:)
1576:(
1465:,
1361:0
1357:0
1321:)
1317:(
1223:)
1211:(
1061:)
1057:(
1026:e
1019:t
1012:v
995:.
981::
951:.
924:.
912::
893:.
881::
875:2
858:.
846::
823:.
803::
780:.
760::
737:.
717::
691:.
671::
647:.
627::
601:.
587::
553:.
533::
507:.
487::
479::
451:.
439::
387:)
380:)
373:)
366:)
359:)
352:)
345:)
338:)
331:)
323:/
315:/
308:)
301:)
294:)
287:)
280:)
273:)
266:)
259:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.