42:
2323:
2242:
988:). Solutions produced in a laboratory may contain a virtually limitless number of species that contribute to alkalinity. Alkalinity is frequently given as molar equivalents per liter of solution or per kilogram of solvent. In commercial (e.g. the swimming pool industry) and regulatory contexts, alkalinity might also be given in parts per million of equivalent calcium carbonate (ppm CaCO
1527:
The lower the pH, the higher the concentration of bicarbonate will be. This shows how a lower pH can lead to higher alkalinity if the amount of bicarbonate produced is greater than the amount of H remaining after the reaction. This is the case since the amount of acid in the rainwater is low. If this
1552:
shows that pH will be related to calcium ion concentration, with lower pH going with higher calcium ion concentration. In this case, the higher the pH, the more bicarbonate and carbonate ion there will be, in contrast to the paradoxical situation described above, where one does not have equilibrium
1460:
will increase alkalinity by 2 units. Increased dissolution of carbonate rock by acidification from acid rain and mining has contributed to increased alkalinity concentrations in some major rivers throughout the eastern U.S. The following reaction shows how acid rain, containing sulfuric acid, can
1741:
techniques add alkalinity to the ocean and therefore immediately buffer pH changes which might help the organisms in the region that the extra alkalinity is added to. The two technologies that fall into this category are ocean alkalinity enhancement and electrochemical methods. Eventually, due to
1102:
Alkalinity can be measured by titrating a sample with a strong acid until all the buffering capacity of the aforementioned ions above the pH of bicarbonate or carbonate is consumed. This point is functionally set to pH 4.5. At this point, all the bases of interest have been protonated to the
879:
1431:
to a solution in contact with a solid can (over time) affect the alkalinity, especially for carbonate minerals in contact with groundwater or seawater. The dissolution (or precipitation) of carbonate rock has a strong influence on the alkalinity. This is because carbonate rock is composed of
1688:
and sedimentation of carbonate minerals (for example, as a function of ocean acidification) are the primary long-term drivers of alkalinity in the ocean. Over human timescales, mean ocean alkalinity is relatively stable. Seasonal and annual variability of mean ocean alkalinity is very low.
390:
are affected by changes in pH, temperature, and pressure. By isolating the conservative ions on one side of this charge balance equation, the nonconservative ions which accept or donate protons and thus define alkalinity are clustered on the other side of the equation.
295:
is 2 molar equivalents because twice as many H ions would be necessary to balance the charge. The total charge of a solution always equals zero. This leads to a parallel definition of alkalinity that is based upon the charge balance of ions in a solution.
1622:(carbonate). The carbonate ion has the potential to absorb two hydrogen ions. Therefore, it causes a net increase in ocean alkalinity. Calcium carbonate dissolution occurs in regions of the ocean which are undersaturated with respect to calcium carbonate.
1131:, although it adds acid and dissolved inorganic carbon, does not change the alkalinity. In natural conditions, the dissolution of basic rocks and addition of ammonia or organic amines leads to the addition of base to natural waters at the CO
397:
1139:
to bicarbonate ion and carbonate ion. At equilibrium, the water contains a certain amount of alkalinity contributed by the concentration of weak acid anions. Conversely, the addition of acid converts weak acid anions to
236:, that is now known as the Principle of Constant Proportions. However, there was one exception. Dittmar found that the concentration of calcium was slightly greater in the deep ocean, and named this increase alkalinity.
200:. It is one of the best measures of the sensitivity of the stream to acid inputs. There can be long-term changes in the alkalinity of streams and rivers in response to human disturbances such as acid rain generated by SO
1746:
is used. Both of these technologies have the potential to operate on a large scale and to be efficient at removing carbon dioxide. However, they are expensive, have many risks and side effects and currently have a low
1103:
zero level species, hence they no longer cause alkalinity. In the carbonate system the bicarbonate ions and the carbonate ions have become converted to carbonic acid at this pH. This pH is also called the CO
345:
1144:
and continuous addition of strong acids can cause the alkalinity to become less than zero. For example, the following reactions take place during the addition of acid to a typical seawater solution:
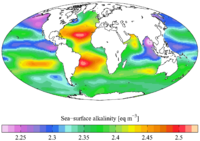
1710:(SST). Therefore, it generally increases with high latitudes and depths. As a result, upwelling areas (where water from the deep ocean is pushed to the surface) also have higher alkalinity values.
402:
888:. Total alkalinity is not (much) affected by temperature, pressure, or pH, and is thus itself a conservative measurement, which increases its usefulness in aquatic systems. All anions except
1095:(Subscript T indicates the total concentration of the species in the solution as measured. This is opposed to the free concentration, which takes into account the significant amount of
1332:
to a solution does not change its alkalinity, since the net reaction produces the same number of equivalents of positively contributing species (H) as negative contributing species (
1713:
There are many programs to measure, record, and study oceanic alkalinity, together with many of the other characteristics of seawater, like temperature and salinity. These include:
1695:
River dominated mixing also occurs close to the shore; it is strongest close to the mouth of a large river. Here, the rivers can act as either a source or a sink of alkalinity. A
2552:
Thomas, H.; Schiettecatte, L.-S.; et al. Enhanced Ocean Carbon
Storage from Anaerobic Alkalinity Generation in Coastal Sediments. Biogeosciences Discussions. 2008, 5, 3575–3591
1257:
It can be seen from the above protonation reactions that most bases consume one proton (H) to become a neutral species, thus increasing alkalinity by one per equivalent.
2732:
Climate Change 2022: Mitigation of
Climate Change. Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change
1532:, precipitate carbonate, and thereby become less alkaline again. When carbonate minerals, water, and the atmosphere are all in equilibrium, the reversible reaction
2270:
Climate Change 2021: The
Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change
2723:
271:
Alkalinity roughly refers to the molar amount of bases in a solution that can be converted to uncharged species by a strong acid. For example, 1 mole of
2181:
1300:. (The conjunction "as" is appropriate in this case because the alkalinity results from a mixture of ions but is reported "as if" all of this is due to CaCO
2261:
874:{\displaystyle {\begin{aligned}&\sum ({\text{conservative cations}})-\sum ({\text{conservative anions}})=\\&\quad +2++++2+++-----\end{aligned}}}
1119:
in an aqueous solution. There are no strong acids or bases at this point. Therefore, the alkalinity is modeled and quantified with respect to the CO
130:
until its pH changes abruptly, or it reaches a known endpoint where that happens. Alkalinity is expressed in units of concentration, such as meq/L (
2561:
Millero, F. J.; Lee, K.; Roche, M. Distribution of alkalinity in the surface waters of the major oceans. Marine
Chemistry. 1998, 60, 111-130.
1684:
The ocean's alkalinity varies over time, most significantly over geologic timescales (millennia). Changes in the balance between terrestrial
1995:
Kaushal, S. S.; Likens, G. E.; Utz, R. M.; Pace, M. L.; Grese, M.; Yepsen, M. (2013). "Increased river alkalinization in the
Eastern U.S.".
1692:
Alkalinity varies by location depending on evaporation/precipitation, advection of water, biological processes, and geochemical processes.
1731:
1717:(Geochemical Ocean Sections Study), TTO/NAS (Transient Tracers in the Ocean/North Atlantic Study), JGOFS (Joint Global Ocean Flux Study),
2105:"An exact definition of total alkalinity and a procedure for the estimation of alkalinity and total inorganic carbon from titration data"
1637:
from the atmosphere into the oceans. This does not affect the ocean's alkalinity but it does result in a reduction in pH value (called
1660:
occur in oxygen-limited environments. Both of these processes consume hydrogen ions (thus increasing alkalinity) and release gases (N
302:
1626:
161:
terrains, alkalinities are low and involve a lot of ions. In the ocean, on the other hand, alkalinity is completely dominated by
2825:
2688:
2471:
2401:
2355:
1949:
1904:
2819:
936:
of carbonate or bicarbonate, defined as pH 4.5 for many oceanographic/limnological studies. The alkalinity is equal to the
1574:
2777:
2755:
1781:
1718:
46:
2854:
2436:
2160:
2048:
1828:
2720:
192:. Moreover, measuring alkalinity is important in determining a stream's ability to neutralize acidic pollution from
2849:
2759:
Handbook of methods for the analysis of the various parameters of the carbon dioxide system in sea water. Version 2
1714:
956:
processes that produce carbonate anions. Other common natural components that can contribute to alkalinity include
1893:
Chester, R.; Jickells, Tim (2012). "Chapter 9: Nutrients oxygen organic carbon and the carbon cycle in seawater".
1648:
Biological processes have a much greater impact on oceanic alkalinity on short (minutes to centuries) timescales.
2734:
2272:
1894:
2258:
2147:, Standard Methods for the Examination of Water and Wastewater, American Public Health Association, 2017-08-27,
2064:
Wolf-Gladrow, Dieter A.; Zeebe, Richard E.; Klaas, Christine; Körtzinger, Arne; Dickson, Andrew G. (July 2007).
2844:
1657:
2620:
1673:
1642:
1285:. and decrease alkalinity, as they act as sources of protons. They are often represented collectively as
2496:
1929:
1645:
has been proposed as one option to add alkalinity to the ocean and therefore buffer against pH changes.
2066:"Total alkalinity: The explicit conservative expression and its application to biogeochemical processes"
260:
1748:
1135:
equivalence point. The dissolved base in water increases the pH and titrates an equivalent amount of CO
1304:.) This can be converted into milliequivalents per Liter (meq/L) by dividing by 50 (the approximate
225:
1702:
Oceanic alkalinity also follows general trends based on latitude and depth. It has been shown that A
2803:
1630:
256:
95:
2595:
2065:
1776:
1707:
233:
17:
2104:
1742:
diffusion, that alkalinity addition will be quite small to distant waters. This is why the term
2799:
2770:
The following packages calculate the state of the carbonate system in seawater (including pH):
232:. He found that in seawater the major ions were in a fixed ratio, confirming the hypothesis of
69:
948:
tends to make up most of the total alkalinity due to the common occurrence and dissolution of
229:
2142:
1853:
Dickson, Andrew G. (1992). "The development of the alkalinity concept in marine chemistry".
1461:
have the effect of increasing river alkalinity by increasing the amount of bicarbonate ion:
2508:
2116:
2077:
1862:
1649:
945:
378:" such that they are unaffected by changes in temperature, pressure or pH. Others such as
123:
2327:
2307:
2290:
2246:
2226:
2209:
8:
2520:
1638:
1570:
1000:
lowers the pH of a solution, thus reducing basicity while alkalinity remains unchanged (
81:
2512:
2120:
2081:
1866:
2702:
2495:
Doney, Scott C.; Fabry, Victoria J.; Feely, Richard A.; Kleypas, Joan A. (2009-01-01).
2369:
2312:
2291:"The Impacts of Ocean Acidification on Marine Ecosystems and Reliant Human Communities"
2289:
Doney, Scott C.; Busch, D. Shallin; Cooley, Sarah R.; Kroeker, Kristy J. (2020-10-17).
2231:
2210:"The Impacts of Ocean Acidification on Marine Ecosystems and Reliant Human Communities"
2208:
Doney, Scott C.; Busch, D. Shallin; Cooley, Sarah R.; Kroeker, Kristy J. (2020-10-17).
2037:
1941:
189:
1970:
2807:
2706:
2694:
2684:
2532:
2524:
2477:
2467:
2442:
2432:
2407:
2397:
2373:
2361:
2351:
2316:
2235:
2175:
2156:
2128:
2044:
2012:
1945:
1910:
1900:
1874:
1721:(World Ocean Circulation Experiment), CARINA (Carbon dioxide in the Atlantic Ocean).
1676:
both decrease alkalinity by releasing protons as a byproduct of oxidation reactions.
1610:. Perhaps the most well known is the dissolution of calcium carbonate to form Ca and
1032:
1016:
933:
912:
have low concentrations in Earth's surface water (streams, rivers, and lakes). Thus
143:
2676:
2571:
2516:
2343:
2302:
2221:
2148:
2124:
2085:
2004:
1937:
1870:
1766:
993:
941:
240:
131:
99:
35:
2089:
228:, analysed 77 pristine seawater samples from around the world brought back by the
2789:
2781:
2738:
2727:
2276:
2265:
1771:
1653:
1528:
alkaline groundwater later comes into contact with the atmosphere, it can lose CO
1024:
221:
107:
60:
1806:
1738:
1699:
follows the outflow of the river and has a linear relationship with salinity.
977:
937:
115:
27:
Capacity of water to resist changes in pH that would make the water more acidic
2838:
2645:
2528:
2481:
2411:
2279:. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA
1914:
1669:
1020:
981:
2774:
2741:, Cambridge University Press, Cambridge, United Kingdom and New York, NY, US
2673:
A Research
Strategy for Ocean-based Carbon Dioxide Removal and Sequestration
2446:
2340:
A Research
Strategy for Ocean-based Carbon Dioxide Removal and Sequestration
1123:
equivalence point. Because the alkalinity is measured with respect to the CO
41:
2698:
2536:
2365:
2016:
1801:
1761:
1012:
185:
181:
146:). Each of these measurements corresponds to an amount of acid added as a
2461:
2391:
2152:
1269:
however, will consume two protons before becoming a zero-level species (CO
2426:
1584:
1044:
166:
154:
49:
2428:
Aquatic chemistry : chemical equilibria and rates in natural waters
1562:
2785:
1685:
1566:
1305:
1028:
953:
197:
2731:
2269:
2008:
1732:
Ocean acidification § Carbon removal technologies which add alkalinity
920:
is also approximately equal to the total alkalinity in surface water.
1580:
1008:
965:
961:
949:
252:
248:
193:
177:
162:
158:
119:
111:
1107:
equivalence point where the major component in water is dissolved CO
2811:
2713:
2680:
2347:
1096:
1048:
969:
87:
2624:
992:). Alkalinity is sometimes incorrectly used interchangeably with
985:
973:
147:
2063:
1652:
of organic matter can decrease alkalinity by releasing protons.
283:
in solution represents 1 molar equivalent, while 1 mole of
243:
submitted his PhD theses in which he advocated the existence of
2829:
2793:
1588:
1360:
to the solution lowers its pH, but does not affect alkalinity.
957:
170:
31:
1899:(3rd ed.). Chichester, West Sussex, UK: Wiley/Blackwell.
340:{\displaystyle \sum ({\text{cations}})=\sum ({\text{anions}})}
2815:
2326:
Text was copied from this source, which is available under a
2322:
2245:
Text was copied from this source, which is available under a
2241:
1607:
135:
91:
1934:
Reference Module in Earth
Systems and Environmental Sciences
928:
Alkalinity measures the ability of a solution to neutralize
1594:
Thus the chemical equation for alkalinity in seawater is:
1015:
are specified for various alkalinity measurement methods.
929:
2667:
2665:
1679:
884:
This combined charge balance and proton balance is called
244:
127:
2662:
2599:
2288:
2207:
2109:
Deep Sea
Research Part A. Oceanographic Research Papers
1606:
There are many methods of alkalinity generation in the
138:), μeq/kg (microequivalents per kilogram), or mg/L CaCO
103:
2494:
2463:
CO2 in seawater : equilibrium, kinetics, isotopes
2328:
Creative
Commons Attribution 4.0 International License
2247:
Creative Commons Attribution 4.0 International License
2251:
1448:
into solution. Ca will not influence alkalinity, but
400:
305:
2198:. 2nd Ed. Long Grove, Illinois: Waveland Press, Inc.
1994:
1579:
In the ocean, alkalinity is completely dominated by
74:
2761:, A. G. Dickson & C. Goyet, eds. ORNL/CDIAC-74.
2460:Zeebe, Richard E.; Wolf-Gladrow, Dieter A. (2001).
1990:
1988:
1273:), thus it increases alkalinity by two per mole of
2596:"Home : Woods Hole Oceanographic Institution"
2039:The Geochemistry of Natural Waters, Second Edition
2036:
1724:
873:
339:
2548:
2546:
2459:
2393:Chemical oceanography and the marine carbon cycle
2180:: CS1 maint: DOI inactive as of September 2024 (
1668:S), which eventually escape into the atmosphere.
1051:, the measured total alkalinity is set equal to:
2836:
1985:
1556:
1422:
176:Although alkalinity is primarily a term used by
2765:
1892:
2543:
1315:
2497:"Ocean Acidification: The Other CO 2 Problem"
2389:
1973:. United States Environment Protection Agency
2424:
944:in solution. In most Earth surface waters
350:Certain ions, including Na, K, Ca, Mg, Cl,
255:ion donors. For that work, he received the
2295:Annual Review of Environment and Resources
2214:Annual Review of Environment and Resources
102:, which is an absolute measurement on the
2396:. Cambridge: Cambridge University Press.
2306:
2225:
1848:
1846:
1292:Alkalinity is typically reported as mg/L
2425:Stumm, Werner; Morgan, James J. (1996).
1633:, results in increasing absorption of CO
1127:equivalence point, the dissolution of CO
1038:
40:
2721:Chapter 12: Cross sectoral perspectives
2102:
2043:. Englewood Cliffs, NJ: Prentice Hall.
1927:
1852:
1680:Global temporal and spatial variability
923:
106:scale. Alkalinity is the strength of a
14:
2837:
2390:Emerson, Steven; Hedges, John (2008).
2034:
1997:Environmental Science & Technology
1843:
1627:carbon dioxide level in the atmosphere
1099:interactions that occur in seawater.)
1023:acids are common acid titrants, while
2385:
2383:
2308:10.1146/annurev-environ-012320-083019
2227:10.1146/annurev-environ-012320-083019
1436:and its dissociation will add Ca and
266:
261:Svante Arrhenius#Ionic disassociation
2521:10.1146/annurev.marine.010908.163834
2201:
2030:
2028:
2026:
1888:
1886:
1884:
1744:local ocean acidification mitigation
1001:
2332:
2282:
1706:is often inversely proportional to
1575:Effects of climate change on oceans
996:. For example, the addition of CO
64:
24:
2380:
1942:10.1016/b978-0-12-409548-9.09397-0
1782:Global Ocean Data Analysis Project
1320:
854:
850:
847:
827:
823:
814:
799:
796:
771:
767:
764:
744:
723:
719:
699:
695:
670:
666:
663:
654:
625:
621:
590:
586:
583:
563:
559:
530:
527:
521:
493:
489:
461:
457:
454:
247:in solution, and defined acids as
25:
2866:
2748:
2431:(3rd ed.). New York: Wiley.
2023:
1881:
98:. It should not be confused with
45:Sea surface alkalinity (from the
2321:
2240:
1730:This section is an excerpt from
1504:Another way of writing this is:
1391:Only at high (basic) pH values:
2638:
2613:
2588:
2564:
2555:
2501:Annual Review of Marine Science
2488:
2453:
2418:
2188:
2135:
2096:
1725:Interventions to add alkalinity
1587:plus a small contribution from
449:
169:plus a small contribution from
2057:
1963:
1921:
1821:
1794:
864:
843:
837:
809:
803:
792:
786:
760:
754:
739:
733:
715:
709:
691:
685:
649:
643:
617:
608:
579:
573:
555:
549:
534:
524:
517:
511:
485:
476:
450:
436:
428:
419:
411:
334:
326:
317:
309:
13:
1:
2784:, available as a stand-alone
2090:10.1016/j.marchem.2007.01.006
1928:Mattson, M. D. (2014-01-01),
1831:. Water Research Center. 2014
1787:
1557:Changes to oceanic alkalinity
1423:Dissolution of carbonate rock
224:of Anderson College, now the
2766:Carbonate system calculators
2623:. 2011-10-16. Archived from
2598:. 2012-03-14. Archived from
2129:10.1016/0198-0149(81)90121-7
1875:10.1016/0304-4203(92)90047-E
1643:Ocean alkalinity enhancement
157:, particularly those on non-
7:
2621:"WOCE Global Data Resource"
2103:Dickson, A.G. (June 1981).
1930:"Alkalinity of Freshwater☆"
1755:
1328:Addition (or removal) of CO
1316:Carbon dioxide interactions
952:rocks and other geological
75:
10:
2871:
1749:technology readiness level
1729:
1560:
215:
90:') is the capacity of
29:
2035:Drever, James I. (1988).
1807:Dictionary.com Unabridged
1567:pH § pH in the ocean
226:University of Strathclyde
222:Wilhelm (William) Dittmar
142:(milligrams per liter of
2855:Water quality indicators
2194:Benjamin. Mark M. 2015.
1631:carbon dioxide emissions
257:Nobel Prize in Chemistry
251:ion donors and bases as
30:Not to be confused with
2726:13 October 2022 at the
2576:iridl.ldeo.columbia.edu
2466:. Amsterdam: Elsevier.
2155:(inactive 2024-09-12),
1777:Dealkalization of water
1708:sea surface temperature
1563:Ocean § Alkalinity
1111:which is converted to H
1035:are common indicators.
234:Johan Georg Forchhammer
875:
341:
53:
2845:Chemical oceanography
2737:2 August 2022 at the
2275:9 August 2021 at the
2153:10.2105/smww.2882.023
1829:"What is alkalinity?"
1553:with the atmosphere.
1039:Theoretical treatment
876:
342:
230:Challenger expedition
184:, it is also used by
126:with an acid such as
44:
946:carbonate alkalinity
924:Detailed description
916:, which is equal to
914:carbonate alkalinity
416:conservative cations
398:
303:
118:. It is measured by
2850:Acid–base chemistry
2513:2009ARMS....1..169D
2264:5 June 2022 at the
2259:Annex VII: Glossary
2121:1981DSRA...28..609D
2082:2007MarCh.106..287W
2003:(18): 10302–10311.
1896:Marine geochemistry
1867:1992MarCh..40...49D
1650:Aerobic respiration
1639:ocean acidification
1571:Ocean acidification
784:
683:
641:
606:
547:
509:
474:
433:conservative anions
220:In 1884, Professor
2780:2011-10-14 at the
2572:"dataset: GEOSECS"
1971:"Total Alkalinity"
1363:At all pH values:
871:
869:
770:
669:
624:
589:
533:
492:
460:
337:
267:Simplified summary
259:in 1903. See also
190:temporary hardness
86:'ashes of the
54:
2690:978-0-309-08761-2
2650:www.pmel.noaa.gov
2473:978-0-08-052922-6
2403:978-0-511-64987-5
2357:978-0-309-08761-2
2009:10.1021/es401046s
1951:978-0-12-409548-9
1906:978-1-118-34909-0
1674:sulfide oxidation
1658:sulfate reduction
1033:bromocresol green
1011:, endpoints, and
1002:see example below
934:equivalence point
434:
417:
332:
315:
144:calcium carbonate
85:
73:
16:(Redirected from
2862:
2818:(also available
2792:spreadsheet, or
2742:
2717:
2711:
2710:
2669:
2660:
2659:
2657:
2656:
2642:
2636:
2635:
2633:
2632:
2617:
2611:
2610:
2608:
2607:
2592:
2586:
2585:
2583:
2582:
2568:
2562:
2559:
2553:
2550:
2541:
2540:
2492:
2486:
2485:
2457:
2451:
2450:
2422:
2416:
2415:
2387:
2378:
2377:
2336:
2330:
2325:
2320:
2310:
2286:
2280:
2255:
2249:
2244:
2239:
2229:
2205:
2199:
2192:
2186:
2185:
2179:
2171:
2170:
2169:
2139:
2133:
2132:
2100:
2094:
2093:
2076:(1–2): 287–300.
2070:Marine Chemistry
2061:
2055:
2054:
2042:
2032:
2021:
2020:
1992:
1983:
1982:
1980:
1978:
1967:
1961:
1960:
1959:
1958:
1925:
1919:
1918:
1890:
1879:
1878:
1855:Marine Chemistry
1850:
1841:
1840:
1838:
1836:
1825:
1819:
1818:
1816:
1815:
1798:
1767:Base (chemistry)
1621:
1620:
1619:
1523:
1522:
1521:
1500:
1499:
1498:
1488:
1487:
1486:
1459:
1458:
1457:
1447:
1446:
1445:
1417:
1416:
1415:
1405:
1404:
1403:
1386:
1385:
1384:
1355:
1354:
1353:
1343:
1342:
1341:
1284:
1283:
1282:
1268:
1267:
1266:
1248:
1247:
1246:
1238:
1237:
1227:
1226:
1225:
1212:
1211:
1210:
1202:
1201:
1191:
1190:
1189:
1158:
1157:
1156:
919:
911:
910:
909:
899:
898:
897:
886:total alkalinity
880:
878:
877:
872:
870:
863:
862:
861:
836:
835:
834:
822:
821:
802:
785:
783:
778:
753:
752:
751:
732:
731:
730:
708:
707:
706:
684:
682:
677:
662:
661:
642:
640:
632:
607:
605:
597:
572:
571:
570:
548:
546:
541:
510:
508:
500:
475:
473:
468:
445:
435:
432:
418:
415:
404:
389:
388:
387:
373:
372:
371:
361:
360:
359:
346:
344:
343:
338:
333:
330:
316:
313:
294:
293:
292:
282:
281:
280:
241:Svante Arrhenius
132:milliequivalents
80:
78:
68:
66:
36:base (chemistry)
21:
2870:
2869:
2865:
2864:
2863:
2861:
2860:
2859:
2835:
2834:
2782:Wayback Machine
2768:
2751:
2746:
2745:
2739:Wayback Machine
2728:Wayback Machine
2718:
2714:
2691:
2671:
2670:
2663:
2654:
2652:
2644:
2643:
2639:
2630:
2628:
2619:
2618:
2614:
2605:
2603:
2594:
2593:
2589:
2580:
2578:
2570:
2569:
2565:
2560:
2556:
2551:
2544:
2493:
2489:
2474:
2458:
2454:
2439:
2423:
2419:
2404:
2388:
2381:
2358:
2338:
2337:
2333:
2287:
2283:
2277:Wayback Machine
2266:Wayback Machine
2256:
2252:
2206:
2202:
2196:Water Chemistry
2193:
2189:
2173:
2172:
2167:
2165:
2163:
2144:2320 alkalinity
2141:
2140:
2136:
2101:
2097:
2062:
2058:
2051:
2033:
2024:
1993:
1986:
1976:
1974:
1969:
1968:
1964:
1956:
1954:
1952:
1926:
1922:
1907:
1891:
1882:
1851:
1844:
1834:
1832:
1827:
1826:
1822:
1813:
1811:
1800:
1799:
1795:
1790:
1772:Biological pump
1758:
1753:
1752:
1735:
1727:
1705:
1698:
1682:
1667:
1663:
1654:Denitrification
1636:
1625:The increasing
1618:
1615:
1614:
1613:
1611:
1601:
1577:
1559:
1547:
1543:
1540:+ 2 H ⇌ Ca + CO
1539:
1531:
1520:
1517:
1516:
1515:
1513:
1511:
1497:
1494:
1493:
1492:
1490:
1485:
1482:
1481:
1480:
1478:
1476:
1472:
1468:
1456:
1453:
1452:
1451:
1449:
1444:
1441:
1440:
1439:
1437:
1435:
1430:
1425:
1414:
1411:
1410:
1409:
1407:
1402:
1399:
1398:
1397:
1395:
1383:
1380:
1379:
1378:
1376:
1374:
1370:
1359:
1352:
1349:
1348:
1347:
1345:
1340:
1337:
1336:
1335:
1333:
1331:
1326:
1324:
1318:
1312:divided by 2).
1311:
1303:
1299:
1288:
1281:
1278:
1277:
1276:
1274:
1272:
1265:
1262:
1261:
1260:
1258:
1245:
1242:
1241:
1240:
1236:
1233:
1232:
1231:
1229:
1224:
1221:
1220:
1219:
1217:
1209:
1206:
1205:
1204:
1200:
1197:
1196:
1195:
1193:
1188:
1185:
1184:
1183:
1181:
1175:
1166:
1162:
1155:
1152:
1151:
1150:
1148:
1143:
1138:
1134:
1130:
1126:
1122:
1118:
1114:
1110:
1106:
1090:
1086:
1082:
1078:
1074:
1070:
1066:
1062:
1058:
1041:
999:
991:
978:conjugate bases
926:
917:
908:
905:
904:
903:
901:
896:
893:
892:
891:
889:
868:
867:
857:
853:
846:
830:
826:
817:
813:
812:
795:
779:
774:
763:
747:
743:
742:
726:
722:
718:
702:
698:
694:
678:
673:
657:
653:
652:
633:
628:
620:
598:
593:
582:
566:
562:
558:
542:
537:
520:
501:
496:
488:
469:
464:
453:
443:
442:
431:
414:
401:
399:
396:
395:
386:
383:
382:
381:
379:
370:
367:
366:
365:
363:
358:
355:
354:
353:
351:
329:
312:
304:
301:
300:
291:
288:
287:
286:
284:
279:
276:
275:
274:
272:
269:
218:
211:
205:
141:
116:conjugate bases
108:buffer solution
39:
28:
23:
22:
15:
12:
11:
5:
2868:
2858:
2857:
2852:
2847:
2833:
2832:
2823:
2797:
2767:
2764:
2763:
2762:
2750:
2749:External links
2747:
2744:
2743:
2712:
2689:
2681:10.17226/26278
2661:
2637:
2612:
2587:
2563:
2554:
2542:
2507:(1): 169–192.
2487:
2472:
2452:
2437:
2417:
2402:
2379:
2356:
2348:10.17226/26278
2331:
2281:
2250:
2200:
2187:
2161:
2134:
2115:(6): 609–623.
2095:
2056:
2049:
2022:
1984:
1962:
1950:
1920:
1905:
1880:
1861:(1–2): 49–63.
1842:
1820:
1792:
1791:
1789:
1786:
1785:
1784:
1779:
1774:
1769:
1764:
1757:
1754:
1739:carbon removal
1736:
1728:
1726:
1723:
1703:
1696:
1681:
1678:
1665:
1661:
1634:
1616:
1604:
1603:
1599:
1558:
1555:
1550:
1549:
1545:
1541:
1537:
1529:
1525:
1524:
1518:
1509:
1502:
1501:
1495:
1483:
1474:
1470:
1466:
1454:
1442:
1433:
1428:
1427:Addition of CO
1424:
1421:
1420:
1419:
1412:
1400:
1389:
1388:
1381:
1372:
1368:
1357:
1350:
1338:
1329:
1325:
1322:
1321:Addition of CO
1319:
1317:
1314:
1309:
1301:
1297:
1286:
1279:
1270:
1263:
1255:
1254:
1250:
1249:
1243:
1234:
1222:
1214:
1213:
1207:
1198:
1186:
1178:
1177:
1173:
1169:
1168:
1164:
1160:
1153:
1141:
1136:
1132:
1128:
1124:
1120:
1116:
1112:
1108:
1104:
1093:
1092:
1088:
1084:
1080:
1076:
1072:
1068:
1064:
1060:
1056:
1040:
1037:
1025:phenolpthalein
997:
989:
938:stoichiometric
925:
922:
906:
894:
882:
881:
866:
860:
856:
852:
849:
845:
842:
839:
833:
829:
825:
820:
816:
811:
808:
805:
801:
798:
794:
791:
788:
782:
777:
773:
769:
766:
762:
759:
756:
750:
746:
741:
738:
735:
729:
725:
721:
717:
714:
711:
705:
701:
697:
693:
690:
687:
681:
676:
672:
668:
665:
660:
656:
651:
648:
645:
639:
636:
631:
627:
623:
619:
616:
613:
610:
604:
601:
596:
592:
588:
585:
581:
578:
575:
569:
565:
561:
557:
554:
551:
545:
540:
536:
532:
529:
526:
523:
519:
516:
513:
507:
504:
499:
495:
491:
487:
484:
481:
478:
472:
467:
463:
459:
456:
452:
448:
446:
444:
441:
438:
430:
427:
424:
421:
413:
410:
407:
405:
403:
384:
368:
356:
348:
347:
336:
328:
325:
322:
319:
311:
308:
289:
277:
268:
265:
239:Also in 1884,
217:
214:
207:
201:
182:oceanographers
139:
26:
9:
6:
4:
3:
2:
2867:
2856:
2853:
2851:
2848:
2846:
2843:
2842:
2840:
2831:
2830:Matlab script
2827:
2824:
2821:
2817:
2813:
2809:
2805:
2801:
2798:
2795:
2791:
2787:
2783:
2779:
2776:
2773:
2772:
2771:
2760:
2756:
2753:
2752:
2740:
2736:
2733:
2729:
2725:
2722:
2716:
2708:
2704:
2700:
2696:
2692:
2686:
2682:
2678:
2674:
2668:
2666:
2651:
2647:
2641:
2627:on 2011-10-16
2626:
2622:
2616:
2602:on 2012-03-14
2601:
2597:
2591:
2577:
2573:
2567:
2558:
2549:
2547:
2538:
2534:
2530:
2526:
2522:
2518:
2514:
2510:
2506:
2502:
2498:
2491:
2483:
2479:
2475:
2469:
2465:
2464:
2456:
2448:
2444:
2440:
2438:0-471-51184-6
2434:
2430:
2429:
2421:
2413:
2409:
2405:
2399:
2395:
2394:
2386:
2384:
2375:
2371:
2367:
2363:
2359:
2353:
2349:
2345:
2341:
2335:
2329:
2324:
2318:
2314:
2309:
2304:
2301:(1): 83–112.
2300:
2296:
2292:
2285:
2278:
2274:
2271:
2267:
2263:
2260:
2254:
2248:
2243:
2237:
2233:
2228:
2223:
2220:(1): 83–112.
2219:
2215:
2211:
2204:
2197:
2191:
2183:
2177:
2164:
2162:9780875532998
2158:
2154:
2150:
2146:
2145:
2138:
2130:
2126:
2122:
2118:
2114:
2110:
2106:
2099:
2091:
2087:
2083:
2079:
2075:
2071:
2067:
2060:
2052:
2050:0-13-351396-3
2046:
2041:
2040:
2031:
2029:
2027:
2018:
2014:
2010:
2006:
2002:
1998:
1991:
1989:
1972:
1966:
1953:
1947:
1943:
1939:
1935:
1931:
1924:
1916:
1912:
1908:
1902:
1898:
1897:
1889:
1887:
1885:
1876:
1872:
1868:
1864:
1860:
1856:
1849:
1847:
1830:
1824:
1810:(Online). n.d
1809:
1808:
1803:
1797:
1793:
1783:
1780:
1778:
1775:
1773:
1770:
1768:
1765:
1763:
1760:
1759:
1750:
1745:
1740:
1733:
1722:
1720:
1716:
1711:
1709:
1700:
1693:
1690:
1687:
1677:
1675:
1671:
1670:Nitrification
1659:
1655:
1651:
1646:
1644:
1640:
1632:
1628:
1623:
1609:
1597:
1596:
1595:
1592:
1590:
1586:
1582:
1576:
1572:
1568:
1564:
1554:
1535:
1534:
1533:
1507:
1506:
1505:
1464:
1463:
1462:
1394:
1393:
1392:
1366:
1365:
1364:
1361:
1313:
1307:
1295:
1290:
1252:
1251:
1216:
1215:
1180:
1179:
1171:
1170:
1147:
1146:
1145:
1100:
1098:
1054:
1053:
1052:
1050:
1046:
1036:
1034:
1030:
1026:
1022:
1018:
1014:
1010:
1007:A variety of
1005:
1003:
995:
987:
983:
982:organic acids
979:
975:
971:
967:
963:
959:
955:
951:
947:
943:
939:
935:
931:
921:
915:
887:
858:
840:
831:
818:
806:
789:
780:
775:
757:
748:
736:
727:
712:
703:
688:
679:
674:
658:
646:
637:
634:
629:
614:
611:
602:
599:
594:
576:
567:
552:
543:
538:
514:
505:
502:
497:
482:
479:
470:
465:
447:
439:
425:
422:
408:
406:
394:
393:
392:
377:
323:
320:
306:
299:
298:
297:
264:
262:
258:
254:
250:
246:
242:
237:
235:
231:
227:
223:
213:
210:
204:
199:
195:
191:
187:
183:
179:
174:
172:
168:
164:
160:
156:
151:
149:
145:
137:
133:
129:
125:
121:
117:
113:
109:
105:
101:
97:
96:acidification
93:
89:
83:
77:
71:
62:
58:
51:
48:
43:
37:
33:
19:
2769:
2758:
2754:DOE (1994) "
2719:IPCC (2022)
2715:
2672:
2653:. Retrieved
2649:
2640:
2629:. Retrieved
2625:the original
2615:
2604:. Retrieved
2600:the original
2590:
2579:. Retrieved
2575:
2566:
2557:
2504:
2500:
2490:
2462:
2455:
2427:
2420:
2392:
2339:
2334:
2298:
2294:
2284:
2257:IPCC, 2021:
2253:
2217:
2213:
2203:
2195:
2190:
2166:, retrieved
2143:
2137:
2112:
2108:
2098:
2073:
2069:
2059:
2038:
2000:
1996:
1975:. Retrieved
1965:
1955:, retrieved
1936:, Elsevier,
1933:
1923:
1895:
1858:
1854:
1833:. Retrieved
1823:
1812:. Retrieved
1805:
1796:
1762:Alkali soils
1743:
1712:
1701:
1694:
1691:
1683:
1647:
1624:
1605:
1593:
1578:
1551:
1526:
1503:
1426:
1390:
1362:
1356:). Adding CO
1327:
1293:
1291:
1256:
1101:
1094:
1042:
1017:Hydrochloric
1006:
972:, dissolved
927:
913:
885:
883:
376:conservative
375:
349:
270:
238:
219:
208:
202:
188:to describe
186:hydrologists
178:limnologists
175:
152:
110:composed of
56:
55:
1585:bicarbonate
1512:+ H ⇌ Ca +
1477:→ 2 Ca + 2
1159:+ H → B(OH)
1045:groundwater
1043:In typical
940:sum of the
212:emissions.
167:bicarbonate
50:climatology
2839:Categories
2786:executable
2655:2023-03-28
2631:2023-03-28
2606:2023-03-28
2581:2023-03-28
2168:2022-12-01
1957:2023-01-09
1835:5 February
1814:2018-09-30
1788:References
1686:weathering
1561:See also:
1172:OH + H → H
1029:methyl red
1013:indicators
976:, and the
954:weathering
198:wastewater
155:freshwater
114:and their
112:weak acids
94:to resist
57:Alkalinity
2804:R package
2707:245089649
2529:1941-1405
2482:246683387
2412:558876135
2374:245089649
2317:225741986
2236:225741986
1915:781078031
1629:, due to
1602:= + 2 +
1581:carbonate
966:phosphate
962:hydroxide
950:carbonate
841:−
807:−
790:−
781:−
758:−
737:−
728:−
680:−
638:−
603:−
568:−
544:−
506:−
471:−
426:∑
423:−
409:∑
324:∑
307:∑
253:hydroxide
249:hydronium
163:carbonate
159:limestone
120:titrating
70:romanized
2812:Mac OS X
2778:Archived
2735:Archived
2724:Archived
2699:35533244
2675:. 2022.
2646:"CARINA"
2537:21141034
2447:31754493
2366:35533244
2342:. 2022.
2273:Archived
2262:Archived
2176:citation
2017:23883395
1802:"alkali"
1756:See also
1192:+ 2 H →
1097:ion pair
1049:seawater
1021:sulfuric
1009:titrants
994:basicity
970:silicate
194:rainfall
124:solution
100:basicity
88:saltwort
2808:Windows
2800:seacarb
2796:script.
2509:Bibcode
2117:Bibcode
2078:Bibcode
1977:6 March
1863:Bibcode
1715:GEOSECS
1344:and/or
1308:of CaCO
986:acetate
984:(e.g.,
974:ammonia
932:to the
314:cations
216:History
148:titrant
84:
76:al-qaly
72::
65:القلوية
18:Alkalic
2794:MATLAB
2775:CO2SYS
2705:
2697:
2687:
2535:
2527:
2480:
2470:
2445:
2435:
2410:
2400:
2372:
2364:
2354:
2315:
2234:
2159:
2047:
2015:
1948:
1913:
1903:
1589:borate
1573:, and
1465:2 CaCO
1406:+ H ⇌
1253:+ H →
1228:+ H →
1031:, and
958:borate
362:, and
331:anions
206:and NO
171:borate
61:Arabic
59:(from
47:GLODAP
32:alkali
2816:Linux
2790:Excel
2703:S2CID
2370:S2CID
2313:S2CID
2268:. In
2232:S2CID
1737:Some
1608:ocean
1418:+ 2 H
1149:B(OH)
942:bases
930:acids
374:are "
136:liter
92:water
2828:, a
2826:CSYS
2820:here
2814:and
2806:for
2802:, a
2695:PMID
2685:ISBN
2533:PMID
2525:ISSN
2478:OCLC
2468:ISBN
2443:OCLC
2433:ISBN
2408:OCLC
2398:ISBN
2362:PMID
2352:ISBN
2182:link
2157:ISBN
2045:ISBN
2013:PMID
1979:2013
1946:ISBN
1911:OCLC
1901:ISBN
1837:2018
1719:WOCE
1672:and
1664:or H
1656:and
1583:and
1536:CaCO
1508:CaCO
1432:CaCO
1375:O ⇌
1296:CaCO
1019:and
1004:).
900:and
245:ions
180:and
165:and
134:per
122:the
82:lit.
2730:in
2677:doi
2517:doi
2344:doi
2303:doi
2222:doi
2149:doi
2125:doi
2086:doi
2074:106
2005:doi
1938:doi
1871:doi
1641:).
1591:.
1544:+ H
1514:HCO
1479:HCO
1469:+ H
1396:HCO
1387:+ H
1377:HCO
1371:+ H
1334:HCO
1218:HPO
1163:+ H
1089:sws
1075:+ 2
1063:+ 2
1047:or
980:of
918:+ 2
890:HCO
380:HCO
273:HCO
196:or
153:In
128:HCl
34:or
2841::
2810:,
2788:,
2757:,"
2701:.
2693:.
2683:.
2664:^
2648:.
2574:.
2545:^
2531:.
2523:.
2515:.
2503:.
2499:.
2476:.
2441:.
2406:.
2382:^
2368:.
2360:.
2350:.
2311:.
2299:45
2297:.
2293:.
2230:.
2218:45
2216:.
2212:.
2178:}}
2174:{{
2123:.
2113:28
2111:.
2107:.
2084:.
2072:.
2068:.
2025:^
2011:.
2001:47
1999:.
1987:^
1944:,
1932:,
1909:.
1883:^
1869:.
1859:40
1857:.
1845:^
1804:.
1612:CO
1569:,
1565:,
1491:SO
1489:+
1473:SO
1450:CO
1438:CO
1408:CO
1367:CO
1346:CO
1306:MW
1294:as
1289:.
1275:CO
1259:CO
1239:PO
1203:PO
1182:PO
1140:CO
1115:CO
1091:−
1087:−
1083:+
1079:+
1071:+
1067:+
1059:=
1027:,
968:,
964:,
960:,
902:CO
364:NO
352:SO
285:CO
263:.
173:.
150:.
104:pH
79:,
67:,
63::
52:).
2822:)
2709:.
2679::
2658:.
2634:.
2609:.
2584:.
2539:.
2519::
2511::
2505:1
2484:.
2449:.
2414:.
2376:.
2346::
2319:.
2305::
2238:.
2224::
2184:)
2151::
2131:.
2127::
2119::
2092:.
2088::
2080::
2053:.
2019:.
2007::
1981:.
1940::
1917:.
1877:.
1873::
1865::
1839:.
1817:.
1751:.
1734:.
1704:T
1697:T
1666:2
1662:2
1635:2
1617:3
1600:T
1598:A
1548:O
1546:2
1542:2
1538:3
1530:2
1519:3
1510:3
1496:4
1484:3
1475:4
1471:2
1467:3
1455:3
1443:3
1434:3
1429:2
1413:3
1401:3
1382:3
1373:2
1369:2
1358:2
1351:3
1339:3
1330:2
1323:2
1310:3
1302:3
1298:3
1287:T
1280:3
1271:2
1264:3
1244:4
1235:2
1230:H
1223:4
1208:4
1199:2
1194:H
1187:4
1176:O
1174:2
1167:O
1165:2
1161:3
1154:4
1142:2
1137:2
1133:2
1129:2
1125:2
1121:2
1117:3
1113:2
1109:2
1105:2
1085:T
1081:T
1077:T
1073:T
1069:T
1065:T
1061:T
1057:T
1055:A
998:2
990:3
907:3
895:3
865:]
859:2
855:O
851:N
848:H
844:[
838:]
832:4
828:O
824:P
819:3
815:H
810:[
804:]
800:F
797:H
793:[
787:]
776:4
772:O
768:S
765:H
761:[
755:]
749:+
745:H
740:[
734:]
724:S
720:H
716:[
713:+
710:]
704:3
700:H
696:N
692:[
689:+
686:]
675:4
671:O
667:i
664:S
659:3
655:H
650:[
647:+
644:]
635:3
630:4
626:O
622:P
618:[
615:2
612:+
609:]
600:2
595:4
591:O
587:P
584:H
580:[
577:+
574:]
564:H
560:O
556:[
553:+
550:]
539:4
535:)
531:H
528:O
525:(
522:B
518:[
515:+
512:]
503:2
498:3
494:O
490:C
486:[
483:2
480:+
477:]
466:3
462:O
458:C
455:H
451:[
440:=
437:)
429:(
420:)
412:(
385:3
369:3
357:4
335:)
327:(
321:=
318:)
310:(
290:3
278:3
209:x
203:x
140:3
38:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.