40:
382:(non-steroidal anti-inflammatory drugs) can be used in both oral or topical formulations for the relief of SVT symptoms. The British Committee for Standards in Haematology guidelines recommend the use of NSAIDs for low-risk SVTs (thrombus <4–5 cm in length, no additional risk factors for thromboembolic events). NSAIDs are used for treatment durations of 8–12 days.
285:
The goal of treatment in SVT is to reduce local inflammation and prevent the SVT from extending from its point of origin. Treatment may entail the use of compression, physical activity, medications, or surgical interventions. The optimal treatment for many SVT sites (i.e. upper limbs, neck, abdominal
127:
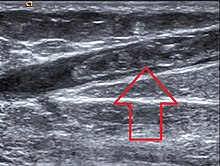
SVT is recognized by the presence of pain, warmth, redness, and tenderness over a superficial vein. The SVT may present as a "cord-like" structure upon palpation. The affected vein may be hard along its entire length. SVTs tend to involve the legs, though they can affect any superficial vein (e.g.
544:
Kalodiki, E; Stvrtinova, V; Allegra, C; Andreozzi, GM; Antignani, P-L; Avram, R; Brkljacic, B; Cadariou, F; Dzsinich, C; Fareed, J; Gaspar, L; Geroulakos, G; Jawien, A; Kozak, M; Lattimer, CR; Minar, E; Partsch, H; Passariello, F; Patel, M; Pecsvarady, Z; Poredos, P; Roztocil, K; Scuderi, A;
451:
SVTs have been historically considered to be benign diseases, for which treatment was limited to conservative measures. However, an increased awareness of the potential risks of SVTs developing into more serious complications has prompted more research into the diagnosis, classification, and
351:
are considered to be equivalent in risk to DVTs. These high risk SVTs are treated identically with therapeutic anticoagulation. Anticoagulation is also used for intermediate risk SVTs that are greater than 3 cm from the saphenofemoral junction or are greater than 4–5 cm in length.
434:
SVT is often a mild, self-resolving medical condition. The inflammatory reaction may last up to 2–3 weeks, with possible recanalization of the thrombosed vein occurring in 6–8 weeks. The superficial vein may continue to be hyperpigmented for several months following the initial event.
118:
SVT has risk factors similar to those for other thrombotic conditions and can arise from a variety of causes. Diagnosis is often based on symptoms. There are multiple possible treatments, with the goal of providing symptomatic relief and preventing complications.
107:. However, SVT can lead to serious complications (as well as signal other serious problems, such as genetic mutations that increase one's risk for clotting), and is therefore no longer regarded as a benign condition. If the blood clot is too near the
237:
The mechanism for the development of an SVT depends upon the specific etiology of the SVT. For example, varicose veins and prolonged bed rest both may induce SVTs due to slowing the flow of blood through superficial veins.
306:
Inactivity is contraindicated in the aftermath of an SVT. Uninterrupted periods of sitting or standing may cause the SVT to elongate from its point of origin, increasing the risk for complications and clinical worsening.
147:
SVTs can recur after they resolve, which is termed "migratory thrombophlebitis." Migratory thrombophlebitis is a complication that may be due to more serious disorders, such as cancer and other hypercoagulable states.
250:. An ultrasound can be useful in situations in which an SVT occurs above the knee and is not associated with a varicose vein, because ultrasounds can detect more serious clots like DVTs. The diagnostic utility of
423:
144:(DVT) was found in 24.6% of people with SVTs. However, because superficial veins lack muscular support, any clots that form are far less likely to be squeezed by muscle contraction, dislodged, and induce a PE.
140:(PE). This is because lower limb SVTs can migrate from superficial veins into deeper veins. In a French population, the percent of people with SVTs that also suffered from PEs was 4.7%. In the same population,
460:
A Cochrane review recommends that future research investigate the utility of oral, topical, and surgical treatments for preventing the progression of SVTs and the development of thromboembolic complications.
406:
Surgical interventions are used for both symptomatic relief of the SVT as well as for preventing the development of more serious complications (e.g. pulmonary embolism). Surgical interventions include
901:
418:. Because of the risk of symptomatic pulmonary embolism with surgery itself, surgical interventions are not recommended for the treatment of lower limb SVTs by the 2012
266:(VV) or non-varicose (NV) associated. NV-SVTs are more likely to be associated with genetic procoagulable states compared to VV-SVTs. SVTs can also be classified by
270:. That is, primary SVTs are characterized by inflammation that is localized to the veins. Secondary SVTs are characterized by systemic inflammatory processes.
163:
Many of the risk factors that are associated with SVT are also associated with other thrombotic conditions (e.g. DVT). These risk factors include age,
592:
911:
17:
160:, though most people with varicose veins do not develop SVTs. SVTs of the arms are often due to the placement of intravenous catheters.
803:
Di Nisio, Marcello; Wichers, Iris M.; Middeldorp, Saskia (2013-04-30). "Treatment for superficial thrombophlebitis of the leg".
298:
have all be used in the treatment of SVTs. The benefit of compression stockings is unclear, though they are frequently used.
419:
246:
SVTs may be diagnosed based upon clinical criteria by a healthcare professional. A more specific evaluation can be made by
294:
Multiple compression bandages exist. Fixed compression bandages, adhesive short stretch bandages, and graduated elastic
136:
SVT in the lower extremities can lead to a dangerous complication in which the clot travels to the lungs, called
205:
due to genetic conditions that increase the risk of clotting may contribute to the development of SVT, such as
368:
222:
180:
921:
192:
593:"Superficial Venous Thrombosis – Heart and Blood Vessel Disorders – Merck Manuals Consumer Version"
545:
Sparovec, M; Szostek, M; Skorski, M (2012). "Superficial vein thrombosis: a consensus statement".
896:
547:
391:
348:
274:
108:
188:
39:
356:
226:
202:
706:
344:
295:
141:
100:
47:
398:
are used for the treatment of SVTs in the setting of vasculitic and autoimmune syndromes.
8:
906:
407:
184:
916:
868:
843:
688:
478:
210:
176:
137:
112:
873:
824:
816:
737:
680:
556:
778:
762:"Tromboflebite superficial: epidemiologia, fisiopatologia, diagnóstico e tratamento"
761:
863:
855:
812:
808:
783:
773:
692:
670:
360:
76:
68:
395:
332:
267:
206:
168:
760:
Sobreira, Marcone Lima; Yoshida, Winston
Bonneti; Lastória, Sidnei (June 2008).
411:
316:
157:
890:
820:
367:
2.5 mg daily for 45 days or the use of intermediate to therapeutic dose
263:
859:
877:
828:
741:
684:
560:
477:
Migratory thrombophlebitis (recurrent SVT) and cancer are the hallmarks of
415:
364:
80:
426:
guidelines. The use of surgery for the treatment of SVT is controversial.
328:
196:
443:
In a French population, SVT occurred in 0.64 per 1000 persons per year.
788:
247:
84:
64:
675:
658:
218:
214:
172:
104:
92:
88:
543:
363:
for 3 months. Anticoagulation for intermediate risk SVTs includes
195:
diseases). Other risk factors include immobilization (stasis) and
324:
251:
254:
testing in the setting of SVTs has yet to be fully established.
732:
Foris, LA; Bhimji, SS (June 2017). "Thrombophlebitis, Septic".
379:
320:
164:
842:
Streiff MB, Bockenstedt PL, Cataland SR, et al. (2011).
96:
286:
and thoracic walls, and the penis) has not been determined.
277:, which are SVTs that occur in the setting of an infection.
72:
841:
91:. SVT itself has limited significance (in terms of direct
802:
759:
355:
Anticoagulation for high risk SVTs includes the use of
539:
537:
535:
533:
531:
529:
527:
525:
523:
521:
519:
517:
755:
753:
751:
515:
513:
511:
509:
507:
505:
503:
501:
499:
497:
902:
Diseases of veins, lymphatic vessels and lymph nodes
707:"Hormonal Birth Control and Blood Clot Risk – NWHN"
748:
494:
315:Medications used for the treatment of SVT include
83:around a thrombosed vein, presenting as a painful
888:
725:
115:, a potentially life-threatening complication.
75:near the surface of the body. Usually there is
424:British Committee for Standards in Haematology
103:(DVT), which occurs deeper in the body at the
652:
650:
648:
646:
644:
642:
640:
638:
636:
634:
410:of the saphenofemoral junction, ligation and
632:
630:
628:
626:
624:
622:
620:
618:
616:
614:
805:The Cochrane Database of Systematic Reviews
731:
659:"Management of superficial vein thrombosis"
38:
867:
787:
777:
674:
611:
390:Antibiotics are used in the treatment of
587:
585:
583:
581:
579:
577:
575:
573:
699:
14:
889:
835:
912:Vascular-related cutaneous conditions
663:Journal of Thrombosis and Haemostasis
656:
570:
122:
420:American College of Chest Physicians
301:
179:medications (containing estrogen),
24:
156:SVTs of the legs are often due to
25:
933:
414:of the affected veins, and local
361:novel oral anticoagulants (NOACs)
338:
262:SVTs can be classified as either
257:
131:
844:"Venous thromboembolic disease"
779:10.1590/S1677-54492008000200007
438:
813:10.1002/14651858.CD004982.pub5
796:
599:. Merck Sharp & Dohme Corp
597:Merck Manuals Consumer Version
471:
310:
289:
183:, recent surgery, and certain
87:(thickening of the skin) with
13:
1:
488:
18:Superficial venous thromboses
464:
429:
369:low molecular weight heparin
280:
241:
232:
7:
455:
343:SVTs that occur within the
181:hormone replacement therapy
57:Superficial vein thrombosis
33:Superficial vein thrombosis
10:
938:
766:Jornal Vascular Brasileiro
446:
401:
111:there is a higher risk of
374:
151:
46:
37:
32:
422:guidelines and the 2012
385:
347:within 3 cm of the
105:deep venous system level
860:10.6004/jnccn.2011.0062
657:Cosmi, B. (July 2015).
566:(subscription required)
548:International Angiology
349:saphenofemoral junction
275:septic thrombophlebitis
273:A subclass of SVTs are
109:saphenofemoral junction
848:J Natl Compr Canc Netw
203:Hypercoagulable states
357:vitamin K antagonists
296:compression stockings
227:factor XII deficiency
99:) when compared to a
81:inflammatory reaction
345:great saphenous vein
142:deep vein thrombosis
128:those in the arms).
101:deep vein thrombosis
48:Great saphenous vein
452:treatment of SVTs.
185:autoimmune diseases
713:. 21 February 2017
479:Trousseau syndrome
211:prothrombin 20210A
177:oral contraceptive
138:pulmonary embolism
123:Signs and symptoms
113:pulmonary embolism
922:Vascular diseases
676:10.1111/jth.12986
302:Physical activity
54:
53:
27:Medical condition
16:(Redirected from
929:
882:
881:
871:
839:
833:
832:
800:
794:
793:
791:
781:
757:
746:
745:
729:
723:
722:
720:
718:
703:
697:
696:
678:
669:(7): 1175–1183.
654:
609:
608:
606:
604:
589:
568:
567:
564:
541:
482:
475:
223:antithrombin III
77:thrombophlebitis
69:superficial vein
42:
30:
29:
21:
937:
936:
932:
931:
930:
928:
927:
926:
887:
886:
885:
840:
836:
807:(4): CD004982.
801:
797:
758:
749:
730:
726:
716:
714:
705:
704:
700:
655:
612:
602:
600:
591:
590:
571:
565:
542:
495:
491:
486:
485:
476:
472:
467:
458:
449:
441:
432:
404:
396:Corticosteroids
388:
377:
371:for 4–6 weeks.
341:
333:corticosteroids
313:
304:
292:
283:
268:pathophysiology
260:
244:
235:
207:factor V Leiden
169:thromboembolism
154:
134:
125:
28:
23:
22:
15:
12:
11:
5:
935:
925:
924:
919:
914:
909:
904:
899:
897:Coagulopathies
884:
883:
834:
795:
772:(2): 131–143.
747:
724:
698:
610:
569:
555:(3): 203–216.
492:
490:
487:
484:
483:
469:
468:
466:
463:
457:
454:
448:
445:
440:
437:
431:
428:
403:
400:
387:
384:
376:
373:
340:
339:Anticoagulants
337:
317:anticoagulants
312:
309:
303:
300:
291:
288:
282:
279:
259:
258:Classification
256:
243:
240:
234:
231:
213:mutation, and
158:varicose veins
153:
150:
133:
130:
124:
121:
79:, which is an
52:
51:
44:
43:
35:
34:
26:
9:
6:
4:
3:
2:
934:
923:
920:
918:
915:
913:
910:
908:
905:
903:
900:
898:
895:
894:
892:
879:
875:
870:
865:
861:
857:
854:(7): 714–77.
853:
849:
845:
838:
830:
826:
822:
818:
814:
810:
806:
799:
790:
785:
780:
775:
771:
767:
763:
756:
754:
752:
743:
739:
735:
728:
712:
708:
702:
694:
690:
686:
682:
677:
672:
668:
664:
660:
653:
651:
649:
647:
645:
643:
641:
639:
637:
635:
633:
631:
629:
627:
625:
623:
621:
619:
617:
615:
598:
594:
588:
586:
584:
582:
580:
578:
576:
574:
562:
558:
554:
550:
549:
540:
538:
536:
534:
532:
530:
528:
526:
524:
522:
520:
518:
516:
514:
512:
510:
508:
506:
504:
502:
500:
498:
493:
480:
474:
470:
462:
453:
444:
436:
427:
425:
421:
417:
413:
409:
399:
397:
393:
383:
381:
372:
370:
366:
362:
358:
353:
350:
346:
336:
334:
330:
326:
322:
318:
308:
299:
297:
287:
278:
276:
271:
269:
265:
264:varicose vein
255:
253:
249:
239:
230:
228:
224:
220:
216:
212:
208:
204:
200:
198:
194:
190:
186:
182:
178:
174:
170:
167:, history of
166:
161:
159:
149:
145:
143:
139:
132:Complications
129:
120:
116:
114:
110:
106:
102:
98:
94:
90:
86:
82:
78:
74:
70:
66:
62:
58:
49:
45:
41:
36:
31:
19:
851:
847:
837:
804:
798:
769:
765:
733:
727:
715:. Retrieved
710:
701:
666:
662:
601:. Retrieved
596:
552:
546:
473:
459:
450:
442:
439:Epidemiology
433:
416:thrombectomy
405:
389:
378:
365:fondaparinux
354:
342:
314:
305:
293:
284:
272:
261:
245:
236:
201:
187:(especially
162:
155:
146:
135:
126:
117:
67:formed in a
60:
56:
55:
789:11449/26900
329:antibiotics
311:Medications
290:Compression
197:laparoscopy
907:Hematology
891:Categories
734:StatPearls
717:29 January
603:7 February
489:References
392:septic SVT
248:ultrasound
85:induration
65:blood clot
50:thrombosis
917:Angiology
821:1469-493X
465:Footnotes
430:Prognosis
412:stripping
281:Treatment
242:Diagnosis
233:Mechanism
215:protein C
193:Buerger's
175:, use of
173:pregnancy
97:mortality
93:morbidity
878:21715723
829:23633322
742:28613482
685:25903684
561:22634973
456:Research
408:ligation
323:(except
189:Behçet's
869:3551573
693:5276848
447:History
402:Surgery
325:aspirin
252:D-dimer
89:redness
63:) is a
876:
866:
827:
819:
740:
691:
683:
559:
380:NSAIDs
375:NSAIDs
331:, and
321:NSAIDs
221:, and
165:cancer
152:Causes
689:S2CID
386:Other
874:PMID
825:PMID
817:ISSN
738:PMID
719:2018
711:NWHN
681:PMID
605:2018
557:PMID
225:and
191:and
95:and
73:vein
71:, a
864:PMC
856:doi
809:doi
784:hdl
774:doi
671:doi
359:or
327:),
61:SVT
893::
872:.
862:.
850:.
846:.
823:.
815:.
782:.
768:.
764:.
750:^
736:.
709:.
687:.
679:.
667:13
665:.
661:.
613:^
595:.
572:^
553:31
551:.
496:^
394:.
335:.
319:,
229:.
217:,
209:,
199:.
171:,
880:.
858::
852:9
831:.
811::
792:.
786::
776::
770:7
744:.
721:.
695:.
673::
607:.
563:.
481:.
219:S
59:(
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.