59:
62:
61:
66:
65:
60:
67:
64:
1305:
1911:
1898:
1954:
476:
653:
63:
1832:
1048:
724:
84:
2240:
1519:
1563:
40:
3310:
Oeters, Franz; Ottow, Manfred; Meiler, Heinrich; Lüngen, Hans Bodo; Koltermann, Manfred; Buhr, Andreas; Yagi, Jun-Ichiro; Formanek, Lothar; Rose, Fritz; Flickenschild, Jürgen; Hauk, Rolf; Steffen, Rolf; Skroch, Reiner; Mayer-Schwinning, Gernot; Bünnagel, Heinz-Lothar; Hoff, Hans-Georg (2006). "Iron".
329:
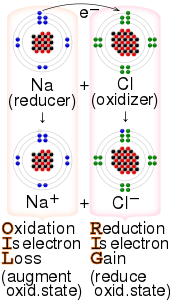
that accomplished chemical reactions similar to those of oxygen. Ultimately, the meaning was generalized to include all processes involving the loss of electrons or the increase in the oxidation state of a chemical species. Substances that have the ability to oxidize other substances (cause them to
525:
demonstrated that this loss of weight was due to the loss of oxygen as a gas. Later, scientists realized that the metal atom gains electrons in this process. The meaning of reduction then became generalized to include all processes involving a gain of electrons. Reducing equivalent refers to
1109:
2312:) relative to the standard hydrogen electrode) or pe (analogous to pH as -log electron activity), is a master variable, along with pH, that controls and is governed by chemical reactions and biological processes. Early theoretical research with applications to flooded soils and
2350:
The key terms involved in redox can be confusing. For example, a reagent that is oxidized loses electrons; however, that reagent is referred to as the reducing agent. Likewise, a reagent that is reduced gains electrons and is referred to as the oxidizing agent. These
694:
The mechanisms of atom-transfer reactions are highly variable because many kinds of atoms can be transferred. Such reactions can also be quite complex, involving many steps. The mechanisms of electron-transfer reactions occur by two distinct pathways,
2316:
production was seminal for subsequent work on thermodynamic aspects of redox and plant root growth in soils. Later work built on this foundation, and expanded it for understanding redox reactions related to heavy metal oxidation state changes,
2139:. In these reactions, an electron detaches from a molecule and then re-attaches almost instantaneously. Free radicals are part of redox molecules and can become harmful to the human body if they do not reattach to the redox molecule or an
238:, and simultaneously, the oxidation state of oxygen decreases as it accepts electrons released by the iron. Although oxidation reactions are commonly associated with forming oxides, other chemical species can serve the same function. In
3090:
The homogeneous proton-transfer reactions described are similar to homogeneous electron-transfer reactions in that the overall electron-transfer reaction can be decomposed into one electronation reaction and one deelectronation
219:– Only one (usually) electron flows from the atom, ion, or molecule being oxidized to the atom, ion, or molecule that is reduced. This type of redox reaction is often discussed in terms of redox couples and electrode potentials.
994:
The oxidation potential is a measure of the tendency of the reducing agent to be oxidized but does not represent the physical potential at an electrode. With this notation, the cell voltage equation is written with a plus sign
278:
gains electrons and is reduced. The pair of an oxidizing and reducing agent that is involved in a particular reaction is called a redox pair. A redox couple is a reducing species and its corresponding oxidizing form, e.g.,
2226:
and regenerate the unchanged parent compound. The net reaction is the oxidation of the flavoenzyme's coenzymes and the reduction of molecular oxygen to form superoxide. This catalytic behavior has been described as a
3123:
IUPAC. Compendium of
Chemical Terminology, 2nd ed. (the "Gold Book"). Compiled by A. D. McNaught and A. Wilkinson. Blackwell Scientific Publications, Oxford (1997). Online version (2019-) created by S. J. Chalk.
3102:
IUPAC. Compendium of
Chemical Terminology, 2nd ed. (the "Gold Book"). Compiled by A. D. McNaught and A. Wilkinson. Blackwell Scientific Publications, Oxford (1997). Online version (2019-) created by S. J. Chalk.
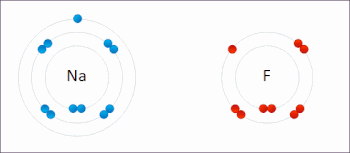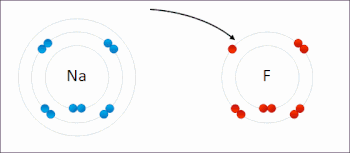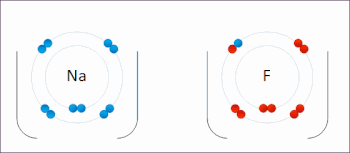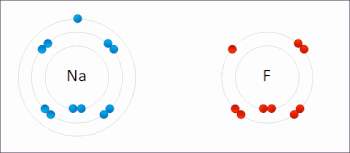
269:
The processes of oxidation and reduction occur simultaneously and cannot occur independently. In redox processes, the reductant transfers electrons to the oxidant. Thus, in the reaction, the reductant or
208:
or an increase in the oxidation state, while reduction is the gain of electrons or a decrease in the oxidation state. The oxidation and reduction processes occur simultaneously in the chemical reaction.
2178:), whose interconversion is dependent on these ratios. Redox mechanisms also control some cellular processes. Redox proteins and their genes must be co-located for redox regulation according to the
1362:
In the above reaction, zinc metal displaces the copper(II) ion from the copper sulfate solution, thus liberating free copper metal. The reaction is spontaneous and releases 213 kJ per 65 g of zinc.
480:
897:), or potential when the half-reaction takes place at a cathode. The reduction potential is a measure of the tendency of the oxidizing agent to be reduced. Its value is zero for H + e →
2103:
into molecular oxygen. The reverse reaction, respiration, oxidizes sugars to produce carbon dioxide and water. As intermediate steps, the reduced carbon compounds are used to reduce
505:, reductants, or reducers. The reductant transfers electrons to another substance and is thus itself oxidized. Because it donates electrons, the reducing agent is also called an
334:, oxidants, or oxidizers. The oxidant removes electrons from another substance, and is thus itself reduced. Because it "accepts" electrons, the oxidizing agent is also called an
580:
2271:). These oxides must be reduced to obtain the corresponding metals, often achieved by heating these oxides with carbon or carbon monoxide as reducing agents.
2275:
are the reactors where iron oxides and coke (a form of carbon) are combined to produce molten iron. The main chemical reaction producing the molten iron is:
1142:
This spontaneous reaction releases 542 kJ per 2 g of hydrogen because the H-F bond is much stronger than the F-F bond. This reaction can be analyzed as two
2430:
which release stored electricity, and can be recharged with electricity. PANIC does not apply to cells that can be recharged with redox materials. These
3313:
3222:
3193:
2720:
501:
Substances that have the ability to reduce other substances (cause them to gain electrons) are said to be reductive or reducing and are known as
2967:
3639:
2417:"LEORA says GEROA" — the loss of electrons is called oxidation (reducing agent); the gain of electrons is called reduction (oxidizing agent).
2787:
Pingarrón, José M.; Labuda, Ján; Barek, Jiří; Brett, Christopher M. A.; Camões, Maria
Filomena; Fojta, Miroslav; Hibbert, D. Brynn (2020).
2151:
3398:
James, Bruce R.; Brose, Dominic A. (2012). "Oxidation-reduction phenomena". In Huang, Pan Ming; Li, Yuncong; Sumner, Malcolm E. (eds.).
3147:"Teaching the fundamentals of electron transfer reactions in mitochondria and the production and detection of reactive oxygen species"
2638:
624:
proposed the words electronation and de-electronation to describe reduction and oxidation processes, respectively, when they occur at
2652:
1657:
Here the overall equation involves adding the reduction equation to twice the oxidation equation, so that the electrons cancel:
2438:, produce electricity from internal redox reactions. Here, the positive electrode is the cathode and the negative is the anode.
3603:
3576:
3539:
3507:
3473:
3443:
3411:
3363:
3034:
3001:
2836:
2051:
and cellular respiration are complementary, but photosynthesis is not the reverse of the redox reaction in cell respiration:
597:, reduce by atom transfer: they transfer the equivalent of hydride or H. These reagents are widely used in the reduction of
2748:
2420:"RED CAT" and "AN OX", or "AnOx RedCat" ("an ox-red cat") — reduction occurs at the cathode and the anode is for oxidation
1755:. The sacrificial metal, instead of the protected metal, then corrodes. A common application of cathodic protection is in
468:
Oxidizers are oxidants, but the term is mainly reserved for sources of oxygen, particularly in the context of explosions.
266:
Oxidation is a process in which a substance loses electrons. Reduction is a process in which a substance gains electrons.
3851:
3728:
1693:
ion with sulfur in oxidation state +2 can react in the presence of acid to form elemental sulfur (oxidation state 0) and
3685:
3330:
2214:
that contain one more electron than their parent compounds. In general, the electron donor is any of a wide variety of
17:
3992:
3632:
3286:
3252:
3129:
3108:
3076:
2943:
2918:
2893:
2700:
2104:
2044:
1879:
1095:
807:
176:
136:
2423:"RED CAT gains what AN OX loses" – reduction at the cathode gains (electrons) what anode oxidation loses (electrons)
1861:
1077:
788:
760:
1785:
often contain metals in oxidized states, such as oxides or sulfides, from which the pure metals are extracted by
325:
Oxidation originally implied a reaction with oxygen to form an oxide. Later, the term was expanded to encompass
3900:
3895:
3705:
1857:
1073:
745:
700:
696:
2244:
513:
with electron acceptors. The word reduction originally referred to the loss in weight upon heating a metallic
4065:
4060:
2980:
1777:
Redox reactions are the foundation of electrochemical cells, which can generate electrical energy or support
767:
4101:
3625:
2852:
4091:
2772:
2474:
2345:
861:
2716:
4030:
3720:
1763:
1459:
774:
1910:
3757:
3657:
2554:
1551:
591:
3987:
3266:
2539:
2504:
1842:
1058:
2091:
is frequently stored and released using redox reactions. Photosynthesis involves the reduction of
756:
4086:
4035:
3836:
1846:
1587:, is a well-known example of electrochemical corrosion: it forms as a result of the oxidation of
1062:
734:
510:
1957:
1897:
3790:
2544:
2484:
2112:
1969:
1853:
1069:
741:
247:
76:
31:
3529:
3497:
3066:
3026:
3019:
1579:
refers to the electrochemical oxidation of metals in reaction with an oxidant such as oxygen.
4020:
3952:
3810:
3800:
3433:
2449:
483:
223:
3559:
Schüring, J.; Schulz, H. D.; Fischer, W. R.; Böttcher, J.; Duijnisveld, W. H., eds. (1999).
664:
4015:
3743:
2630:
2171:
1975:
1936:
1744:
1309:
845:
340:
8:
4025:
3957:
3942:
3885:
2594:
2589:
2529:
2499:
2211:
1968:
processes involve redox reactions. Before some of these processes can begin iron must be
1736:
1728:
Thus one sulfur atom is reduced from +2 to 0, while the other is oxidized from +2 to +4.
1490:
821:
338:. Oxidants are usually chemical substances with elements in high oxidation states (e.g.,
885:
The electrode potential of each half-reaction is also known as its reduction potential (
4050:
3820:
3649:
3292:
3171:
3146:
2961:
2549:
2524:
2123:
1689:
reaction is one in which a single substance is both oxidized and reduced. For example,
1686:
1341:
691:. Electron transfer reactions are generally fast, occurring within the time of mixing.
584:
414:
326:
79:(strong oxidizing agent), a violent redox reaction accompanied by self-ignition starts.
3355:
2333:, and various methodological approaches for characterizing the redox status of soils.
4045:
4040:
4002:
3947:
3866:
3846:
3782:
3599:
3572:
3535:
3503:
3469:
3439:
3407:
3359:
3326:
3296:
3282:
3176:
3125:
3104:
3072:
3030:
2997:
2949:
2939:
2914:
2889:
2832:
2696:
2660:
2559:
2479:
2469:
2427:
2301:
2088:
1748:
1623:
1486:
1332:
atom in a compound or solution is replaced by an atom of another metal. For example,
1270:
707:
522:
427:
377:
335:
216:
193:
3232:
3203:
2162:. The redox state is reflected in the balance of several sets of metabolites (e.g.,
1747:. A simple method of protection connects protected metal to a more easily corroded "
1304:
3977:
3926:
3880:
3591:
3564:
3351:
3318:
3274:
3236:
3227:
3207:
3198:
3166:
3158:
2800:
2509:
2489:
2454:
2330:
2100:
1778:
1759:
steel, in which a sacrificial zinc coating on steel parts protects them from rust.
915:) and negative for oxidizing agents that are weaker than H (e.g., −0.763V for Zn).
781:
618:
527:
263:
of the words "reduction" and "oxidation." The term "redox" was first used in 1928.
147:
107:
3322:
911:
by definition, positive for oxidizing agents stronger than H (e.g., +2.866 V for F
4055:
3967:
3916:
3568:
2789:"Terminology of electrochemical methods of analysis (IUPAC Recommendations 2019)"
2604:
2574:
2534:
2305:
2179:
2159:
2108:
1960:
is an example of a redox reaction that takes place in most fruits and vegetables.
1940:
1739:
is a technique used to control the corrosion of a metal surface by making it the
1592:
1531:
1523:
1502:
1471:
1414:
960:
However, the potential of the reaction at the anode is sometimes expressed as an
546:
331:
320:
275:
197:
51:
3162:
1801:
1762:
Oxidation is used in a wide variety of industries, such as in the production of
1320:
solution connected with a wire and a porous disk to a copper electrode in a CuSO
4096:
3762:
3751:
3133:
3112:
2584:
2569:
2459:
2155:
2092:
2048:
1995:
1926:
1794:
1790:
1694:
1467:
841:
539:
506:
502:
496:
271:
3278:
918:
For a redox reaction that takes place in a cell, the potential difference is:
577:, are good reducing agents. These metals donate electrons relatively readily.
4080:
4010:
3982:
3890:
3841:
3815:
3231:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
3202:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
2953:
2579:
2519:
2514:
2431:
2272:
2251:
Minerals are generally oxidized derivatives of metals. Iron is mined as its
2187:
2136:
1922:
1808:
1756:
1376:
1313:
1143:
853:
710:
in water allows calculation of the thermodynamic aspects of redox reactions.
633:
307:
239:
3617:
3240:
3211:
1953:
1793:
uses redox reactions to coat objects with a thin layer of a material, as in
3962:
3768:
3675:
3665:
3379:
Bartlett, Richmond J.; James, Bruce R. (1991). "Redox chemistry of soils".
3180:
2609:
2494:
2464:
2322:
2228:
2175:
2128:
2116:
1160:
621:
535:
363:
311:
because two half-reactions always occur together to form a whole reaction.
3586:
Tratnyek, Paul G.; Grundl, Timothy J.; Haderlein, Stefan B., eds. (2011).
2981:
https://chemed.chem.purdue.edu/genchem/topicreview/bp/ch19/oxred_3.php#top
2805:
2788:
3921:
3856:
3595:
2767:
2564:
2318:
2215:
2203:
2191:
2163:
2147:
2140:
2132:
2043:
The process of cell respiration also depends heavily on the reduction of
1905:
1892:
1812:
1789:
at high temperatures in the presence of a reducing agent. The process of
1771:
1690:
1455:
629:
518:
475:
469:
260:
234:, the oxidation state of iron atoms increases as the iron converts to an
652:
330:
lose electrons) are said to be oxidative or oxidizing, and are known as
3267:"Titles of Volumes 1–44 in the Metal Ions in Biological Systems Series"
2599:
2313:
2304:
reactions are central to myriad processes and properties in soils, and
2223:
1797:
1584:
1451:
748: in this section. Unsourced material may be challenged and removed.
713:
391:
2827:
Petrucci, Ralph H.; Harwood, William S.; Herring, F. Geoffrey (2017).
2691:
Petrucci, Ralph H.; Harwood, William S.; Herring, F. Geoffrey (2002).
3972:
3403:
2435:
2352:
2252:
2115:(ATP) and is maintained by the reduction of oxygen. In animal cells,
1944:
1930:
1576:
625:
574:
562:
3459:
3457:
3455:
2222:. Once formed, these anion free radicals reduce molecular oxygen to
1831:
1047:
723:
636:. They have not been widely adopted by chemists worldwide, although
605:. A related method of reduction involves the use of hydrogen gas (H
3071:. Vol. 1. Springer Science & Business Media. p. 494.
2884:
Whitten, Kenneth W.; Gailey, Kenneth D.; Davis, Raymond E. (1992).
2219:
2167:
1786:
1506:
1494:
1410:
1185:
1172:
1164:
1121:
1117:
602:
598:
538:. A reducing equivalent can be an electron or a hydrogen atom as a
531:
445:
438:
205:
201:
72:
47:
2426:"PANIC" – Positive Anode and Negative is Cathode. This applies to
521:
to extract the metal. In other words, ore was "reduced" to metal.
3452:
2360:
2326:
2309:
2047:
to NADH and the reverse reaction (the oxidation of NADH to NAD).
1979:
1965:
1815:
1804:
1767:
1740:
1406:
1108:
849:
837:
554:
452:
227:
3271:
Metals, Microbes, and
Minerals - the Biogeochemical Side of Life
2355:
are commonly used by students to help memorise the terminology:
465:) that can gain extra electrons by oxidizing another substance.
83:
27:
Chemical reaction in which oxidation states of atoms are changed
2264:
2207:
2065:
2002:
1567:
1498:
1479:
1475:
1333:
1316:
pictured. The battery is made out of a zinc electrode in a ZnSO
1147:
558:
459:
431:
305:.The oxidation alone and the reduction alone are each called a
243:
43:
3499:
Descriptive
Inorganic, Coordination, and Solid-State Chemistry
2653:"redox – definition of redox in English | Oxford Dictionaries"
2239:
2146:
The term redox state is often used to describe the balance of
1192:
The half-reactions are combined so that the electrons cancel:
156:
3558:
2860:
2263:). Titanium is mined as its dioxide, usually in the form of
2096:
2006:
1752:
1518:
1463:
1329:
857:
640:
has recognized the terms electronation and de-electronation.
637:
550:
235:
2996:. Washington, D.C.: American Chemical Society. p. 429.
2321:
and morphology, organic compound degradation and formation,
1124:, hydrogen is being oxidized and fluorine is being reduced:
2107:(NAD) to NADH, which then contributes to the creation of a
1627:
1588:
1580:
1527:
1337:
688:
684:
570:
566:
294:
281:
231:
116:
162:
122:
3695:
3464:
Phillips, John; Strozak, Victor; Wistrom, Cheryl (2000).
2888:(4th ed.). Saunders College Publishin. p. 147.
2786:
2183:
1782:
683:
Redox reactions can occur slowly, as in the formation of
514:
3025:. Washington, D.C.: American Chemical Society. pp.
3585:
3309:
1562:
54:. The sodium atom is oxidized, and fluorine is reduced.
3463:
3350:. Advances in Agronomy. Vol. 24. pp. 29–96.
2826:
2690:
1485:
The stepwise oxidation of a hydrocarbon by oxygen, in
39:
3438:. National Science Teachers Association. p. 82.
2829:
General
Chemistry: Principles and Modern applications
177:
168:
165:
137:
128:
125:
2131:
reactions are redox reactions that occur as part of
714:
Standard electrode potentials (reduction potentials)
612:
274:
loses electrons and is oxidized, and the oxidant or
159:
153:
119:
113:
3400:
Handbook of soil sciences: properties and processes
2934:Lehninger AL, Nelson DL, Cox MM (January 1, 2017).
150:
110:
3018:
2933:
2883:
2751:. In K. Lee Lerner; Brenda Wilmoth Lerner (eds.).
2308:, quantified as Eh (platinum electrode potential (
643:
2755:(5th ed.). Farmington Hills, MI: Gale Group.
1558:), form when oxygen combines with other elements.
836:), which is equal to the potential difference or
4078:
3065:Bockris, John O'M.; Reddy, Amulya K.N. (2013) .
3050:Bockris, John O'M.; Reddy, Amulya K. N. (1970).
2009:. The summary equation for cell respiration is:
3561:Redox: Fundamentals, Processes and Applications
91:reaction between sodium and chlorine, with the
3527:
3502:. Brooks/Cole, Cengage Learning. p. 330.
3314:Ullmann's Encyclopedia of Industrial Chemistry
2927:
2822:
2820:
2818:
2816:
2686:
2684:
2682:
2680:
2678:
1731:
1146:. The oxidation reaction converts hydrogen to
1037:
3647:
3633:
3378:
2234:
1821:
1478:. Complete oxidation of materials containing
3563:. Heidelberg: Springer-Verlag. p. 246.
3427:
3425:
3423:
3345:
3134:https://goldbook.iupac.org/terms/view/O04362
3113:https://goldbook.iupac.org/terms/view/R05222
3064:
3049:
2550:Oxidative addition and reductive elimination
1595:, formed in the following chemical reaction:
75:(mild reducing agent) are added to powdered
2813:
2675:
2296:
1860:. Unsourced material may be challenged and
1076:. Unsourced material may be challenged and
222:Atom transfer – An atom transfers from one
3640:
3626:
3523:
3521:
3519:
3491:
3489:
3487:
3485:
3397:
2966:: CS1 maint: location missing publisher (
2879:
2877:
1622:The oxidation of iron(II) to iron(III) by
545:Reductants in chemistry are very diverse.
534:in redox reactions. The term is common in
212:There are two classes of redox reactions:
3431:
3420:
3273:. De Gruyter. 2021. pp. xxiii–xxiv.
3170:
2804:
1880:Learn how and when to remove this message
1365:The ionic equation for this reaction is:
1269:The protons and fluoride combine to form
1096:Learn how and when to remove this message
808:Learn how and when to remove this message
3590:. ACS Symposium Series. Vol. 1071.
3528:Zumdahl, Steven; Zumdahl, Susan (2009).
3402:(second ed.). Boca Raton, Florida:
3144:
3016:
2991:
2746:
2238:
1952:
1561:
1517:
1513:
1470:, some partially oxidized forms such as
1379:, it is seen that the zinc is oxidized:
1308:A redox reaction is the force behind an
1303:
1107:
687:, or rapidly, as in the case of burning
474:
82:
57:
38:
3516:
3495:
3482:
2874:
1489:, produces water and, successively: an
14:
4079:
3666:Unimolecular nucleophilic substitution
2742:
2740:
2738:
2641:from the original on November 3, 2022.
3676:Bimolecular nucleophilic substitution
3621:
1680:
1299:
530:which transfer the equivalent of one
3468:. Glencoe McGraw-Hill. p. 558.
3466:Chemistry: Concepts and Applications
2936:Lehninger Principles of Biochemistry
2908:
1978:, for instance, is the oxidation of
1858:adding citations to reliable sources
1825:
1074:adding citations to reliable sources
1041:
746:adding citations to reliable sources
717:
647:
3729:Electrophilic aromatic substitution
2831:(11th ed.). Toronto: Pearson.
2735:
1591:metal. Common rust often refers to
24:
3696:Nucleophilic internal substitution
3686:Nucleophilic aromatic substitution
3552:
3228:Compendium of Chemical Terminology
3199:Compendium of Chemical Terminology
2938:(Seventh ed.). New York, NY.
2765:
2747:Haustein, Catherine Hinga (2014).
820:Each half-reaction has a standard
25:
4113:
3613:
3534:. Houghton Mifflin. p. 160.
2723:from the original on May 30, 2012
2154:in a biological system such as a
2105:nicotinamide adenine dinucleotide
1399:
613:Electronation and deelectronation
226:to another. For example, in the
204:change. Oxidation is the loss of
3348:The Chemistry of Submerged Soils
3145:Mailloux, Ryan J. (April 2015).
2753:The Gale Encyclopedia of Science
2197:
2111:, which drives the synthesis of
1909:
1896:
1830:
1171:The reduction reaction converts
1112:Illustration of a redox reaction
1046:
722:
651:
509:. Electron donors can also form
146:
106:
3852:Lindemann–Hinshelwood mechanism
3391:
3372:
3339:
3303:
3259:
3245:
3216:
3187:
3138:
3117:
3096:
3058:
3054:. Plenum Press. pp. 352–3.
3043:
3021:Oxidations in Organic Chemistry
3010:
2994:Reductions in Organic Chemistry
2985:
2974:
2902:
2695:(8th ed.). Prentice-Hall.
733:needs additional citations for
644:Rates, mechanisms, and energies
628:. These words are analogous to
3901:Outer sphere electron transfer
3896:Inner sphere electron transfer
3706:Nucleophilic acyl substitution
3233:Outer-sphere electron transfer
3204:Inner-sphere electron transfer
2845:
2780:
2759:
2749:"Oxidation-reduction reaction"
2709:
2645:
2623:
706:Analysis of bond energies and
701:outer sphere electron transfer
697:inner sphere electron transfer
254:
46:"gives" one outer electron to
13:
1:
4066:Diffusion-controlled reaction
3356:10.1016/S0065-2113(08)60633-1
3323:10.1002/14356007.a14_461.pub2
2657:Oxford Dictionaries | English
2616:
1530:, which consists of hydrated
490:
2911:Fundamentals of Biochemistry
2339:
1413:in the presence of an acid (
1328:In this type of reaction, a
864:where hydrogen is oxidized:
7:
3721:Electrophilic substitutions
3432:Robertson, William (2010).
3346:Ponnamperuma, F.N. (1972).
3163:10.1016/j.redox.2015.02.001
2773:Online Etymology Dictionary
2475:Chemical looping combustion
2442:
2390:"LEO the lion says GER " —
2346:List of chemistry mnemonics
2245:Třinec Iron and Steel Works
2119:perform similar functions.
1732:Redox reactions in industry
1389:And the copper is reduced:
1247:
1220:
1199:
1038:Examples of redox reactions
862:standard hydrogen electrode
314:
10:
4118:
4031:Energy profile (chemistry)
3993:More O'Ferrall–Jencks plot
3658:Nucleophilic substitutions
3406:. pp. 14-1 -- 14-24.
3253:"Bond Energy and Enthalpy"
2793:Pure and Applied Chemistry
2343:
2235:Redox reactions in geology
2121:
1822:Redox reactions in biology
1460:internal combustion engine
1260:
1237:
1215:
494:
318:
248:transfer of hydrogen atoms
29:
4061:Michaelis–Menten kinetics
4001:
3935:
3909:
3865:
3829:
3781:
3742:
3719:
3656:
3279:10.1515/9783110589771-005
2555:Oxidative phosphorylation
2432:galvanic or voltaic cells
1552:iron(III) oxide-hydroxide
1281:The overall reaction is:
1273:in a non-redox reaction:
1242:
609:) as sources of H atoms.
581:Hydride transfer reagents
511:charge transfer complexes
3988:Potential energy surface
3867:Electron/Proton transfer
3752:Unimolecular elimination
3017:Hudlický, Miloš (1990).
2992:Hudlický, Miloš (1996).
2862:. University of Maryland
2853:"Nitric Acid Fact Sheet"
2540:Nucleophilic abstraction
2505:Electron transport chain
2297:Redox reactions in soils
1482:produces carbon dioxide.
1116:In the reaction between
4036:Transition state theory
3837:Intramolecular reaction
3763:Bimolecular elimination
3588:Aquatic Redox Chemistry
3317:. Weinheim: Wiley-VCH.
3241:10.1351/goldbook.O04351
3212:10.1351/goldbook.I03052
3068:Modern Electrochemistry
3052:Modern Electrochemistry
1175:to the fluoride anion:
486:for oxidizing chemicals
50:, bonding them to form
3830:Unimolecular reactions
3791:Electrophilic addition
3496:Rodgers, Glen (2012).
2545:Organic redox reaction
2485:Electrochemical series
2288:+ 3 CO → 2 Fe + 3 CO
2248:
2113:adenosine triphosphate
1972:from the environment.
1961:
1697:(oxidation state +4).
1626:in the presence of an
1571:
1559:
1325:
1113:
487:
472:is a strong oxidizer.
98:
80:
77:potassium permanganate
55:
32:Redox (disambiguation)
4021:Rate-determining step
3953:Reactive intermediate
3811:Free-radical addition
3801:Nucleophilic addition
3744:Elimination reactions
3569:10013/epic.31694.d001
3435:More Chemistry Basics
2806:10.1515/pac-2018-0109
2450:Anaerobic respiration
2242:
2099:and the oxidation of
2001:and the reduction of
1956:
1565:
1521:
1514:Corrosion and rusting
1340:metal is placed in a
1307:
1111:
840:at equilibrium under
478:
86:
70:
42:
4016:Equilibrium constant
3596:10.1021/bk-2011-1071
3381:Advances in Agronomy
2182:for the function of
2172:beta-hydroxybutyrate
1976:Cellular respiration
1937:dehydroascorbic acid
1854:improve this section
1745:electrochemical cell
1669:+ 2 H → 2 Fe + 2 H
1310:electrochemical cell
1070:improve this section
856:considered, and the
846:electrochemical cell
742:improve this article
71:When a few drops of
30:For other uses, see
4102:Reaction mechanisms
4026:Reaction coordinate
3958:Radical (chemistry)
3943:Elementary reaction
3886:Grotthuss mechanism
3650:reaction mechanisms
2595:Reduction potential
2590:Reducing atmosphere
2530:Microbial fuel cell
2500:Electron equivalent
1737:Cathodic protection
1583:, the formation of
1434:+ 10 e + 12 H → N
1370:Zn + Cu → Zn + Cu
962:oxidation potential
842:standard conditions
822:electrode potential
708:ionization energies
190:oxidation–reduction
186:reduction–oxidation
89:reduction–oxidation
4092:Chemical reactions
4051:Arrhenius equation
3821:Oxidative addition
3783:Addition reactions
2663:on October 1, 2017
2525:Membrane potential
2428:electrolytic cells
2375:oss of electrons,
2249:
2243:Blast furnaces of
2231:or redox cycling.
2204:aromatic compounds
2202:Wide varieties of
2124:Membrane potential
1962:
1958:Enzymatic browning
1687:disproportionation
1681:Disproportionation
1572:
1560:
1342:copper(II) sulfate
1336:is deposited when
1326:
1300:Metal displacement
1114:
663:. You can help by
488:
426:), or else highly
99:
81:
56:
18:Chemical reduction
4074:
4073:
4046:Activated complex
4041:Activation energy
4003:Chemical kinetics
3948:Reaction dynamics
3847:Photodissociation
3605:978-0-8412-2652-4
3578:978-3-540-66528-1
3541:978-0-547-05405-6
3509:978-0-8400-6846-0
3475:978-0-02-828210-7
3445:978-1-936137-74-9
3413:978-1-4398-0305-9
3365:978-0-12-000724-0
3036:978-0-8412-1780-5
3003:978-0-8412-3344-7
2886:General Chemistry
2838:978-0-13-293128-1
2766:Harper, Douglas.
2717:"Redox Reactions"
2693:General Chemistry
2560:Partial oxidation
2480:Citric acid cycle
2470:Chemical equation
2302:Electron transfer
2089:Biological energy
1948:
1917:
1916:
1904:
1903:
1890:
1889:
1882:
1764:cleaning products
1749:sacrificial anode
1624:hydrogen peroxide
1487:organic chemistry
1405:The reduction of
1271:hydrogen fluoride
1265:
1264:
1106:
1105:
1098:
818:
817:
810:
792:
681:
680:
523:Antoine Lavoisier
336:electron acceptor
217:Electron-transfer
194:chemical reaction
68:
16:(Redirected from
4109:
3978:Collision theory
3927:Matrix isolation
3881:Harpoon reaction
3758:E1cB-elimination
3642:
3635:
3628:
3619:
3618:
3609:
3582:
3546:
3545:
3525:
3514:
3513:
3493:
3480:
3479:
3461:
3450:
3449:
3429:
3418:
3417:
3395:
3389:
3388:
3376:
3370:
3369:
3343:
3337:
3336:
3307:
3301:
3300:
3263:
3257:
3256:
3249:
3243:
3220:
3214:
3191:
3185:
3184:
3174:
3142:
3136:
3121:
3115:
3100:
3094:
3093:
3087:
3085:
3062:
3056:
3055:
3047:
3041:
3040:
3024:
3014:
3008:
3007:
2989:
2983:
2978:
2972:
2971:
2965:
2957:
2931:
2925:
2924:
2909:Jain JL (2004).
2906:
2900:
2899:
2881:
2872:
2871:
2869:
2867:
2857:
2849:
2843:
2842:
2824:
2811:
2810:
2808:
2784:
2778:
2777:
2763:
2757:
2756:
2744:
2733:
2732:
2730:
2728:
2713:
2707:
2706:
2688:
2673:
2672:
2670:
2668:
2659:. Archived from
2649:
2643:
2642:
2627:
2600:Thermic reaction
2510:Electrosynthesis
2490:Electrochemistry
2455:Bessemer process
2387:ain of electrons
2331:soil remediation
2292:
2247:, Czech Republic
2210:reduced to form
2084:
2039:
1920:
1913:
1906:
1900:
1893:
1885:
1878:
1874:
1871:
1865:
1834:
1826:
1779:electrosynthesis
1751:" to act as the
1724:
1715:+ 2 H → S + SO
1714:
1713:
1710:
1674:
1652:
1639:
1615:
1566:Iron rusting in
1554:(FeO(OH), Fe(OH)
1532:iron(III) oxides
1522:Oxides, such as
1458:, such as in an
1443:
1433:
1432:
1429:
1395:
1385:
1371:
1358:
1295:
1277:2 H + 2 F → 2 HF
1231:
1230:
1229:
1210:
1209:
1208:
1197:
1196:
1188:
1167:
1138:
1101:
1094:
1090:
1087:
1081:
1050:
1042:
1033:
1032:
1021:
1020:
1009:
1008:
990:
989:
978:
977:
956:
955:
944:
943:
932:
931:
906:
905:
901:
896:
895:
876:
875:
871:
852:reaction is the
835:
834:
813:
806:
802:
799:
793:
791:
750:
726:
718:
676:
673:
655:
648:
528:chemical species
425:
423:
422:
411:
409:
408:
400:
399:
388:
386:
385:
374:
372:
371:
360:
358:
357:
349:
348:
332:oxidizing agents
304:
302:
301:
291:
289:
288:
198:oxidation states
180:
175:
174:
171:
170:
167:
164:
161:
158:
155:
152:
140:
135:
134:
131:
130:
127:
124:
121:
118:
115:
112:
96:
69:
21:
4117:
4116:
4112:
4111:
4110:
4108:
4107:
4106:
4077:
4076:
4075:
4070:
4056:Eyring equation
3997:
3968:Stereochemistry
3931:
3917:Solvent effects
3905:
3861:
3825:
3806:
3796:
3777:
3772:
3738:
3734:
3715:
3711:
3701:
3691:
3681:
3671:
3652:
3646:
3616:
3606:
3579:
3555:
3553:Further reading
3550:
3549:
3542:
3526:
3517:
3510:
3494:
3483:
3476:
3462:
3453:
3446:
3430:
3421:
3414:
3396:
3392:
3377:
3373:
3366:
3344:
3340:
3333:
3308:
3304:
3289:
3265:
3264:
3260:
3251:
3250:
3246:
3221:
3217:
3192:
3188:
3143:
3139:
3122:
3118:
3101:
3097:
3083:
3081:
3079:
3063:
3059:
3048:
3044:
3037:
3015:
3011:
3004:
2990:
2986:
2979:
2975:
2959:
2958:
2946:
2932:
2928:
2921:
2907:
2903:
2896:
2882:
2875:
2865:
2863:
2855:
2851:
2850:
2846:
2839:
2825:
2814:
2785:
2781:
2764:
2760:
2745:
2736:
2726:
2724:
2715:
2714:
2710:
2703:
2689:
2676:
2666:
2664:
2651:
2650:
2646:
2629:
2628:
2624:
2619:
2614:
2605:Transmetalation
2575:Redox potential
2535:Murburn concept
2445:
2348:
2342:
2336:
2306:redox potential
2299:
2291:
2287:
2283:
2279:
2270:
2262:
2258:
2237:
2200:
2180:CoRR hypothesis
2150:, NAD/NADH and
2126:
2109:proton gradient
2083:
2079:
2075:
2071:
2063:
2059:
2055:
2037:
2033:
2029:
2025:
2021:
2017:
2013:
1999:
1993:
1989:
1985:
1964:Many essential
1951:
1950:
1949:
1934:
1886:
1875:
1869:
1866:
1851:
1835:
1824:
1734:
1722:
1718:
1711:
1708:
1707:
1705:
1701:
1683:
1672:
1668:
1664:
1660:
1650:
1646:
1642:
1637:
1614:
1610:
1606:
1602:
1593:iron(III) oxide
1557:
1549:
1541:
1537:
1524:iron(III) oxide
1516:
1503:carboxylic acid
1472:carbon monoxide
1441:
1437:
1430:
1427:
1426:
1424:
1415:denitrification
1402:
1394:Cu + 2 e → Cu
1393:
1384:Zn → Zn + 2 e
1383:
1369:
1356:
1352:
1348:
1323:
1319:
1302:
1293:
1289:
1285:
1255:
1251:
1243:
1228:
1225:
1224:
1223:
1221:
1207:
1204:
1203:
1202:
1200:
1183:
1179:
1158:
1154:
1136:
1132:
1128:
1102:
1091:
1085:
1082:
1067:
1051:
1040:
1031:
1028:
1027:
1026:
1019:
1016:
1015:
1014:
1007:
1004:
1003:
1002:
988:
985:
984:
983:
976:
973:
972:
971:
954:
951:
950:
949:
942:
939:
938:
937:
930:
927:
926:
925:
914:
910:
903:
899:
898:
894:
891:
890:
889:
880:
873:
869:
868:
833:
830:
829:
828:
814:
803:
797:
794:
751:
749:
739:
727:
716:
677:
671:
668:
661:needs expansion
646:
615:
608:
595:
588:
547:Electropositive
503:reducing agents
499:
493:
463:
456:
449:
442:
435:
430:elements (e.g.
428:electronegative
421:
418:
417:
416:
413:
407:
404:
403:
402:
398:
395:
394:
393:
390:
384:
381:
380:
379:
376:
370:
367:
366:
365:
362:
356:
353:
352:
351:
347:
344:
343:
342:
339:
323:
321:Oxidizing agent
317:
300:
298:
297:
296:
293:
287:
285:
284:
283:
280:
276:oxidizing agent
257:
246:are reduced by
192:) is a type of
178:
149:
145:
138:
109:
105:
92:
58:
52:sodium fluoride
35:
28:
23:
22:
15:
12:
11:
5:
4115:
4105:
4104:
4099:
4094:
4089:
4087:Soil chemistry
4072:
4071:
4069:
4068:
4063:
4058:
4053:
4048:
4043:
4038:
4033:
4028:
4023:
4018:
4013:
4007:
4005:
3999:
3998:
3996:
3995:
3990:
3985:
3980:
3975:
3970:
3965:
3960:
3955:
3950:
3945:
3939:
3937:
3936:Related topics
3933:
3932:
3930:
3929:
3924:
3919:
3913:
3911:
3910:Medium effects
3907:
3906:
3904:
3903:
3898:
3893:
3888:
3883:
3878:
3872:
3870:
3863:
3862:
3860:
3859:
3854:
3849:
3844:
3839:
3833:
3831:
3827:
3826:
3824:
3823:
3818:
3813:
3808:
3804:
3798:
3794:
3787:
3785:
3779:
3778:
3776:
3775:
3770:
3766:
3760:
3755:
3748:
3746:
3740:
3739:
3737:
3736:
3732:
3725:
3723:
3717:
3716:
3714:
3713:
3709:
3703:
3699:
3693:
3689:
3683:
3679:
3673:
3669:
3662:
3660:
3654:
3653:
3645:
3644:
3637:
3630:
3622:
3615:
3614:External links
3612:
3611:
3610:
3604:
3583:
3577:
3554:
3551:
3548:
3547:
3540:
3515:
3508:
3481:
3474:
3451:
3444:
3419:
3412:
3390:
3371:
3364:
3338:
3332:978-3527306732
3331:
3302:
3287:
3258:
3244:
3215:
3186:
3137:
3116:
3095:
3077:
3057:
3042:
3035:
3009:
3002:
2984:
2973:
2944:
2926:
2919:
2901:
2894:
2873:
2844:
2837:
2812:
2799:(4): 641–694.
2779:
2758:
2734:
2708:
2701:
2674:
2644:
2621:
2620:
2618:
2615:
2613:
2612:
2607:
2602:
2597:
2592:
2587:
2585:Reducing agent
2582:
2577:
2572:
2570:Redox gradient
2567:
2562:
2557:
2552:
2547:
2542:
2537:
2532:
2527:
2522:
2517:
2512:
2507:
2502:
2497:
2492:
2487:
2482:
2477:
2472:
2467:
2462:
2460:Bioremediation
2457:
2452:
2446:
2444:
2441:
2440:
2439:
2424:
2421:
2418:
2415:
2388:
2344:Main article:
2341:
2338:
2298:
2295:
2294:
2293:
2289:
2285:
2281:
2273:Blast furnaces
2268:
2260:
2256:
2236:
2233:
2199:
2196:
2137:microorganisms
2093:carbon dioxide
2086:
2085:
2081:
2077:
2073:
2069:
2061:
2057:
2049:Photosynthesis
2041:
2040:
2035:
2031:
2027:
2023:
2019:
2015:
1997:
1991:
1987:
1983:
1919:
1918:
1915:
1914:
1902:
1901:
1891:
1888:
1887:
1838:
1836:
1829:
1823:
1820:
1802:silver plating
1791:electroplating
1766:and oxidizing
1733:
1730:
1726:
1725:
1720:
1716:
1703:
1695:sulfur dioxide
1682:
1679:
1678:
1677:
1676:
1675:
1670:
1666:
1662:
1655:
1654:
1653:
1651:+ 2 e → 2 OH
1648:
1644:
1640:
1632:
1631:
1619:
1618:
1617:
1616:
1612:
1608:
1604:
1597:
1596:
1555:
1547:
1539:
1535:
1515:
1512:
1511:
1510:
1483:
1468:carbon dioxide
1447:
1446:
1445:
1444:
1439:
1435:
1419:
1418:
1401:
1400:Other examples
1398:
1397:
1396:
1387:
1386:
1377:half-reactions
1373:
1372:
1360:
1359:
1354:
1350:
1321:
1317:
1301:
1298:
1297:
1296:
1291:
1287:
1279:
1278:
1267:
1266:
1263:
1262:
1259:
1256:
1253:
1249:
1245:
1244:
1240:
1239:
1236:
1233:
1226:
1218:
1217:
1214:
1211:
1205:
1190:
1189:
1181:
1169:
1168:
1156:
1144:half-reactions
1140:
1139:
1134:
1130:
1104:
1103:
1054:
1052:
1045:
1039:
1036:
1035:
1034:
1029:
1017:
1005:
992:
991:
986:
974:
958:
957:
952:
940:
928:
912:
908:
892:
883:
882:
878:
831:
816:
815:
730:
728:
721:
715:
712:
679:
678:
658:
656:
645:
642:
619:electrochemist
614:
611:
606:
593:
586:
507:electron donor
497:Reducing agent
495:Main article:
492:
489:
461:
454:
447:
440:
433:
419:
405:
396:
382:
368:
354:
345:
319:Main article:
316:
313:
299:
286:
272:reducing agent
256:
253:
252:
251:
220:
26:
9:
6:
4:
3:
2:
4114:
4103:
4100:
4098:
4095:
4093:
4090:
4088:
4085:
4084:
4082:
4067:
4064:
4062:
4059:
4057:
4054:
4052:
4049:
4047:
4044:
4042:
4039:
4037:
4034:
4032:
4029:
4027:
4024:
4022:
4019:
4017:
4014:
4012:
4011:Rate equation
4009:
4008:
4006:
4004:
4000:
3994:
3991:
3989:
3986:
3984:
3983:Arrow pushing
3981:
3979:
3976:
3974:
3971:
3969:
3966:
3964:
3961:
3959:
3956:
3954:
3951:
3949:
3946:
3944:
3941:
3940:
3938:
3934:
3928:
3925:
3923:
3920:
3918:
3915:
3914:
3912:
3908:
3902:
3899:
3897:
3894:
3892:
3891:Marcus theory
3889:
3887:
3884:
3882:
3879:
3877:
3874:
3873:
3871:
3868:
3864:
3858:
3855:
3853:
3850:
3848:
3845:
3843:
3842:Isomerization
3840:
3838:
3835:
3834:
3832:
3828:
3822:
3819:
3817:
3816:Cycloaddition
3814:
3812:
3809:
3802:
3799:
3792:
3789:
3788:
3786:
3784:
3780:
3774:
3767:
3764:
3761:
3759:
3756:
3753:
3750:
3749:
3747:
3745:
3741:
3730:
3727:
3726:
3724:
3722:
3718:
3707:
3704:
3697:
3694:
3687:
3684:
3677:
3674:
3667:
3664:
3663:
3661:
3659:
3655:
3651:
3643:
3638:
3636:
3631:
3629:
3624:
3623:
3620:
3607:
3601:
3597:
3593:
3589:
3584:
3580:
3574:
3570:
3566:
3562:
3557:
3556:
3543:
3537:
3533:
3532:
3524:
3522:
3520:
3511:
3505:
3501:
3500:
3492:
3490:
3488:
3486:
3477:
3471:
3467:
3460:
3458:
3456:
3447:
3441:
3437:
3436:
3428:
3426:
3424:
3415:
3409:
3405:
3401:
3394:
3386:
3382:
3375:
3367:
3361:
3357:
3353:
3349:
3342:
3334:
3328:
3324:
3320:
3316:
3315:
3306:
3298:
3294:
3290:
3288:9783110588903
3284:
3280:
3276:
3272:
3268:
3262:
3254:
3248:
3242:
3238:
3234:
3230:
3229:
3224:
3219:
3213:
3209:
3205:
3201:
3200:
3195:
3190:
3182:
3178:
3173:
3168:
3164:
3160:
3156:
3152:
3151:Redox Biology
3148:
3141:
3135:
3131:
3130:0-9678550-9-8
3127:
3120:
3114:
3110:
3109:0-9678550-9-8
3106:
3099:
3092:
3080:
3078:9781461574675
3074:
3070:
3069:
3061:
3053:
3046:
3038:
3032:
3028:
3023:
3022:
3013:
3005:
2999:
2995:
2988:
2982:
2977:
2969:
2963:
2955:
2951:
2947:
2945:9781464126116
2941:
2937:
2930:
2922:
2920:81-219-2453-7
2916:
2912:
2905:
2897:
2895:0-03-072373-6
2891:
2887:
2880:
2878:
2861:
2854:
2848:
2840:
2834:
2830:
2823:
2821:
2819:
2817:
2807:
2802:
2798:
2794:
2790:
2783:
2775:
2774:
2769:
2762:
2754:
2750:
2743:
2741:
2739:
2722:
2719:. wiley.com.
2718:
2712:
2704:
2702:0-13-014329-4
2698:
2694:
2687:
2685:
2683:
2681:
2679:
2662:
2658:
2654:
2648:
2640:
2636:
2632:
2626:
2622:
2611:
2608:
2606:
2603:
2601:
2598:
2596:
2593:
2591:
2588:
2586:
2583:
2581:
2580:Redox therapy
2578:
2576:
2573:
2571:
2568:
2566:
2563:
2561:
2558:
2556:
2553:
2551:
2548:
2546:
2543:
2541:
2538:
2536:
2533:
2531:
2528:
2526:
2523:
2521:
2520:Hydrogenation
2518:
2516:
2515:Galvanic cell
2513:
2511:
2508:
2506:
2503:
2501:
2498:
2496:
2493:
2491:
2488:
2486:
2483:
2481:
2478:
2476:
2473:
2471:
2468:
2466:
2463:
2461:
2458:
2456:
2453:
2451:
2448:
2447:
2437:
2433:
2429:
2425:
2422:
2419:
2416:
2413:
2409:
2405:
2401:
2397:
2393:
2389:
2386:
2382:
2378:
2374:
2370:
2366:
2362:
2358:
2357:
2356:
2354:
2347:
2337:
2334:
2332:
2329:delineation,
2328:
2324:
2320:
2315:
2311:
2307:
2303:
2278:
2277:
2276:
2274:
2266:
2254:
2246:
2241:
2232:
2230:
2225:
2221:
2217:
2213:
2212:free radicals
2209:
2208:enzymatically
2205:
2198:Redox cycling
2195:
2193:
2189:
2185:
2181:
2177:
2173:
2169:
2165:
2161:
2157:
2153:
2149:
2144:
2142:
2138:
2134:
2130:
2125:
2120:
2118:
2114:
2110:
2106:
2102:
2098:
2094:
2090:
2067:
2054:
2053:
2052:
2050:
2046:
2012:
2011:
2010:
2008:
2004:
2000:
1981:
1977:
1973:
1971:
1967:
1959:
1955:
1946:
1942:
1941:oxidized form
1938:
1932:
1928:
1924:
1923:ascorbic acid
1912:
1908:
1907:
1899:
1895:
1894:
1884:
1881:
1873:
1870:December 2023
1863:
1859:
1855:
1849:
1848:
1844:
1839:This section
1837:
1833:
1828:
1827:
1819:
1817:
1814:
1810:
1809:galvanization
1806:
1803:
1799:
1796:
1795:chrome-plated
1792:
1788:
1784:
1780:
1775:
1773:
1769:
1765:
1760:
1758:
1754:
1750:
1746:
1742:
1738:
1729:
1700:
1699:
1698:
1696:
1692:
1688:
1659:
1658:
1656:
1641:
1638:Fe → Fe + e
1636:
1635:
1634:
1633:
1629:
1625:
1621:
1620:
1601:
1600:
1599:
1598:
1594:
1590:
1586:
1582:
1578:
1574:
1573:
1569:
1564:
1553:
1545:
1533:
1529:
1525:
1520:
1508:
1505:, and then a
1504:
1500:
1496:
1492:
1488:
1484:
1481:
1477:
1473:
1469:
1465:
1461:
1457:
1453:
1449:
1448:
1423:
1422:
1421:
1420:
1416:
1412:
1408:
1404:
1403:
1392:
1391:
1390:
1382:
1381:
1380:
1378:
1368:
1367:
1366:
1363:
1357:(aq) + Cu (s)
1353:(aq) → ZnSO
1349:Zn (s) + CuSO
1347:
1346:
1345:
1343:
1339:
1335:
1331:
1315:
1314:Galvanic cell
1311:
1306:
1284:
1283:
1282:
1276:
1275:
1274:
1272:
1257:
1246:
1241:
1234:
1219:
1212:
1198:
1195:
1194:
1193:
1187:
1178:
1177:
1176:
1174:
1166:
1162:
1153:
1152:
1151:
1149:
1145:
1127:
1126:
1125:
1123:
1119:
1110:
1100:
1097:
1089:
1086:December 2023
1079:
1075:
1071:
1065:
1064:
1060:
1055:This section
1053:
1049:
1044:
1043:
1025:
1013:
1001:
998:
997:
996:
982:
970:
967:
966:
965:
963:
948:
936:
924:
921:
920:
919:
916:
888:
867:
866:
865:
863:
859:
855:
854:half-reaction
851:
848:in which the
847:
843:
839:
827:
823:
812:
809:
801:
798:December 2023
790:
787:
783:
780:
776:
773:
769:
766:
762:
759: –
758:
754:
753:Find sources:
747:
743:
737:
736:
731:This section
729:
725:
720:
719:
711:
709:
704:
702:
698:
692:
690:
686:
675:
666:
662:
659:This section
657:
654:
650:
649:
641:
639:
635:
634:deprotonation
631:
627:
623:
620:
610:
604:
601:compounds to
600:
596:
589:
582:
578:
576:
572:
568:
564:
560:
556:
552:
548:
543:
541:
537:
533:
529:
524:
520:
516:
512:
508:
504:
498:
485:
482:
481:international
477:
473:
471:
466:
464:
457:
450:
443:
436:
429:
424:
410:
387:
373:
359:
337:
333:
328:
322:
312:
310:
309:
308:half-reaction
303:
290:
277:
273:
267:
264:
262:
259:"Redox" is a
249:
245:
242:, bonds like
241:
240:hydrogenation
237:
233:
229:
225:
221:
218:
215:
214:
213:
210:
207:
203:
199:
196:in which the
195:
191:
187:
183:
182:
173:
143:
142:
133:
103:
95:
90:
87:Example of a
85:
78:
74:
53:
49:
45:
41:
37:
33:
19:
3963:Molecularity
3875:
3587:
3560:
3530:
3498:
3465:
3434:
3399:
3393:
3384:
3380:
3374:
3347:
3341:
3312:
3305:
3270:
3261:
3247:
3226:
3218:
3197:
3189:
3154:
3150:
3140:
3119:
3098:
3089:
3082:. Retrieved
3067:
3060:
3051:
3045:
3020:
3012:
2993:
2987:
2976:
2935:
2929:
2913:. S. Chand.
2910:
2904:
2885:
2866:February 12,
2864:. Retrieved
2859:
2847:
2828:
2796:
2792:
2782:
2771:
2761:
2752:
2725:. Retrieved
2711:
2692:
2665:. Retrieved
2661:the original
2656:
2647:
2634:
2625:
2610:Sulfur cycle
2495:Electrolysis
2465:Calvin cycle
2411:
2410:lectrons is
2407:
2403:
2399:
2398:lectrons is
2395:
2391:
2384:
2380:
2376:
2372:
2368:
2364:
2349:
2335:
2323:free radical
2300:
2250:
2229:futile cycle
2216:flavoenzymes
2201:
2192:chloroplasts
2188:mitochondria
2176:acetoacetate
2145:
2135:and killing
2129:Free radical
2127:
2117:mitochondria
2087:
2066:light energy
2042:
1974:
1963:
1927:reduced form
1876:
1867:
1852:Please help
1840:
1776:
1761:
1735:
1727:
1684:
1543:
1456:hydrocarbons
1388:
1374:
1364:
1361:
1327:
1280:
1268:
1191:
1184:+ 2 e → 2
1170:
1141:
1115:
1092:
1083:
1068:Please help
1056:
1023:
1018:red(cathode)
1011:
999:
993:
980:
968:
961:
959:
946:
934:
922:
917:
886:
884:
825:
819:
804:
795:
785:
778:
771:
764:
752:
740:Please help
735:verification
732:
705:
693:
682:
669:
665:adding to it
660:
622:John Bockris
616:
579:
544:
536:biochemistry
500:
467:
324:
306:
268:
265:
258:
211:
189:
185:
101:
100:
93:
88:
36:
3922:Cage effect
3857:RRKM theory
3773:elimination
3157:: 381–398.
2565:Pro-oxidant
2325:chemistry,
2319:pedogenesis
2141:antioxidant
2133:homeostasis
1970:assimilated
1813:gold-plated
1772:nitric acid
1770:to produce
1691:thiosulfate
1585:iron oxides
1474:, and heat
1462:, produces
630:protonation
540:hydride ion
519:metal oxide
470:Nitric acid
261:portmanteau
255:Terminology
4081:Categories
3387:: 151–208.
2617:References
2436:fuel cells
2434:, such as
2402:xidation,
2314:paddy rice
2224:superoxide
2218:and their
2152:NADP/NADPH
2122:See also:
2038:O + Energy
1966:biological
1798:automotive
1757:galvanized
1603:4 Fe + 3 O
1452:combustion
1344:solution:
1261:2 H + 2 F
1216:2 H + 2 e
768:newspapers
672:April 2023
626:electrodes
583:, such as
553:, such as
549:elemental
517:such as a
491:Reductants
327:substances
3973:Catalysis
3869:reactions
3531:Chemistry
3404:CRC Press
3297:242013948
3091:reaction.
3084:March 29,
2962:cite book
2954:986827885
2379:eduction
2367:xidation
2353:mnemonics
2340:Mnemonics
2253:magnetite
2220:coenzymes
1945:Vitamin C
1931:Vitamin C
1841:does not
1577:corrosion
1575:The term
1324:solution.
1312:like the
1057:does not
1030:ox(anode)
575:aluminium
563:magnesium
484:pictogram
224:substrate
206:electrons
202:reactants
3181:25744690
2721:Archived
2639:Archived
2635:Bitesize
2631:"Metals"
2443:See also
2414:eduction
2168:pyruvate
2148:GSH/GSSG
1935:Bottom:
1787:smelting
1781:. Metal
1661:2 Fe + H
1507:peroxide
1495:aldehyde
1411:nitrogen
1173:fluorine
1122:fluorine
1118:hydrogen
603:alcohols
599:carbonyl
532:electron
315:Oxidants
97:mnemonic
73:glycerol
48:fluorine
3172:4348434
2768:"redox"
2667:May 15,
2637:. BBC.
2406:ain of
2394:oss of
2361:OIL RIG
2327:wetland
2310:voltage
2164:lactate
2030:→ 6 CO
1980:glucose
1862:removed
1847:sources
1816:jewelry
1805:cutlery
1800:parts,
1768:ammonia
1741:cathode
1607:→ 2 Fe
1581:Rusting
1491:alcohol
1407:nitrate
1375:As two
1294:→ 2 HF
1148:protons
1137:→ 2 HF
1078:removed
1063:sources
941:cathode
902:⁄
881:→ H + e
872:⁄
850:cathode
838:voltage
782:scholar
757:"Redox"
555:lithium
228:rusting
200:of the
94:OIL RIG
3648:Basic
3602:
3575:
3538:
3506:
3472:
3442:
3410:
3362:
3329:
3295:
3285:
3179:
3169:
3128:
3107:
3075:
3033:
3000:
2952:
2942:
2917:
2892:
2835:
2727:May 9,
2699:
2265:rutile
2174:, and
2097:sugars
2003:oxygen
1743:of an
1568:pyrite
1550:O and
1499:ketone
1480:carbon
1476:energy
1334:copper
1232:+ 2 e
844:of an
784:
777:
770:
763:
755:
573:, and
559:sodium
551:metals
44:Sodium
4097:Redox
3876:Redox
3712:Acyl)
3293:S2CID
3223:IUPAC
3194:IUPAC
2856:(PDF)
2160:organ
2101:water
2095:into
2080:+ 6 O
2060:+ 6 H
2034:+ 6 H
2026:+ 6 O
2007:water
1994:) to
1921:Top:
1753:anode
1570:cubes
1497:or a
1493:, an
1464:water
1438:+ 6 H
1330:metal
1159:→ 2
953:anode
860:is a
858:anode
789:JSTOR
775:books
638:IUPAC
592:LiAlH
236:oxide
181:-doks
102:Redox
3765:(E2)
3754:(E1)
3600:ISBN
3573:ISBN
3536:ISBN
3504:ISBN
3470:ISBN
3440:ISBN
3408:ISBN
3360:ISBN
3327:ISBN
3283:ISBN
3177:PMID
3126:ISBN
3105:ISBN
3086:2020
3073:ISBN
3031:ISBN
2998:ISBN
2968:link
2950:OCLC
2940:ISBN
2915:ISBN
2890:ISBN
2868:2024
2833:ISBN
2729:2012
2697:ISBN
2669:2017
2363:" —
2267:(TiO
2206:are
2190:and
2166:and
2156:cell
2068:→ C
2064:O +
2056:6 CO
1845:any
1843:cite
1811:and
1783:ores
1628:acid
1589:iron
1528:rust
1501:, a
1450:The
1425:2 NO
1338:zinc
1238:2 F
1163:+ 2
1120:and
1061:any
1059:cite
1006:cell
929:cell
832:cell
761:news
699:and
689:fuel
685:rust
632:and
617:The
590:and
585:NaBH
571:zinc
567:iron
479:The
232:iron
141:-oks
3735:Ar)
3692:Ar)
3592:doi
3565:hdl
3352:doi
3319:doi
3275:doi
3237:doi
3235:".
3208:doi
3206:".
3167:PMC
3159:doi
3027:456
2801:doi
2255:(Fe
2186:in
2184:DNA
2158:or
2045:NAD
2005:to
1943:of
1929:of
1856:by
1719:+ H
1526:or
1454:of
1409:to
1290:+ F
1252:+ F
1133:+ F
1072:by
987:red
979:= –
893:red
744:by
667:.
515:ore
415:OsO
378:CrO
364:MnO
244:C=C
230:of
188:or
179:REE
139:RED
4083::
3803:(A
3793:(A
3731:(S
3708:(S
3702:i)
3698:(S
3688:(S
3682:2)
3678:(S
3672:1)
3668:(S
3598:.
3571:.
3518:^
3484:^
3454:^
3422:^
3385:39
3383:.
3358:.
3325:.
3291:.
3281:.
3269:.
3225:,
3196:,
3175:.
3165:.
3153:.
3149:.
3132:.
3111:.
3088:.
3029:.
2964:}}
2960:{{
2948:.
2876:^
2858:.
2815:^
2797:92
2795:.
2791:.
2770:.
2737:^
2677:^
2655:.
2633:.
2383:s
2371:s
2280:Fe
2194:.
2170:,
2143:.
2074:12
2020:12
1996:CO
1988:12
1982:(C
1818:.
1807:,
1774:.
1709:2−
1685:A
1534:Fe
1466:,
1417:):
1258:→
1235:→
1213:→
1150::
1022:+
1010:=
975:ox
964::
945:–
933:=
703:.
569:,
565:,
561:,
557:,
542:.
458:,
453:Br
451:,
446:Cl
444:,
437:,
412:,
392:Cr
389:,
375:,
361:,
295:Fe
292:/
282:Fe
184:,
157:iː
144:,
3807:)
3805:N
3797:)
3795:E
3771:i
3769:E
3733:E
3710:N
3700:N
3690:N
3680:N
3670:N
3641:e
3634:t
3627:v
3608:.
3594::
3581:.
3567::
3544:.
3512:.
3478:.
3448:.
3416:.
3368:.
3354::
3335:.
3321::
3299:.
3277::
3255:.
3239::
3210::
3183:.
3161::
3155:4
3039:.
3006:.
2970:)
2956:.
2923:.
2898:.
2870:.
2841:.
2809:.
2803::
2776:.
2731:.
2705:.
2671:.
2412:r
2408:e
2404:g
2400:o
2396:e
2392:l
2385:g
2381:i
2377:r
2373:l
2369:i
2365:o
2359:"
2290:2
2286:3
2284:O
2282:2
2269:2
2261:4
2259:O
2257:3
2082:2
2078:6
2076:O
2072:H
2070:6
2062:2
2058:2
2036:2
2032:2
2028:2
2024:6
2022:O
2018:H
2016:6
2014:C
1998:2
1992:6
1990:O
1986:H
1984:6
1947:)
1939:(
1933:)
1925:(
1883:)
1877:(
1872:)
1868:(
1864:.
1850:.
1723:O
1721:2
1717:2
1712:3
1706:O
1704:2
1702:S
1673:O
1671:2
1667:2
1665:O
1663:2
1649:2
1647:O
1645:2
1643:H
1630::
1613:3
1611:O
1609:2
1605:2
1556:3
1548:2
1546:H
1544:n
1542:·
1540:3
1538:O
1536:2
1509:.
1442:O
1440:2
1436:2
1431:3
1428:−
1355:4
1351:4
1322:4
1318:4
1292:2
1288:2
1286:H
1254:2
1250:2
1248:H
1227:2
1222:F
1206:2
1201:H
1186:F
1182:2
1180:F
1165:e
1161:H
1157:2
1155:H
1135:2
1131:2
1129:H
1099:)
1093:(
1088:)
1084:(
1080:.
1066:.
1024:E
1012:E
1000:E
981:E
969:E
947:E
935:E
923:E
913:2
909:2
907:H
904:2
900:1
887:E
879:2
877:H
874:2
870:1
826:E
824:(
811:)
805:(
800:)
796:(
786:·
779:·
772:·
765:·
738:.
674:)
670:(
607:2
594:4
587:4
462:2
460:I
455:2
448:2
441:2
439:F
434:2
432:O
420:4
406:7
401:O
397:2
383:3
369:4
355:4
350:O
346:2
341:N
250:.
172:/
169:s
166:k
163:ɒ
160:d
154:r
151:ˈ
148:/
132:/
129:s
126:k
123:ɒ
120:d
117:ɛ
114:r
111:ˈ
108:/
104:(
34:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.